US20050263900A1 - Semiconductor device having silicon carbide and conductive pathway interface - Google Patents
Semiconductor device having silicon carbide and conductive pathway interface Download PDFInfo
- Publication number
- US20050263900A1 US20050263900A1 US11/169,337 US16933705A US2005263900A1 US 20050263900 A1 US20050263900 A1 US 20050263900A1 US 16933705 A US16933705 A US 16933705A US 2005263900 A1 US2005263900 A1 US 2005263900A1
- Authority
- US
- United States
- Prior art keywords
- layer
- plasma
- interface
- copper
- semiconductor device
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 title claims description 6
- 229910010271 silicon carbide Inorganic materials 0.000 title claims description 6
- 239000004065 semiconductor Substances 0.000 title claims 7
- 230000037361 pathway Effects 0.000 title claims 4
- 239000010949 copper Substances 0.000 claims abstract description 33
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims abstract description 32
- 229910052802 copper Inorganic materials 0.000 claims abstract description 32
- 230000004888 barrier function Effects 0.000 claims description 9
- 230000003647 oxidation Effects 0.000 claims description 9
- 238000007254 oxidation reaction Methods 0.000 claims description 9
- 239000010936 titanium Substances 0.000 claims description 3
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims description 3
- 229910052721 tungsten Inorganic materials 0.000 claims description 3
- 239000010937 tungsten Substances 0.000 claims description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims 2
- 229910052715 tantalum Inorganic materials 0.000 claims 2
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims 2
- 229910052719 titanium Inorganic materials 0.000 claims 2
- 238000009832 plasma treatment Methods 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 63
- 230000008569 process Effects 0.000 abstract description 57
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 abstract description 29
- 238000000151 deposition Methods 0.000 abstract description 20
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 abstract description 19
- 239000001301 oxygen Substances 0.000 abstract description 19
- 229910052760 oxygen Inorganic materials 0.000 abstract description 19
- 229910021529 ammonia Inorganic materials 0.000 abstract description 14
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 abstract description 12
- 239000000356 contaminant Substances 0.000 abstract description 12
- 238000011065 in-situ storage Methods 0.000 abstract description 12
- 238000004140 cleaning Methods 0.000 abstract description 11
- 230000009977 dual effect Effects 0.000 abstract description 11
- 239000001257 hydrogen Substances 0.000 abstract description 6
- 229910052739 hydrogen Inorganic materials 0.000 abstract description 6
- 229910052757 nitrogen Inorganic materials 0.000 abstract description 6
- 150000001875 compounds Chemical class 0.000 abstract description 4
- 125000004435 hydrogen atom Chemical class [H]* 0.000 abstract 1
- 239000000758 substrate Substances 0.000 description 35
- 229910052751 metal Inorganic materials 0.000 description 23
- 239000002184 metal Substances 0.000 description 23
- 239000003638 chemical reducing agent Substances 0.000 description 21
- 239000004020 conductor Substances 0.000 description 16
- 150000004767 nitrides Chemical class 0.000 description 12
- 239000000463 material Substances 0.000 description 10
- 238000005229 chemical vapour deposition Methods 0.000 description 9
- 239000007789 gas Substances 0.000 description 9
- 229910052782 aluminium Inorganic materials 0.000 description 6
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 6
- 230000008021 deposition Effects 0.000 description 6
- 230000009467 reduction Effects 0.000 description 6
- QPLDLSVMHZLSFG-UHFFFAOYSA-N Copper oxide Chemical compound [Cu]=O QPLDLSVMHZLSFG-UHFFFAOYSA-N 0.000 description 5
- 239000005751 Copper oxide Substances 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 229910000431 copper oxide Inorganic materials 0.000 description 5
- 238000005516 engineering process Methods 0.000 description 5
- 230000000977 initiatory effect Effects 0.000 description 5
- 150000002500 ions Chemical class 0.000 description 5
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 230000027455 binding Effects 0.000 description 3
- 238000011109 contamination Methods 0.000 description 3
- -1 copper oxide (Cu2O) Chemical class 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000005137 deposition process Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 238000005530 etching Methods 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 238000000059 patterning Methods 0.000 description 3
- 229920002120 photoresistant polymer Polymers 0.000 description 3
- 238000005240 physical vapour deposition Methods 0.000 description 3
- 238000000623 plasma-assisted chemical vapour deposition Methods 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- 239000012159 carrier gas Substances 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000009713 electroplating Methods 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 238000001465 metallisation Methods 0.000 description 2
- 238000002161 passivation Methods 0.000 description 2
- 238000011946 reduction process Methods 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 238000002230 thermal chemical vapour deposition Methods 0.000 description 2
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- BERDEBHAJNAUOM-UHFFFAOYSA-N copper(I) oxide Inorganic materials [Cu]O[Cu] BERDEBHAJNAUOM-UHFFFAOYSA-N 0.000 description 1
- LBJNMUFDOHXDFG-UHFFFAOYSA-N copper;hydrate Chemical compound O.[Cu].[Cu] LBJNMUFDOHXDFG-UHFFFAOYSA-N 0.000 description 1
- KRFJLUBVMFXRPN-UHFFFAOYSA-N cuprous oxide Chemical compound [O-2].[Cu+].[Cu+] KRFJLUBVMFXRPN-UHFFFAOYSA-N 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 239000003989 dielectric material Substances 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 238000010849 ion bombardment Methods 0.000 description 1
- 230000002045 lasting effect Effects 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 229910000069 nitrogen hydride Inorganic materials 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 238000001020 plasma etching Methods 0.000 description 1
- 239000005368 silicate glass Substances 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- LIVNPJMFVYWSIS-UHFFFAOYSA-N silicon monoxide Chemical class [Si-]#[O+] LIVNPJMFVYWSIS-UHFFFAOYSA-N 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 230000009870 specific binding Effects 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/31—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to form insulating layers thereon, e.g. for masking or by using photolithographic techniques; After treatment of these layers; Selection of materials for these layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B08—CLEANING
- B08B—CLEANING IN GENERAL; PREVENTION OF FOULING IN GENERAL
- B08B7/00—Cleaning by methods not provided for in a single other subclass or a single group in this subclass
- B08B7/0035—Cleaning by methods not provided for in a single other subclass or a single group in this subclass by radiant energy, e.g. UV, laser, light beam or the like
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C16/00—Chemical coating by decomposition of gaseous compounds, without leaving reaction products of surface material in the coating, i.e. chemical vapour deposition [CVD] processes
- C23C16/02—Pretreatment of the material to be coated
- C23C16/0227—Pretreatment of the material to be coated by cleaning or etching
- C23C16/0245—Pretreatment of the material to be coated by cleaning or etching by etching with a plasma
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23G—CLEANING OR DE-GREASING OF METALLIC MATERIAL BY CHEMICAL METHODS OTHER THAN ELECTROLYSIS
- C23G5/00—Cleaning or de-greasing metallic material by other methods; Apparatus for cleaning or de-greasing metallic material with organic solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02041—Cleaning
- H01L21/02057—Cleaning during device manufacture
- H01L21/0206—Cleaning during device manufacture during, before or after processing of insulating layers
- H01L21/02063—Cleaning during device manufacture during, before or after processing of insulating layers the processing being the formation of vias or contact holes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02041—Cleaning
- H01L21/02057—Cleaning during device manufacture
- H01L21/02068—Cleaning during device manufacture during, before or after processing of conductive layers, e.g. polysilicon or amorphous silicon layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02041—Cleaning
- H01L21/02057—Cleaning during device manufacture
- H01L21/02068—Cleaning during device manufacture during, before or after processing of conductive layers, e.g. polysilicon or amorphous silicon layers
- H01L21/02074—Cleaning during device manufacture during, before or after processing of conductive layers, e.g. polysilicon or amorphous silicon layers the processing being a planarization of conductive layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02109—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates
- H01L21/02112—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer
- H01L21/02123—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon
- H01L21/0217—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon the material being a silicon nitride not containing oxygen, e.g. SixNy or SixByNz
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02109—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates
- H01L21/02205—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates the layer being characterised by the precursor material for deposition
- H01L21/02208—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates the layer being characterised by the precursor material for deposition the precursor containing a compound comprising Si
- H01L21/02211—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates the layer being characterised by the precursor material for deposition the precursor containing a compound comprising Si the compound being a silane, e.g. disilane, methylsilane or chlorosilane
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02225—Forming insulating materials on a substrate characterised by the process for the formation of the insulating layer
- H01L21/0226—Forming insulating materials on a substrate characterised by the process for the formation of the insulating layer formation by a deposition process
- H01L21/02263—Forming insulating materials on a substrate characterised by the process for the formation of the insulating layer formation by a deposition process deposition from the gas or vapour phase
- H01L21/02271—Forming insulating materials on a substrate characterised by the process for the formation of the insulating layer formation by a deposition process deposition from the gas or vapour phase deposition by decomposition or reaction of gaseous or vapour phase compounds, i.e. chemical vapour deposition
- H01L21/02274—Forming insulating materials on a substrate characterised by the process for the formation of the insulating layer formation by a deposition process deposition from the gas or vapour phase deposition by decomposition or reaction of gaseous or vapour phase compounds, i.e. chemical vapour deposition in the presence of a plasma [PECVD]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02296—Forming insulating materials on a substrate characterised by the treatment performed before or after the formation of the layer
- H01L21/02318—Forming insulating materials on a substrate characterised by the treatment performed before or after the formation of the layer post-treatment
- H01L21/02337—Forming insulating materials on a substrate characterised by the treatment performed before or after the formation of the layer post-treatment treatment by exposure to a gas or vapour
- H01L21/0234—Forming insulating materials on a substrate characterised by the treatment performed before or after the formation of the layer post-treatment treatment by exposure to a gas or vapour treatment by exposure to a plasma
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/31—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to form insulating layers thereon, e.g. for masking or by using photolithographic techniques; After treatment of these layers; Selection of materials for these layers
- H01L21/3105—After-treatment
- H01L21/311—Etching the insulating layers by chemical or physical means
- H01L21/31105—Etching inorganic layers
- H01L21/31111—Etching inorganic layers by chemical means
- H01L21/31116—Etching inorganic layers by chemical means by dry-etching
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/31—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to form insulating layers thereon, e.g. for masking or by using photolithographic techniques; After treatment of these layers; Selection of materials for these layers
- H01L21/314—Inorganic layers
- H01L21/3148—Silicon Carbide layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/31—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to form insulating layers thereon, e.g. for masking or by using photolithographic techniques; After treatment of these layers; Selection of materials for these layers
- H01L21/314—Inorganic layers
- H01L21/318—Inorganic layers composed of nitrides
- H01L21/3185—Inorganic layers composed of nitrides of siliconnitrides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/31—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to form insulating layers thereon, e.g. for masking or by using photolithographic techniques; After treatment of these layers; Selection of materials for these layers
- H01L21/3205—Deposition of non-insulating-, e.g. conductive- or resistive-, layers on insulating layers; After-treatment of these layers
- H01L21/321—After treatment
- H01L21/3213—Physical or chemical etching of the layers, e.g. to produce a patterned layer from a pre-deposited extensive layer
- H01L21/32133—Physical or chemical etching of the layers, e.g. to produce a patterned layer from a pre-deposited extensive layer by chemical means only
- H01L21/32135—Physical or chemical etching of the layers, e.g. to produce a patterned layer from a pre-deposited extensive layer by chemical means only by vapour etching only
- H01L21/32136—Physical or chemical etching of the layers, e.g. to produce a patterned layer from a pre-deposited extensive layer by chemical means only by vapour etching only using plasmas
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/70—Manufacture or treatment of devices consisting of a plurality of solid state components formed in or on a common substrate or of parts thereof; Manufacture of integrated circuit devices or of parts thereof
- H01L21/71—Manufacture of specific parts of devices defined in group H01L21/70
- H01L21/768—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics
- H01L21/76801—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing
- H01L21/76829—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing characterised by the formation of thin functional dielectric layers, e.g. dielectric etch-stop, barrier, capping or liner layers
- H01L21/76834—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing characterised by the formation of thin functional dielectric layers, e.g. dielectric etch-stop, barrier, capping or liner layers formation of thin insulating films on the sidewalls or on top of conductors
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/70—Manufacture or treatment of devices consisting of a plurality of solid state components formed in or on a common substrate or of parts thereof; Manufacture of integrated circuit devices or of parts thereof
- H01L21/71—Manufacture of specific parts of devices defined in group H01L21/70
- H01L21/768—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics
- H01L21/76838—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the conductors
- H01L21/76877—Filling of holes, grooves or trenches, e.g. vias, with conductive material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/70—Manufacture or treatment of devices consisting of a plurality of solid state components formed in or on a common substrate or of parts thereof; Manufacture of integrated circuit devices or of parts thereof
- H01L21/71—Manufacture of specific parts of devices defined in group H01L21/70
- H01L21/768—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics
- H01L21/76838—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the conductors
- H01L21/76877—Filling of holes, grooves or trenches, e.g. vias, with conductive material
- H01L21/76883—Post-treatment or after-treatment of the conductive material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/52—Arrangements for conducting electric current within the device in operation from one component to another, i.e. interconnections, e.g. wires, lead frames
- H01L23/522—Arrangements for conducting electric current within the device in operation from one component to another, i.e. interconnections, e.g. wires, lead frames including external interconnections consisting of a multilayer structure of conductive and insulating layers inseparably formed on the semiconductor body
- H01L23/532—Arrangements for conducting electric current within the device in operation from one component to another, i.e. interconnections, e.g. wires, lead frames including external interconnections consisting of a multilayer structure of conductive and insulating layers inseparably formed on the semiconductor body characterised by the materials
- H01L23/53204—Conductive materials
- H01L23/53209—Conductive materials based on metals, e.g. alloys, metal silicides
- H01L23/53228—Conductive materials based on metals, e.g. alloys, metal silicides the principal metal being copper
- H01L23/53238—Additional layers associated with copper layers, e.g. adhesion, barrier, cladding layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02041—Cleaning
- H01L21/02057—Cleaning during device manufacture
- H01L21/02068—Cleaning during device manufacture during, before or after processing of conductive layers, e.g. polysilicon or amorphous silicon layers
- H01L21/02071—Cleaning during device manufacture during, before or after processing of conductive layers, e.g. polysilicon or amorphous silicon layers the processing being a delineation, e.g. RIE, of conductive layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02109—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates
- H01L21/02112—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer
- H01L21/02115—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material being carbon, e.g. alpha-C, diamond or hydrogen doped carbon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02109—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates
- H01L21/02112—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer
- H01L21/02123—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon
- H01L21/02164—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon the material being a silicon oxide, e.g. SiO2
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/02104—Forming layers
- H01L21/02107—Forming insulating materials on a substrate
- H01L21/02109—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates
- H01L21/02112—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer
- H01L21/02123—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon
- H01L21/02167—Forming insulating materials on a substrate characterised by the type of layer, e.g. type of material, porous/non-porous, pre-cursors, mixtures or laminates characterised by the material of the layer the material containing silicon the material being a silicon carbide not containing oxygen, e.g. SiC, SiC:H or silicon carbonitrides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/70—Manufacture or treatment of devices consisting of a plurality of solid state components formed in or on a common substrate or of parts thereof; Manufacture of integrated circuit devices or of parts thereof
- H01L21/71—Manufacture of specific parts of devices defined in group H01L21/70
- H01L21/768—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics
- H01L21/76801—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing
- H01L21/76802—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing by forming openings in dielectrics
- H01L21/76807—Applying interconnections to be used for carrying current between separate components within a device comprising conductors and dielectrics characterised by the formation and the after-treatment of the dielectrics, e.g. smoothing by forming openings in dielectrics for dual damascene structures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L2924/00—Indexing scheme for arrangements or methods for connecting or disconnecting semiconductor or solid-state bodies as covered by H01L24/00
- H01L2924/0001—Technical content checked by a classifier
- H01L2924/0002—Not covered by any one of groups H01L24/00, H01L24/00 and H01L2224/00
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L2924/00—Indexing scheme for arrangements or methods for connecting or disconnecting semiconductor or solid-state bodies as covered by H01L24/00
- H01L2924/30—Technical effects
- H01L2924/301—Electrical effects
- H01L2924/3011—Impedance
Definitions
- the present invention relates generally to the fabrication of integrated circuits on substrates. More particularly, the invention relates to a method of reducing oxides on a substrate prior to depositing a layer thereover in the fabrication process.
- VLSI very large scale integration
- ULSI ultra large scale integration
- the multilevel interconnects that lie at the heart of this technology require careful processing of high aspect ratio features, such as vias and other interconnects. Reliable formation of these interconnects is very important to the VLSI and ULSI success and to the continued effort to increase circuit density and quality of individual substrates and die.
- CVD chemical vapor deposition
- PVD physical vapor deposition
- electroplating techniques are used to deposit electrically conductive material into the contacts, vias, lines, or other features formed on the substrate. Considerable effort has focused on reliably depositing material in these high aspect ratio, smaller interconnects.
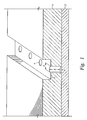
- FIG. 1 shows a substrate 2 with a via 4 formed within an electrically insulative or dielectric layer 6 .
- the aspect ratio has increased to approximately 5:1 for the height to width ratio, shown in FIG. 1 as x with respect to d.
- this attention to improved cleaning is due to a desired change in the conductor metal.
- copper is now being considered as an interconnect material in place of aluminum, because copper has a lower resistivity (1.7 ⁇ -cm compared to 3.1 ⁇ -cm for aluminum) and higher current carrying capacity.
- copper is highly susceptible to oxidation. With copper depositions, oxidation is considered a detriment and interferes with adhesion on the adjacent layer, affects conductivity of the copper feature, and reduces the reliability of the overall circuit.
- present processes utilize oxygen for a variety of reasons in some instances and in other instances, oxygen is a byproduct of the reactions. Thus, even carefully controlled environments may contain oxygen that may oxidize copper or other conductive materials, such as aluminum, to the detriment of the circuit.
- Copper has other difficulties. Because copper is difficult to etch in a precise pattern, traditional deposition/etch processes for forming interconnects has become unworkable, and accordingly, a “dual damascene” structure is being used for copper interconnects. In a typical dual damascene structure, the dielectric layer is etched to define both the contacts/vias and the interconnect lines. Metal is then inlaid into the defined pattern and any excess metal is typically removed from the top of the structure in a planarization process, such as CMP. This complex approach increases the importance of obtaining properly cleaned surfaces within the interconnects.
- an inert gas plasma such as an Argon (Ar) plasma
- Ar Argon
- interconnects and metal layers such as aluminum and copper
- ions were attracted to the substrate surface to physically bombard the surface and remove the surface of the uppermost layer.
- Ar ions in the plasma depend on directionality to clean and with the decreasing sizes of the interconnects, the increasing aspect ratios, and the resulting shading that can occur, this process is ineffective in removing oxides in the small features.
- the present invention provides a process for removing oxides and other contaminants comprising initiating a plasma containing a reducing agent in a chamber and exposing at least a portion of a substrate surface having a reducible contaminant to the reducing agent.
- the reducing agent comprises a compound containing nitrogen and hydrogen, preferably ammonia.
- One example may include introducing a reducing agent comprising nitrogen and hydrogen into a chamber, initiating a plasma in the chamber, and exposing an oxide to the reducing agent.
- the plasma process parameters to reduce an oxide, such as copper oxide, using ammonia include a pressure range of about 1 to about 9 mTorr, an RF power of about 100 to about 1000 watts for a 200 mm wafer to the chamber with a power density of about 1.43 to 14.3 watts/cm 2 , a substrate temperature of about 100° to about 450° C., a showerhead to substrate spacing of about 200 to about 600 mils, and a reducing agent flow rate of about 100 to about 1000 sccm.
- An exemplary process sequence of the invention such as for forming a dual damascene structure, includes depositing a dielectric on a substrate, depositing an etch stop, etching the etch stop, depositing a barrier layer, depositing a metal layer, initiating a reducing agent plasma, reducing oxides which may form on at least some of the metal surface with the reducing agent, such as ammonia, and in situ depositing a layer, such as a nitride layer, over the reduced surface.
- FIG. 1 shows a schematic of a multilevel substrate with interconnects
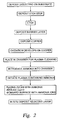
- FIG. 2 is a flow chart of one example of a deposition/etching process using the plasma reducing process
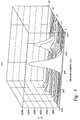
- FIG. 3 is a graph of dielectric compatibility showing oxygen levels obtained from a copper wafer after a CMP process
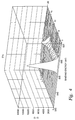
- FIG. 4 is a graph in comparison to FIG. 3 showing an improvement by the plasma reducing process of the present invention, having decreased oxygen levels;
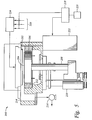
- FIG. 5 shows a cross-sectional view of one commercially available CVD plasma reactor in which the plasma reducing process of the present invention may be performed
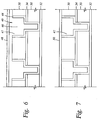
- FIG. 6 is a dual damascene structure showing an oxide layer on a conductor
- FIG. 7 is a dual damascene structure with a layer deposited on the cleaned conductor.
- the present invention provides a process for removing oxides and other contaminants comprising initiating a plasma containing a reducing agent and exposing at least a portion of a substrate surface having a reducible contaminant to the plasma and the reducing agent.
- the reducing process is believed to increase the adhesion of the adjoining layer and to decrease the electrical resistance of the combined layers by decreasing the oxygen content in the oxidized layer. At least in part, these adverse oxide effects are believed to be due to the metal oxides, such as copper oxide (Cu 2 O), reducing the mobility of the metal, e.g., copper (Cu).
- the process is performed in situ to minimize re-contamination before the deposition of a subsequent layer. In situ processing may be particularly important with copper, because of its rapid susceptibility to oxidation.
- a metal layer is deposited at some point in the deposition process and typically comprises aluminum or copper. Because copper is being considered for the conducting material, much of the discussion herein is addressed to copper. However, the present invention may be used for any oxidized metal layers, such as Ti, TiN, Ta, TaN, Al, and others. It may also be used for other layers, including silicon oxides.
- the present invention combines the chemical reactive cleaning of a reducing agent, such as a compound containing nitrogen and hydrogen, including ammonia, with the physical bombardment of the ions from a plasma, and so may be used on a variety of materials to effectuate the reduction of contaminants, such as oxides. While oxides are clearly discussed in the specification, other contaminants would fall within the scope of the present invention. It is believed that the nitrogen combined with hydrogen allows a reduced energy level to break the hydrogen bonds and otherwise disassociate the molecules and more effectively utilize the reducing agent to clean the contaminants.
- the metal is cleaned according to the teaching of the present invention.
- cleaning the term is meant to include a reduction of an oxide or other contaminants. Cleaning may be necessary due to exposure to an oxygen source (such as air, diatomic oxygen, or oxygen contained in a molecular compound). This cleaning may take place in the same CVD or plasma enhanced chemical vapor deposition (“PECVD”) chamber in which the subsequent layer is deposited, as an in situ process.
- PECVD plasma enhanced chemical vapor deposition
- the term “in situ” is intended to include in a given chamber, such as in a plasma chamber, or in a system, such as an integrated cluster tool arrangement, without exposing the material to intervening contamination environments. An in situ process typically minimizes process time and possible contaminants compared to relocating the substrate to other processing chambers or areas.
- the reduction process typically includes introducing the reducing agent, such as ammonia, into a vacuum chamber and initiating a plasma where the plasma excites the ammonia into an energized ionic state.
- the energized ions chemically react with the oxide and the oxides are removed according to the following equation: 3 Cu 2 O+2 NH 3 ⁇ 6 Cu+3 H 2 O+N 2
- the plasma provides the energy necessary to disassociate the ammonia and to provide the desired ion bombardment.
- the ionized particles impact the oxidized surfaces in the reduction process to further enhance the cleaning.
- the combination of chemical reactions and physical bombardment of ions increases the likelihood that all surfaces in small features are cleaned or oxides reduced.
- the process of the invention is believed to afford at least two advantages.
- First, the cleaned surface is better prepared for increased adhesion to an adjoining layer. Removal of the oxide allows a better bond to the base conducting material.
- oxides are known to increase resistance of a layer or combined layers. Thus, the reduction of the oxide decreases the resistance or impedance of the combined layers.
- the plasma process parameters for at least one embodiment, using ammonia to reduce the copper oxide include a pressure range of about 1 to about 9 mTorr, an RF power of about 100 to about 1000 watts to a chamber, that may have a reaction zone, to create the plasma having a power density of about 1.43 to about 14.3 watts/cm 2 , a substrate surface temperature of about 100° to about 450° C., a showerhead to substrate spacing of about 200 to about 600 mils, and a reducing agent flowing at a rate of about 100 to about 1000 sccm into the chamber.
- the gas dispersion element such as a “showerhead”, is commonly known to those with ordinary skill in the art and is used interchangeably herein, and includes other gas dispersion elements.
- the “reaction zone” is the zone between the showerhead and the substrate surface in the chamber, such as one in a CENTURA DxZTM CVD reactor, fabricated and sold by Applied Materials, Inc. of Santa Clara, Calif.
- a preferred process range includes a pressure range of about 3 to about 7 mTorr, an RF power of about 100 to about 500 watts for a 200 mm wafer having a power density of about 1.43 to about 7.14 watts/cm 2 , a substrate temperature of about 200° to about 400° C., a showerhead to substrate spacing of about 200 to about 500 mils, and a reducing agent flowing at a rate of about 100 to about 500 sccm.
- a most preferred process range includes a pressure range of about 4 to about 6 mTorr, an RF power of about 200 to about 400 watts having a power density of about 2.86 to about 5.72 watts/cm 2 , a substrate temperature of about 300° to about 400° C., a showerhead to substrate spacing of about 300 to about 400 mils, and a reducing agent flowing at a rate of about 200 to about 300 sccm.
- carrier gases may be used in conjunction with the above process parameters to assist in stabilizing the gas flow and the plasma reaction.
- the flow rate of the carrier gases such as helium, argon, and nitrogen, could be approximately 0 to 2000 sccm.
- the plasma reducing process reduces, treats, or otherwise modifies the surface in about 5 to about 60 seconds.
- the ammonia plasma is generated in one or more treatment cycles and purged between cycles.
- one treatment cycle lasting 10 seconds effectively removes oxygen from an oxidized copper surface.
- the parameters could be adjusted for other materials besides copper and other contaminants besides oxides.
- FIG. 2 shows a deposition/plasma reducing sequence of one example of the present invention.
- a dielectric such as silicon dioxide, silicon nitride, or silicon carbide is deposited on a substrate.
- substrate includes the IC base or the IC including deposited materials or levels thereon, as the context may indicate.
- An etch stop is deposited over the dielectric and interconnects are etched therethrough to form a pattern.
- Horizontal interconnects are typically referred to as lines and vertical interconnects are typically referred to as contacts or vias; contacts extend to a device on the underlying substrate, while vias extend to an underlying metal layer, such as M 1 , M 2 , etc.
- a barrier layer such as a TiN layer, is deposited over the pattern to restrict the diffusion of the conductor into the dielectric layer(s).
- the conducting material may then be deposited over the barrier layer.
- An oxidation may form on the conducting material, impeding adhesion and conductance.
- the substrate may be placed in a processing chamber for plasma cleaning that may be in situ with prior or subsequent processes. Typically, the system would initiate a plasma and introduce a reducing agent into the chamber, whereupon the plasma cleaning would occur. The plasma would assist in energizing the reducing agent molecules to clean and otherwise reduce the oxide.
- another layer such as a nitride, may be in situ deposited over the conductor to reduce further contamination from an adverse environment, such as one with oxygen.
- this layer is a dielectric layer, but can include other types of layers, such as a barrier layer, an etch stop, or a passivation layer.
- the reduced substrate may be transported to a different chamber for subsequent processing.
- the cleaning is not restricted to the conductor—other layers before and after the conductor layer could be plasma cleaned using the underlying concepts of the present invention.
- FIG. 3 shows the oxygen detected through a 500 ⁇ nitride layer deposited on a copper surface after a CMP process without a plasma reducing process.
- the x-axis represents the binding energy in electron volts (ev)
- the y-axis represents counts per signal (c/s)
- the z-axis represents a relative depth profile through the nitride film layer.
- the x-axis, showing the binding energy is element specific and the substrate layers have been tested at an oxygen binding energy level to detect its presence.
- the y-axis represents the oxygen level detected at an oxygen-specific binding energy. Because the z-axis is relative, the distance between the two largest peaks along the z-axis is the approximate thickness of the 500 ⁇ nitride layer.
- FIG. 3 shows a first high peak closest to the origin of the z-axis of ⁇ 11000 c/s. This first and highest peak represents the surface of the nitride layer and may be ignored for the present purposes.
- the last large peak at a depth of ⁇ 500 ⁇ represents the oxygen level of ⁇ 6000 c/s at the nitride/copper interface. This interface has a quantity of copper oxide that has not been reduced in accordance with the teaching of the present invention.
- FIG. 4 is a graph corresponding to FIG. 3 , showing the results from an exemplary copper surface substrate treated by an ammonia plasma reducing process of the present invention.
- FIG. 4 can be compared to FIG. 3 and the axes represent similar scales and values. Similar to the substrate surface of FIG. 3 , a 500 ⁇ nitride layer was deposited on the copper after applying the plasma reducing process of the present invention.
- FIG. 4 shows an overall lower oxygen level beyond the initial surface peak, where the initial surface peak may again be ignored for the present purposes.
- the oxygen level at the nitride/copper interface represented by the second peak at a depth of about 500 ⁇ , has been lowered to a level of ⁇ 3000 c/s due to the elimination or reduction of the oxide from the copper surface.
- FIG. 5 is a cross-sectional view of a CVD plasma reactor in which the above plasma reducing process may be performed, such as a CENTURA DxZTM mentioned above.
- the present invention could be used in other reactors, such as a lamp heated reactor.
- Reactor 10 contains a gas distribution manifold 11 , which may be the above described showerhead, for dispersing process gases through perforated holes (not shown) in the manifold to a substrate or wafer 16 that rests on a substrate support plate or susceptor 12 .
- Susceptor 12 is resistivity heated and is mounted on a support stem 13 , so that susceptor 12 and the wafer supported on the upper surface of susceptor 12 can be controllably moved by a lift motor 14 between a lower loading/off-loading position and an upper processing position, which is spaced closely adjacent to the manifold 11 .
- a lift motor 14 When susceptor 12 and the wafer 16 are in the processing position, they are surrounded by an insulator ring 17 .
- gases inlet to manifold 11 are uniformly distributed radially across the substrate surface. The gases exhaust through a port 24 by a vacuum pump system 32 .
- the deposition process performed in reactor 10 can be either a thermal process or a plasma enhanced process.
- a controlled plasma is formed adjacent to the wafer by RF energy applied to distribution manifold 11 from RF power supply 25 with susceptor 12 grounded.
- Gas distribution manifold 11 is also an RF electrode, while susceptor 12 is grounded.
- RF power supply 25 can supply either single or mixed frequency RF power to manifold 11 to enhance the decomposition of any reactive species introduced into chamber 15 .
- a mixed frequency RF power supply typically supplies power at a high RF frequency (RF 1 ) of 13.56 MHz and at a low RF frequency (RF 2 ) of 350 kHz.
- the system controller 34 and memory 38 control the activities of the CVD reactor.
- the above chamber can be used to plasma reduce an oxide with a reducing agent and particularly a copper oxide with ammonia.
- the reducing agent can be introduced through manifold 11 and be uniformly distributed radially across the wafer surface for the plasma reducing process in the manner described above, followed by the gases exhausting through the port 24 .
- This discussion applies in general to the improvement provided by the plasma reducing process and can be used in multiple environments, on substrates, and in a variety of processes.
- This plasma reducing process has particular applicability to the increased density and complexity of a dual damascene structure.
- the following discussion briefly discusses aspects from a dual damascene structure and how the plasma reducing process may be utilized.
- FIGS. 6 and 7 represent a dual damascene structure which can be cleaned using the techniques of the present invention.
- a first dielectric layer 30 is deposited on a substrate 32 , followed by an etch stop 34 deposition.
- the etch stop is pattern etched to define contact/via openings 40 and to expose the first dielectric layer in the areas where the contacts/vias are to be formed.
- a second dielectric layer 38 is deposited over the etch stop and patterned to define interconnect lines, preferably using conventional photolithography processes with a photoresist layer, as would be known to those with ordinary skill in the art.
- interconnects and contacts/vias are then etched using reactive ion etching or other anisotropic etching techniques and any photoresist or other material used to pattern the layers is removed using an oxygen strip or other suitable process.
- a barrier layer 44 is then preferably deposited conformally in the metallization pattern to prevent metal migration into the surrounding silicon and/or dielectric material.
- the above plasma reducing process may be useful on the barrier layer or other layers deposited prior or subsequent to the metal layer.
- the regime and parameters discussed above for the plasma reduction of the conductor could be adjusted for the particular layer in question.
- the metal layer 47 is deposited in the vias and lines and is preferably a conductive material such as aluminum, copper, tungsten or combinations thereof with the recent trend being copper.
- the metal layer is deposited using either CVD, PVD, electroplating, or combinations thereof to form the conductive structure.
- a CMP process may be used to planarize the metal surface.
- a sacrificial layer may be deposited on the field areas between the interconnects prior to the metal deposition, and then the sacrificial layer stripped after the metal is deposited, leaving a surface suitable for the next stage of the deposition process.
- the structure surface may be planarized, using a CMP process at this stage and/or at other stages.
- the above plasma reducing process may be initiated to remove or reduce a contaminant layer 48 , such as copper oxide, that may have formed on the metal layer 47 , including the interconnects 46 .
- the plasma reducing process is applied in situ with the deposit of the adjoining layer 50 , shown in FIG. 7 .
- This layer may be another dielectric layer, a barrier layer, a passivation layer, or some other layer beside the metal layer.
- the process steps could include: depositing a first dielectric layer, such as a fluorinated silicate glass (FSG) layer, on a substrate, depositing a low k dielectric etch stop on the first dielectric layer, patterning the etch stops to define the contacts/vias, stripping the patterning medium such as a photoresist layer, depositing a second dielectric layer, such as an FSG layer, and patterning a resist layer on the second dielectric layer to define one or more interconnects.
- the process can include: depositing a barrier layer in the structure, depositing a metal layer such as copper, and exposing an oxidized surface of the metal layer to a plasma with the reducing agent to reduce the oxide.
- Another layer such as a nitride, may then be deposited over the surface in situ with the reducing process.
Landscapes
- Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Power Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Computer Hardware Design (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Manufacturing & Machinery (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Plasma & Fusion (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Optics & Photonics (AREA)
- Internal Circuitry In Semiconductor Integrated Circuit Devices (AREA)
- Formation Of Insulating Films (AREA)
- Drying Of Semiconductors (AREA)
- Element Separation (AREA)
Abstract
The present invention provides an in situ plasma reducing process to reduce oxides or other contaminants, using a compound of nitrogen and hydrogen, typically ammonia, at relatively low temperatures prior to depositing a subsequent layer thereon. The adhesion characteristics of the layers are improved and oxygen presence is reduced compared to the typical physical sputter cleaning process of an oxide layer. This process may be particularly useful for the complex requirements of a dual damascene structure, especially with copper applications.
Description
- This application is a divisional application of co-pending U.S. patent application Ser. No. 10/655,438, filed Sep. 4, 2003, which is a continuation of U.S. patent application Ser. No. 10/013,182, filed Dec. 7, 2001, which is a divisional of U.S. patent application Ser. No. 09/365,129, filed Jul. 30, 1999, which is a continuation-in-part of U.S. patent application Ser. No. 09/193,920, filed Nov. 17, 1998, all of which are hereby incorporated by reference in their entireties.
- 1. Field of the Invention
- The present invention relates generally to the fabrication of integrated circuits on substrates. More particularly, the invention relates to a method of reducing oxides on a substrate prior to depositing a layer thereover in the fabrication process.
- 2. Background of the Invention
- Reliably producing sub-half micron and smaller features is one of the key technologies for the next generation of very large scale integration (VLSI) and ultra large scale integration (ULSI) integrated circuits. However, as the fringes of circuit technology are pressed, the shrinking dimensions of interconnects in VLSI and ULSI technology has placed additional demands on the processing capabilities. The multilevel interconnects that lie at the heart of this technology require careful processing of high aspect ratio features, such as vias and other interconnects. Reliable formation of these interconnects is very important to the VLSI and ULSI success and to the continued effort to increase circuit density and quality of individual substrates and die.
- Conventional chemical vapor deposition (CVD) and physical vapor deposition (PVD), and now electroplating, techniques are used to deposit electrically conductive material into the contacts, vias, lines, or other features formed on the substrate. Considerable effort has focused on reliably depositing material in these high aspect ratio, smaller interconnects.
- One issue that still needs improvement as feature sizes shrink is the reduction of oxides in these very small features.
FIG. 1 shows asubstrate 2 with avia 4 formed within an electrically insulative ordielectric layer 6. With current technology, the aspect ratio has increased to approximately 5:1 for the height to width ratio, shown inFIG. 1 as x with respect to d. As a result, it is becoming more difficult to properly prepare the surfaces within the small features for subsequent processing, especially in the lower interconnect portions, such as in theinterconnect areas - In part, this attention to improved cleaning is due to a desired change in the conductor metal. For example, copper is now being considered as an interconnect material in place of aluminum, because copper has a lower resistivity (1.7 μΩ-cm compared to 3.1 μΩ-cm for aluminum) and higher current carrying capacity. However, copper is highly susceptible to oxidation. With copper depositions, oxidation is considered a detriment and interferes with adhesion on the adjacent layer, affects conductivity of the copper feature, and reduces the reliability of the overall circuit. Furthermore, present processes utilize oxygen for a variety of reasons in some instances and in other instances, oxygen is a byproduct of the reactions. Thus, even carefully controlled environments may contain oxygen that may oxidize copper or other conductive materials, such as aluminum, to the detriment of the circuit.
- Copper has other difficulties. Because copper is difficult to etch in a precise pattern, traditional deposition/etch processes for forming interconnects has become unworkable, and accordingly, a “dual damascene” structure is being used for copper interconnects. In a typical dual damascene structure, the dielectric layer is etched to define both the contacts/vias and the interconnect lines. Metal is then inlaid into the defined pattern and any excess metal is typically removed from the top of the structure in a planarization process, such as CMP. This complex approach increases the importance of obtaining properly cleaned surfaces within the interconnects.
- Prior to the present invention, an inert gas plasma, such as an Argon (Ar) plasma, physically cleaned the surfaces of interconnects and metal layers, such as aluminum and copper, as ions were attracted to the substrate surface to physically bombard the surface and remove the surface of the uppermost layer. However, the Ar ions in the plasma depend on directionality to clean and with the decreasing sizes of the interconnects, the increasing aspect ratios, and the resulting shading that can occur, this process is ineffective in removing oxides in the small features.
- Therefore, there is a need for an improved cleaning process to reduce oxides formed on the surface of substrates and materials deposited thereon.
- The present invention provides a process for removing oxides and other contaminants comprising initiating a plasma containing a reducing agent in a chamber and exposing at least a portion of a substrate surface having a reducible contaminant to the reducing agent. In a preferred embodiment, the reducing agent comprises a compound containing nitrogen and hydrogen, preferably ammonia. One example may include introducing a reducing agent comprising nitrogen and hydrogen into a chamber, initiating a plasma in the chamber, and exposing an oxide to the reducing agent. The plasma process parameters to reduce an oxide, such as copper oxide, using ammonia include a pressure range of about 1 to about 9 mTorr, an RF power of about 100 to about 1000 watts for a 200 mm wafer to the chamber with a power density of about 1.43 to 14.3 watts/cm2, a substrate temperature of about 100° to about 450° C., a showerhead to substrate spacing of about 200 to about 600 mils, and a reducing agent flow rate of about 100 to about 1000 sccm.
- An exemplary process sequence of the invention, such as for forming a dual damascene structure, includes depositing a dielectric on a substrate, depositing an etch stop, etching the etch stop, depositing a barrier layer, depositing a metal layer, initiating a reducing agent plasma, reducing oxides which may form on at least some of the metal surface with the reducing agent, such as ammonia, and in situ depositing a layer, such as a nitride layer, over the reduced surface.
- So that the manner in which the above recited features, advantages and objects of the present invention are attained and can be understood in detail, a more particular description of the invention, briefly summarized above, may be had by reference to the embodiments thereof which are illustrated in the appended drawings.
- It is to be noted, however, that the appended drawings illustrate only typical embodiments of this invention and are therefore not to be considered limiting of its scope, for the invention may admit to other equally effective embodiments.
-
FIG. 1 shows a schematic of a multilevel substrate with interconnects; -
FIG. 2 is a flow chart of one example of a deposition/etching process using the plasma reducing process; -
FIG. 3 is a graph of dielectric compatibility showing oxygen levels obtained from a copper wafer after a CMP process; -
FIG. 4 is a graph in comparison toFIG. 3 showing an improvement by the plasma reducing process of the present invention, having decreased oxygen levels; -
FIG. 5 shows a cross-sectional view of one commercially available CVD plasma reactor in which the plasma reducing process of the present invention may be performed; -
FIG. 6 is a dual damascene structure showing an oxide layer on a conductor; and -
FIG. 7 is a dual damascene structure with a layer deposited on the cleaned conductor. - The present invention provides a process for removing oxides and other contaminants comprising initiating a plasma containing a reducing agent and exposing at least a portion of a substrate surface having a reducible contaminant to the plasma and the reducing agent. The reducing process is believed to increase the adhesion of the adjoining layer and to decrease the electrical resistance of the combined layers by decreasing the oxygen content in the oxidized layer. At least in part, these adverse oxide effects are believed to be due to the metal oxides, such as copper oxide (Cu2O), reducing the mobility of the metal, e.g., copper (Cu). In the preferred embodiment, the process is performed in situ to minimize re-contamination before the deposition of a subsequent layer. In situ processing may be particularly important with copper, because of its rapid susceptibility to oxidation.
- In an integrated circuit (IC) fabrication, a metal layer is deposited at some point in the deposition process and typically comprises aluminum or copper. Because copper is being considered for the conducting material, much of the discussion herein is addressed to copper. However, the present invention may be used for any oxidized metal layers, such as Ti, TiN, Ta, TaN, Al, and others. It may also be used for other layers, including silicon oxides. The present invention combines the chemical reactive cleaning of a reducing agent, such as a compound containing nitrogen and hydrogen, including ammonia, with the physical bombardment of the ions from a plasma, and so may be used on a variety of materials to effectuate the reduction of contaminants, such as oxides. While oxides are clearly discussed in the specification, other contaminants would fall within the scope of the present invention. It is believed that the nitrogen combined with hydrogen allows a reduced energy level to break the hydrogen bonds and otherwise disassociate the molecules and more effectively utilize the reducing agent to clean the contaminants.
- Before depositing a layer over the metal, such as a nitride, the metal is cleaned according to the teaching of the present invention. By “cleaning”, the term is meant to include a reduction of an oxide or other contaminants. Cleaning may be necessary due to exposure to an oxygen source (such as air, diatomic oxygen, or oxygen contained in a molecular compound). This cleaning may take place in the same CVD or plasma enhanced chemical vapor deposition (“PECVD”) chamber in which the subsequent layer is deposited, as an in situ process. The term “in situ” is intended to include in a given chamber, such as in a plasma chamber, or in a system, such as an integrated cluster tool arrangement, without exposing the material to intervening contamination environments. An in situ process typically minimizes process time and possible contaminants compared to relocating the substrate to other processing chambers or areas.
- In one embodiment, the reduction process typically includes introducing the reducing agent, such as ammonia, into a vacuum chamber and initiating a plasma where the plasma excites the ammonia into an energized ionic state. The energized ions chemically react with the oxide and the oxides are removed according to the following equation:
3 Cu2O+2 NH3→6 Cu+3 H2O+N2
The plasma provides the energy necessary to disassociate the ammonia and to provide the desired ion bombardment. The ionized particles impact the oxidized surfaces in the reduction process to further enhance the cleaning. The combination of chemical reactions and physical bombardment of ions increases the likelihood that all surfaces in small features are cleaned or oxides reduced. - The process of the invention is believed to afford at least two advantages. First, the cleaned surface is better prepared for increased adhesion to an adjoining layer. Removal of the oxide allows a better bond to the base conducting material. Secondly, oxides are known to increase resistance of a layer or combined layers. Thus, the reduction of the oxide decreases the resistance or impedance of the combined layers.
- The plasma process parameters for at least one embodiment, using ammonia to reduce the copper oxide, include a pressure range of about 1 to about 9 mTorr, an RF power of about 100 to about 1000 watts to a chamber, that may have a reaction zone, to create the plasma having a power density of about 1.43 to about 14.3 watts/cm2, a substrate surface temperature of about 100° to about 450° C., a showerhead to substrate spacing of about 200 to about 600 mils, and a reducing agent flowing at a rate of about 100 to about 1000 sccm into the chamber. The gas dispersion element, such as a “showerhead”, is commonly known to those with ordinary skill in the art and is used interchangeably herein, and includes other gas dispersion elements. The “reaction zone” is the zone between the showerhead and the substrate surface in the chamber, such as one in a CENTURA DxZ™ CVD reactor, fabricated and sold by Applied Materials, Inc. of Santa Clara, Calif.
- A preferred process range includes a pressure range of about 3 to about 7 mTorr, an RF power of about 100 to about 500 watts for a 200 mm wafer having a power density of about 1.43 to about 7.14 watts/cm2, a substrate temperature of about 200° to about 400° C., a showerhead to substrate spacing of about 200 to about 500 mils, and a reducing agent flowing at a rate of about 100 to about 500 sccm. A most preferred process range includes a pressure range of about 4 to about 6 mTorr, an RF power of about 200 to about 400 watts having a power density of about 2.86 to about 5.72 watts/cm2, a substrate temperature of about 300° to about 400° C., a showerhead to substrate spacing of about 300 to about 400 mils, and a reducing agent flowing at a rate of about 200 to about 300 sccm. Additionally, carrier gases may be used in conjunction with the above process parameters to assist in stabilizing the gas flow and the plasma reaction. The flow rate of the carrier gases, such as helium, argon, and nitrogen, could be approximately 0 to 2000 sccm.
- The plasma reducing process reduces, treats, or otherwise modifies the surface in about 5 to about 60 seconds. Preferably, the ammonia plasma is generated in one or more treatment cycles and purged between cycles. However, in most cases, one treatment cycle lasting 10 seconds effectively removes oxygen from an oxidized copper surface. Naturally, the parameters could be adjusted for other materials besides copper and other contaminants besides oxides.
-
FIG. 2 shows a deposition/plasma reducing sequence of one example of the present invention. Other sequences, fabrication techniques, and processes may be used. Typically, a dielectric such as silicon dioxide, silicon nitride, or silicon carbide is deposited on a substrate. The term “substrate” herein includes the IC base or the IC including deposited materials or levels thereon, as the context may indicate. An etch stop is deposited over the dielectric and interconnects are etched therethrough to form a pattern. Horizontal interconnects are typically referred to as lines and vertical interconnects are typically referred to as contacts or vias; contacts extend to a device on the underlying substrate, while vias extend to an underlying metal layer, such as M1, M2, etc. Once the lines and contacts/vias are patterned, such as shown inFIG. 1 , a barrier layer, such as a TiN layer, is deposited over the pattern to restrict the diffusion of the conductor into the dielectric layer(s). The conducting material may then be deposited over the barrier layer. An oxidation may form on the conducting material, impeding adhesion and conductance. The substrate may be placed in a processing chamber for plasma cleaning that may be in situ with prior or subsequent processes. Typically, the system would initiate a plasma and introduce a reducing agent into the chamber, whereupon the plasma cleaning would occur. The plasma would assist in energizing the reducing agent molecules to clean and otherwise reduce the oxide. - After the conductor is cleaned, another layer, such as a nitride, may be in situ deposited over the conductor to reduce further contamination from an adverse environment, such as one with oxygen. Typically, this layer is a dielectric layer, but can include other types of layers, such as a barrier layer, an etch stop, or a passivation layer. Alternatively, the reduced substrate may be transported to a different chamber for subsequent processing. The cleaning is not restricted to the conductor—other layers before and after the conductor layer could be plasma cleaned using the underlying concepts of the present invention.
-
FIG. 3 shows the oxygen detected through a 500 Å nitride layer deposited on a copper surface after a CMP process without a plasma reducing process. The x-axis represents the binding energy in electron volts (ev), the y-axis represents counts per signal (c/s), and the z-axis represents a relative depth profile through the nitride film layer. The x-axis, showing the binding energy, is element specific and the substrate layers have been tested at an oxygen binding energy level to detect its presence. The y-axis represents the oxygen level detected at an oxygen-specific binding energy. Because the z-axis is relative, the distance between the two largest peaks along the z-axis is the approximate thickness of the 500 Å nitride layer. Beyond the 500 Å nitride layer, the signal count drops to approximately zero because copper is a conductor.FIG. 3 shows a first high peak closest to the origin of the z-axis of ˜11000 c/s. This first and highest peak represents the surface of the nitride layer and may be ignored for the present purposes. The last large peak at a depth of ˜500 Å represents the oxygen level of ˜6000 c/s at the nitride/copper interface. This interface has a quantity of copper oxide that has not been reduced in accordance with the teaching of the present invention. -
FIG. 4 is a graph corresponding toFIG. 3 , showing the results from an exemplary copper surface substrate treated by an ammonia plasma reducing process of the present invention.FIG. 4 can be compared toFIG. 3 and the axes represent similar scales and values. Similar to the substrate surface ofFIG. 3 , a 500 Å nitride layer was deposited on the copper after applying the plasma reducing process of the present invention.FIG. 4 shows an overall lower oxygen level beyond the initial surface peak, where the initial surface peak may again be ignored for the present purposes. Noticeably, the oxygen level at the nitride/copper interface, represented by the second peak at a depth of about 500 Å, has been lowered to a level of ˜3000 c/s due to the elimination or reduction of the oxide from the copper surface. -
FIG. 5 is a cross-sectional view of a CVD plasma reactor in which the above plasma reducing process may be performed, such as a CENTURA DxZ™ mentioned above. The present invention could be used in other reactors, such as a lamp heated reactor.Reactor 10 contains a gas distribution manifold 11, which may be the above described showerhead, for dispersing process gases through perforated holes (not shown) in the manifold to a substrate or wafer 16 that rests on a substrate support plate or susceptor 12. Susceptor 12 is resistivity heated and is mounted on a support stem 13, so that susceptor 12 and the wafer supported on the upper surface of susceptor 12 can be controllably moved by a lift motor 14 between a lower loading/off-loading position and an upper processing position, which is spaced closely adjacent to the manifold 11. When susceptor 12 and the wafer 16 are in the processing position, they are surrounded by an insulator ring 17. During processing, gases inlet to manifold 11 are uniformly distributed radially across the substrate surface. The gases exhaust through a port 24 by avacuum pump system 32. - The deposition process performed in
reactor 10 can be either a thermal process or a plasma enhanced process. In a plasma process, a controlled plasma is formed adjacent to the wafer by RF energy applied to distribution manifold 11 from RF power supply 25 with susceptor 12 grounded. Gas distribution manifold 11 is also an RF electrode, while susceptor 12 is grounded. RF power supply 25 can supply either single or mixed frequency RF power to manifold 11 to enhance the decomposition of any reactive species introduced intochamber 15. A mixed frequency RF power supply typically supplies power at a high RF frequency (RF1) of 13.56 MHz and at a low RF frequency (RF2) of 350 kHz. Thesystem controller 34 andmemory 38 control the activities of the CVD reactor. An example of such a CVD reactor is described in U.S. Pat. No. 5,000,113, which is incorporated by reference and entitled “Thermal CVD/PECVD Reactor and Use for Thermal Chemical Vapor Deposition of Silicon Dioxide and In-situ Multi-step Planarized Process,” issued to Wang et al. and assigned to Applied Materials, Inc., the assignee of the present invention. - With the present invention, the above chamber can be used to plasma reduce an oxide with a reducing agent and particularly a copper oxide with ammonia. The reducing agent can be introduced through manifold 11 and be uniformly distributed radially across the wafer surface for the plasma reducing process in the manner described above, followed by the gases exhausting through the port 24.
- The above discussion applies in general to the improvement provided by the plasma reducing process and can be used in multiple environments, on substrates, and in a variety of processes. This plasma reducing process has particular applicability to the increased density and complexity of a dual damascene structure. The following discussion briefly discusses aspects from a dual damascene structure and how the plasma reducing process may be utilized.
-
FIGS. 6 and 7 represent a dual damascene structure which can be cleaned using the techniques of the present invention. In forming one type of dual damascene structure, afirst dielectric layer 30 is deposited on asubstrate 32, followed by anetch stop 34 deposition. The etch stop is pattern etched to define contact/viaopenings 40 and to expose the first dielectric layer in the areas where the contacts/vias are to be formed. Asecond dielectric layer 38 is deposited over the etch stop and patterned to define interconnect lines, preferably using conventional photolithography processes with a photoresist layer, as would be known to those with ordinary skill in the art. The interconnects and contacts/vias are then etched using reactive ion etching or other anisotropic etching techniques and any photoresist or other material used to pattern the layers is removed using an oxygen strip or other suitable process. Abarrier layer 44 is then preferably deposited conformally in the metallization pattern to prevent metal migration into the surrounding silicon and/or dielectric material. - With the present invention, the above plasma reducing process may be useful on the barrier layer or other layers deposited prior or subsequent to the metal layer. The regime and parameters discussed above for the plasma reduction of the conductor could be adjusted for the particular layer in question.
- The
metal layer 47 is deposited in the vias and lines and is preferably a conductive material such as aluminum, copper, tungsten or combinations thereof with the recent trend being copper. The metal layer is deposited using either CVD, PVD, electroplating, or combinations thereof to form the conductive structure. Once the structure has been filled with copper or other metal, a CMP process may be used to planarize the metal surface. In other embodiments, a sacrificial layer may be deposited on the field areas between the interconnects prior to the metal deposition, and then the sacrificial layer stripped after the metal is deposited, leaving a surface suitable for the next stage of the deposition process. The structure surface may be planarized, using a CMP process at this stage and/or at other stages. The above plasma reducing process may be initiated to remove or reduce acontaminant layer 48, such as copper oxide, that may have formed on themetal layer 47, including theinterconnects 46. In the preferred embodiment, the plasma reducing process is applied in situ with the deposit of the adjoininglayer 50, shown inFIG. 7 . This layer may be another dielectric layer, a barrier layer, a passivation layer, or some other layer beside the metal layer. - Specifically, the process steps could include: depositing a first dielectric layer, such as a fluorinated silicate glass (FSG) layer, on a substrate, depositing a low k dielectric etch stop on the first dielectric layer, patterning the etch stops to define the contacts/vias, stripping the patterning medium such as a photoresist layer, depositing a second dielectric layer, such as an FSG layer, and patterning a resist layer on the second dielectric layer to define one or more interconnects. Once the dual damascene structure has been formed, the process can include: depositing a barrier layer in the structure, depositing a metal layer such as copper, and exposing an oxidized surface of the metal layer to a plasma with the reducing agent to reduce the oxide. Another layer, such as a nitride, may then be deposited over the surface in situ with the reducing process.
- While the foregoing is directed to the preferred embodiment of the present invention, other and further embodiments of the invention may be devised without departing from the basis scope thereof, and the scope thereof is determined by the claims that follow. Furthermore, in this specification, including particularly the claims, the use of “comprising” with “a” or “the”, and variations thereof means that the item(s) or list(s) referenced includes at least the enumerated item(s) or list(s) and furthermore may include a plurality of the enumerated item(s) or list(s), unless otherwise stated.
Claims (6)
1. A semiconductor device comprising:
an interface defined by a first insulating layer comprising silicon carbide and one or more conductive pathways disposed within said layer, the interface having oxidation reduced portions; and
a second insulating layer disposed upon said interface coincident with removal of said oxidation reduced portions.
2. The semiconductor device of claim 1 wherein the second insulating layer comprises at least one layer selected from the group consisting of a patterned etch stop layer, a barrier layer, and a film comprising silicon carbide.
3. The semiconductor device of claim 1 wherein the conductive pathways are formed from at least one of copper, titanium, tantalum, and tungsten.
4. A semiconductor device comprising:
an interface defined by a first insulating layer and one or more conductive pathways disposed within said layer, the interface having oxidation reduced portions; and
a second insulating layer comprising silicon carbide disposed upon said interface coincident with removal of said oxidation reduced portions.
5. A semiconductor device interface comprising:
a layer comprising silicon carbide;
one or more conductive devices disposed within said layer, said layer and conductive devices defining said interface,
wherein said interface is treated with a continuous plasma treatment to remove oxidation and deposit a second layer thereupon.
6. The semiconductor device interface of claim 5 wherein the conductive devices are formed from at least one of copper, titanium, tantalum, and tungsten.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/169,337 US20050263900A1 (en) | 1998-11-17 | 2005-06-29 | Semiconductor device having silicon carbide and conductive pathway interface |
| US12/257,806 US8183150B2 (en) | 1998-11-17 | 2008-10-24 | Semiconductor device having silicon carbide and conductive pathway interface |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/193,920 US20010049181A1 (en) | 1998-11-17 | 1998-11-17 | Plasma treatment for cooper oxide reduction |
| US09/365,129 US6355571B1 (en) | 1998-11-17 | 1999-07-30 | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US10/013,182 US6700202B2 (en) | 1998-11-17 | 2001-12-07 | Semiconductor device having reduced oxidation interface |
| US10/655,438 US6946401B2 (en) | 1998-11-17 | 2003-09-04 | Plasma treatment for copper oxide reduction |
| US11/169,337 US20050263900A1 (en) | 1998-11-17 | 2005-06-29 | Semiconductor device having silicon carbide and conductive pathway interface |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/655,438 Division US6946401B2 (en) | 1998-11-17 | 2003-09-04 | Plasma treatment for copper oxide reduction |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/257,806 Division US8183150B2 (en) | 1998-11-17 | 2008-10-24 | Semiconductor device having silicon carbide and conductive pathway interface |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20050263900A1 true US20050263900A1 (en) | 2005-12-01 |
Family
ID=23437585
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/365,129 Expired - Lifetime US6355571B1 (en) | 1998-11-17 | 1999-07-30 | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US10/013,182 Expired - Lifetime US6700202B2 (en) | 1998-11-17 | 2001-12-07 | Semiconductor device having reduced oxidation interface |
| US10/655,438 Expired - Fee Related US6946401B2 (en) | 1998-11-17 | 2003-09-04 | Plasma treatment for copper oxide reduction |
| US11/169,337 Abandoned US20050263900A1 (en) | 1998-11-17 | 2005-06-29 | Semiconductor device having silicon carbide and conductive pathway interface |
| US12/257,806 Expired - Fee Related US8183150B2 (en) | 1998-11-17 | 2008-10-24 | Semiconductor device having silicon carbide and conductive pathway interface |
Family Applications Before (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/365,129 Expired - Lifetime US6355571B1 (en) | 1998-11-17 | 1999-07-30 | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US10/013,182 Expired - Lifetime US6700202B2 (en) | 1998-11-17 | 2001-12-07 | Semiconductor device having reduced oxidation interface |
| US10/655,438 Expired - Fee Related US6946401B2 (en) | 1998-11-17 | 2003-09-04 | Plasma treatment for copper oxide reduction |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/257,806 Expired - Fee Related US8183150B2 (en) | 1998-11-17 | 2008-10-24 | Semiconductor device having silicon carbide and conductive pathway interface |
Country Status (6)
| Country | Link |
|---|---|
| US (5) | US6355571B1 (en) |
| EP (1) | EP1073106B1 (en) |
| JP (1) | JP2001093902A (en) |
| KR (1) | KR100773188B1 (en) |
| DE (1) | DE60040507D1 (en) |
| TW (1) | TW459304B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150001728A1 (en) * | 2013-06-26 | 2015-01-01 | Taiwan Semiconductor Manufacturing Company, Ltd. | Pre-treatment method for metal-oxide reduction and device formed |
Families Citing this family (74)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20010049181A1 (en) | 1998-11-17 | 2001-12-06 | Sudha Rathi | Plasma treatment for cooper oxide reduction |
| US6355571B1 (en) * | 1998-11-17 | 2002-03-12 | Applied Materials, Inc. | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US6821571B2 (en) * | 1999-06-18 | 2004-11-23 | Applied Materials Inc. | Plasma treatment to enhance adhesion and to minimize oxidation of carbon-containing layers |
| JP4554011B2 (en) * | 1999-08-10 | 2010-09-29 | ルネサスエレクトロニクス株式会社 | Manufacturing method of semiconductor integrated circuit device |
| JP2001160558A (en) * | 1999-12-02 | 2001-06-12 | Nec Corp | Method and apparatus for manufacturing semiconductor device |
| JP2001291720A (en) * | 2000-04-05 | 2001-10-19 | Hitachi Ltd | Semiconductor integrated circuit device and its manufacturing method |
| US6794311B2 (en) | 2000-07-14 | 2004-09-21 | Applied Materials Inc. | Method and apparatus for treating low k dielectric layers to reduce diffusion |
| JP2002110679A (en) * | 2000-09-29 | 2002-04-12 | Hitachi Ltd | Method for manufacturing semiconductor integrated circuit device |
| DE10059143B4 (en) * | 2000-11-29 | 2006-12-28 | Advanced Micro Devices, Inc., Sunnyvale | Surface treatment and cover layer method for producing a copper interface in a semiconductor device |
| JP2002164428A (en) * | 2000-11-29 | 2002-06-07 | Hitachi Ltd | Semiconductor device and its manufacturing method |
| US6693020B2 (en) * | 2001-03-12 | 2004-02-17 | Motorola, Inc. | Method of preparing copper metallization die for wirebonding |
| US7084070B1 (en) | 2001-03-30 | 2006-08-01 | Lam Research Corporation | Treatment for corrosion in substrate processing |
| US6506677B1 (en) * | 2001-05-02 | 2003-01-14 | Advanced Micro Devices, Inc. | Method of forming capped copper interconnects with reduced hillock formation and improved electromigration resistance |
| US6951823B2 (en) * | 2001-05-14 | 2005-10-04 | Axcelis Technologies, Inc. | Plasma ashing process |
| US6630406B2 (en) | 2001-05-14 | 2003-10-07 | Axcelis Technologies | Plasma ashing process |
| JP2003051481A (en) * | 2001-08-07 | 2003-02-21 | Hitachi Ltd | Manufacturing method for semiconductor integrated circuit device |
| US6797652B1 (en) * | 2002-03-15 | 2004-09-28 | Advanced Micro Devices, Inc. | Copper damascene with low-k capping layer and improved electromigration reliability |
| US6664202B2 (en) * | 2002-04-18 | 2003-12-16 | Applied Materials Inc. | Mixed frequency high temperature nitride CVD process |
| DE10224167B4 (en) * | 2002-05-31 | 2007-01-25 | Advanced Micro Devices, Inc., Sunnyvale | A method of making a copper wire with increased resistance to electromigration in a semiconductor element |
| US20040018715A1 (en) * | 2002-07-25 | 2004-01-29 | Applied Materials, Inc. | Method of cleaning a surface of a material layer |
| US6790777B2 (en) * | 2002-11-06 | 2004-09-14 | Texas Instruments Incorporated | Method for reducing contamination, copper reduction, and depositing a dielectric layer on a semiconductor device |
| US7582185B2 (en) * | 2002-12-26 | 2009-09-01 | Canon Kabushiki Kaisha | Plasma-processing apparatus |
| KR100483594B1 (en) * | 2002-12-27 | 2005-04-15 | 매그나칩 반도체 유한회사 | Method of forming metal line of semiconductor device |
| US7273808B1 (en) | 2003-02-03 | 2007-09-25 | Novellus Systems, Inc. | Reactive barrier/seed preclean process for damascene process |
| KR101276694B1 (en) * | 2003-02-14 | 2013-06-19 | 어플라이드 머티어리얼스, 인코포레이티드 | Cleaning of native oxide with hydrogen-containing radicals |
| JP2004247675A (en) * | 2003-02-17 | 2004-09-02 | Renesas Technology Corp | Method of manufacturing semiconductor device |
| US7387738B2 (en) * | 2003-04-28 | 2008-06-17 | Air Products And Chemicals, Inc. | Removal of surface oxides by electron attachment for wafer bumping applications |
| US7897029B2 (en) * | 2008-03-04 | 2011-03-01 | Air Products And Chemicals, Inc. | Removal of surface oxides by electron attachment |
| US8361340B2 (en) * | 2003-04-28 | 2013-01-29 | Air Products And Chemicals, Inc. | Removal of surface oxides by electron attachment |
| US20040219795A1 (en) * | 2003-05-01 | 2004-11-04 | Taiwan Semiconductor Manufacturing Co. | Method to improve breakdown voltage by H2 plasma treat |
| US7371427B2 (en) * | 2003-05-20 | 2008-05-13 | Applied Materials, Inc. | Reduction of hillocks prior to dielectric barrier deposition in Cu damascene |
| US7723228B2 (en) * | 2003-05-20 | 2010-05-25 | Applied Materials, Inc. | Reduction of hillocks prior to dielectric barrier deposition in Cu damascene |
| US6790778B1 (en) * | 2003-09-10 | 2004-09-14 | Taiwan Semiconductor Manufacturing Co., Ltd. | Method for capping over a copper layer |
| US7309651B2 (en) * | 2003-10-30 | 2007-12-18 | Texas Instruments Incorporated | Method for improving reliability of copper interconnects |
| KR100587659B1 (en) * | 2003-12-31 | 2006-06-08 | 동부일렉트로닉스 주식회사 | Method for preventing copper oxidation in dual damascene process |
| US6897147B1 (en) | 2004-01-15 | 2005-05-24 | Taiwan Semiconductor Manufacturing Company | Solution for copper hillock induced by thermal strain with buffer zone for strain relaxation |
| US20050186339A1 (en) * | 2004-02-20 | 2005-08-25 | Applied Materials, Inc., A Delaware Corporation | Methods and apparatuses promoting adhesion of dielectric barrier film to copper |
| US7229911B2 (en) * | 2004-04-19 | 2007-06-12 | Applied Materials, Inc. | Adhesion improvement for low k dielectrics to conductive materials |
| US20050233555A1 (en) * | 2004-04-19 | 2005-10-20 | Nagarajan Rajagopalan | Adhesion improvement for low k dielectrics to conductive materials |
| US7338903B2 (en) * | 2004-04-24 | 2008-03-04 | Taiwan Semiconductor Manufacturing Co., Ltd. | Sequential reducing plasma and inert plasma pre-treatment method for oxidizable conductor layer |
| US20060081965A1 (en) * | 2004-10-15 | 2006-04-20 | Ju-Ai Ruan | Plasma treatment of an etch stop layer |
| US20070012661A1 (en) * | 2005-07-13 | 2007-01-18 | Vasilyeva Irina V | Silicon nitride passivation layers having oxidized interface |
| KR20090047211A (en) * | 2007-11-07 | 2009-05-12 | 삼성전자주식회사 | Method of forming a conductive pattern and method of manufacturing a semiconductor device using the same |
| KR20100099094A (en) * | 2007-12-27 | 2010-09-10 | 램 리써치 코포레이션 | Copper discoloration prevention following bevel etch process |
| US7737029B2 (en) * | 2008-03-18 | 2010-06-15 | Samsung Electronics Co., Ltd. | Methods of forming metal interconnect structures on semiconductor substrates using oxygen-removing plasmas and interconnect structures formed thereby |
| US8043976B2 (en) * | 2008-03-24 | 2011-10-25 | Air Products And Chemicals, Inc. | Adhesion to copper and copper electromigration resistance |
| US20090269507A1 (en) * | 2008-04-29 | 2009-10-29 | Sang-Ho Yu | Selective cobalt deposition on copper surfaces |
| US20100270262A1 (en) * | 2009-04-22 | 2010-10-28 | Applied Materials, Inc. | Etching low-k dielectric or removing resist with a filtered ionized gas |
| US8454850B2 (en) * | 2009-09-02 | 2013-06-04 | Air Products And Chemicals, Inc. | Method for the removal of surface oxides by electron attachment |
| US8329575B2 (en) | 2010-12-22 | 2012-12-11 | Applied Materials, Inc. | Fabrication of through-silicon vias on silicon wafers |
| US8758638B2 (en) | 2011-05-10 | 2014-06-24 | Applied Materials, Inc. | Copper oxide removal techniques |
| US20140117545A1 (en) * | 2012-10-26 | 2014-05-01 | Globalfoundries Singapore Pte. Ltd | Copper hillock prevention with hydrogen plasma treatment in a dedicated chamber |
| US20140187045A1 (en) * | 2013-01-02 | 2014-07-03 | Applied Materials, Inc. | Silicon nitride gapfill implementing high density plasma |
| US9018108B2 (en) | 2013-01-25 | 2015-04-28 | Applied Materials, Inc. | Low shrinkage dielectric films |
| US8980740B2 (en) | 2013-03-06 | 2015-03-17 | Globalfoundries Inc. | Barrier layer conformality in copper interconnects |
| US9070750B2 (en) | 2013-03-06 | 2015-06-30 | Novellus Systems, Inc. | Methods for reducing metal oxide surfaces to modified metal surfaces using a gaseous reducing environment |
| US9865501B2 (en) | 2013-03-06 | 2018-01-09 | Lam Research Corporation | Method and apparatus for remote plasma treatment for reducing metal oxides on a metal seed layer |
| KR101734821B1 (en) | 2013-03-15 | 2017-05-12 | 어플라이드 머티어리얼스, 인코포레이티드 | Processing systems, apparatus, and methods adapted to process substrates in electronic device manufacturing |
| US20150206798A1 (en) * | 2014-01-17 | 2015-07-23 | Taiwan Semiconductor Manufacturing Company, Ltd. | Interconnect Structure And Method of Forming |
| US9054308B1 (en) | 2014-03-04 | 2015-06-09 | Sandisk 3D Llc | Plasma reduction method for modifying metal oxide stoichiometry in ReRAM |
| US10854472B2 (en) * | 2014-03-19 | 2020-12-01 | Globalfoundries Inc. | Method for forming a metal gate including de-oxidation of an oxidized surface of the metal gate utilizing a reducing agent |
| US9469912B2 (en) | 2014-04-21 | 2016-10-18 | Lam Research Corporation | Pretreatment method for photoresist wafer processing |
| US20150380296A1 (en) * | 2014-06-25 | 2015-12-31 | Lam Research Corporation | Cleaning of carbon-based contaminants in metal interconnects for interconnect capping applications |
| US9472377B2 (en) | 2014-10-17 | 2016-10-18 | Lam Research Corporation | Method and apparatus for characterizing metal oxide reduction |
| US9558955B2 (en) * | 2015-02-03 | 2017-01-31 | Taiwan Semiconductor Manufacturing Co., Ltd. | Formation method of semiconductor device that includes performing hydrogen-containing plasma treatment on metal gate stack |
| US9397011B1 (en) * | 2015-04-13 | 2016-07-19 | Lam Research Corporation | Systems and methods for reducing copper contamination due to substrate processing chambers with components made of alloys including copper |
| US9524905B1 (en) | 2015-08-31 | 2016-12-20 | Sandisk Technologies Llc | Nitrided tungsten via |
| US20180144973A1 (en) * | 2016-11-01 | 2018-05-24 | Applied Materials, Inc. | Electromigration Improvement Using Tungsten For Selective Cobalt Deposition On Copper Surfaces |
| US10443146B2 (en) | 2017-03-30 | 2019-10-15 | Lam Research Corporation | Monitoring surface oxide on seed layers during electroplating |
| KR102331718B1 (en) | 2017-06-08 | 2021-11-26 | 삼성전자주식회사 | Methods of manufacturing a semiconductor device |
| US11152455B2 (en) | 2019-09-23 | 2021-10-19 | Taiwan Semiconductor Manufacturing Company, Ltd. | Method to reduce breakdown failure in a MIM capacitor |
| US11508617B2 (en) | 2019-10-24 | 2022-11-22 | Applied Materials, Inc. | Method of forming interconnect for semiconductor device |
| US11257677B2 (en) * | 2020-01-24 | 2022-02-22 | Applied Materials, Inc. | Methods and devices for subtractive self-alignment |
| US11430729B2 (en) | 2020-09-16 | 2022-08-30 | Taiwan Semiconductor Manufacturing Company, Ltd. | MIM capacitor with a symmetrical capacitor insulator structure |
Citations (49)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4308089A (en) * | 1979-05-28 | 1981-12-29 | Hitachi, Ltd. | Method for preventing corrosion of Al and Al alloys |
| US4420386A (en) * | 1983-04-22 | 1983-12-13 | White Engineering Corporation | Method for pure ion plating using magnetic fields |
| US4436761A (en) * | 1981-07-30 | 1984-03-13 | Agency Of Industrial Science & Technology | Method for treatment of metal substrate for growth of hydrogen-containing semiconductor film |
| US4849797A (en) * | 1987-01-23 | 1989-07-18 | Hosiden Electronics Co., Ltd. | Thin film transistor |
| US4980196A (en) * | 1990-02-14 | 1990-12-25 | E. I. Du Pont De Nemours And Company | Method of coating steel substrate using low temperature plasma processes and priming |
| US4994410A (en) * | 1988-04-04 | 1991-02-19 | Motorola, Inc. | Method for device metallization by forming a contact plug and interconnect using a silicide/nitride process |
| US5000113A (en) * | 1986-12-19 | 1991-03-19 | Applied Materials, Inc. | Thermal CVD/PECVD reactor and use for thermal chemical vapor deposition of silicon dioxide and in-situ multi-step planarized process |
| US5121706A (en) * | 1987-10-16 | 1992-06-16 | The Curators Of The University Of Missouri | Apparatus for applying a composite insulative coating to a substrate |
| US5135775A (en) * | 1990-11-02 | 1992-08-04 | Thyssen Edelstalhwerke Ag | Process for plasma-chemical cleaning prior to pvd or pecvd coating |
| US5232872A (en) * | 1989-05-09 | 1993-08-03 | Fujitsu Limited | Method for manufacturing semiconductor device |
| US5232871A (en) * | 1990-12-27 | 1993-08-03 | Intel Corporation | Method for forming a titanium nitride barrier layer |
| US5288527A (en) * | 1990-09-03 | 1994-02-22 | Saint Gobain Vitrage International C/O Saint Gobain Recherche | Silicon nitride thin films with improved properties |
| US5409543A (en) * | 1992-12-22 | 1995-04-25 | Sandia Corporation | Dry soldering with hot filament produced atomic hydrogen |
| US5420044A (en) * | 1991-05-21 | 1995-05-30 | Canon Kabushiki Kaisha | Method for producing non-monocrystalline semiconductor device |
| US5441768A (en) * | 1994-02-08 | 1995-08-15 | Applied Materials, Inc. | Multi-step chemical vapor deposition method for thin film transistors |
| US5451263A (en) * | 1994-02-03 | 1995-09-19 | Harris Corporation | Plasma cleaning method for improved ink brand permanency on IC packages with metallic parts |
| US5458907A (en) * | 1993-02-12 | 1995-10-17 | Nec Corporation | Method of manufacturing printed circuit boards having an oxidation proof coating on a copper or copper alloy circuit pattern |
| US5599736A (en) * | 1995-06-28 | 1997-02-04 | Vanguard International Semiconductor Corporation | Fabrication method for polysilicon contact plugs |
| US5643834A (en) * | 1991-07-01 | 1997-07-01 | Sumitomo Electric Industries, Ltd. | Process for manufacturing a semiconductor substrate comprising laminated copper, silicon oxide and silicon nitride layers |
| US5725938A (en) * | 1994-08-23 | 1998-03-10 | Lucent Technologies Inc. | Metallization of ceramic through application of an adherent reducible layer |
| US5726097A (en) * | 1994-07-28 | 1998-03-10 | Sony Corporation | Method of forming multilevel interconnections using high density plasma metal clean |
| US5736002A (en) * | 1994-08-22 | 1998-04-07 | Sharp Microelectronics Technology, Inc. | Methods and equipment for anisotropic, patterned conversion of copper into selectively removable compounds and for removal of same |
| US5780163A (en) * | 1996-06-05 | 1998-07-14 | Dow Corning Corporation | Multilayer coating for microelectronic devices |
| US5801098A (en) * | 1996-09-03 | 1998-09-01 | Motorola, Inc. | Method of decreasing resistivity in an electrically conductive layer |
| US5821168A (en) * | 1997-07-16 | 1998-10-13 | Motorola, Inc. | Process for forming a semiconductor device |
| US5834371A (en) * | 1997-01-31 | 1998-11-10 | Tokyo Electron Limited | Method and apparatus for preparing and metallizing high aspect ratio silicon semiconductor device contacts to reduce the resistivity thereof |
| US5843847A (en) * | 1996-04-29 | 1998-12-01 | Applied Materials, Inc. | Method for etching dielectric layers with high selectivity and low microloading |
| US5866213A (en) * | 1994-06-03 | 1999-02-02 | Tokyo Electron Limited | Method for producing thin films by low temperature plasma-enhanced chemical vapor deposition using a rotating susceptor reactor |
| US5893752A (en) * | 1997-12-22 | 1999-04-13 | Motorola, Inc. | Process for forming a semiconductor device |
| US5899720A (en) * | 1994-12-28 | 1999-05-04 | Nec Corporation | Process of fabricating salicide structure from high-purity reproducible cobalt layer without sacrifice of leakage current and breakdown voltage of P-N junction |
| US5950083A (en) * | 1994-09-29 | 1999-09-07 | Nec Corporation | Method for fabricating CMOS transistor with self-aligned silicide (salicide) structure |
| US5970378A (en) * | 1996-09-03 | 1999-10-19 | Taiwan Semiconductor Manufacturing Company, Ltd. | Multi-step plasma treatment process for forming low resistance titanium nitride layer |
| US5975912A (en) * | 1994-06-03 | 1999-11-02 | Materials Research Corporation | Low temperature plasma-enhanced formation of integrated circuits |
| US6013574A (en) * | 1996-01-30 | 2000-01-11 | Advanced Micro Devices, Inc. | Method of forming low resistance contact structures in vias arranged between two levels of interconnect lines |
| US6033537A (en) * | 1996-12-26 | 2000-03-07 | Kabushiki Kaisha Toshiba | Sputtering target and method of manufacturing a semiconductor device |
| US6033584A (en) * | 1997-12-22 | 2000-03-07 | Advanced Micro Devices, Inc. | Process for reducing copper oxide during integrated circuit fabrication |
| US6071813A (en) * | 1997-10-20 | 2000-06-06 | Advanced Micro Devices, Inc. | Method and system for electrical coupling to copper interconnects |
| US6100184A (en) * | 1997-08-20 | 2000-08-08 | Sematech, Inc. | Method of making a dual damascene interconnect structure using low dielectric constant material for an inter-level dielectric layer |
| US6143640A (en) * | 1997-09-23 | 2000-11-07 | International Business Machines Corporation | Method of fabricating a stacked via in copper/polyimide beol |
| US6172421B1 (en) * | 1998-08-11 | 2001-01-09 | Advanced Micro Devices, Inc. | Semiconductor device having an intermetallic layer on metal interconnects |
| US6174810B1 (en) * | 1998-04-06 | 2001-01-16 | Motorola, Inc. | Copper interconnect structure and method of formation |
| US6251775B1 (en) * | 1999-04-23 | 2001-06-26 | International Business Machines Corporation | Self-aligned copper silicide formation for improved adhesion/electromigration |
| US6287977B1 (en) * | 1998-07-31 | 2001-09-11 | Applied Materials, Inc. | Method and apparatus for forming improved metal interconnects |
| US6303505B1 (en) * | 1998-07-09 | 2001-10-16 | Advanced Micro Devices, Inc. | Copper interconnect with improved electromigration resistance |
| US6319728B1 (en) * | 1998-06-05 | 2001-11-20 | Applied Materials, Inc. | Method for treating a deposited film for resistivity reduction |
| US6355571B1 (en) * | 1998-11-17 | 2002-03-12 | Applied Materials, Inc. | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US6365527B1 (en) * | 2000-10-06 | 2002-04-02 | United Microelectronics Corp. | Method for depositing silicon carbide in semiconductor devices |
| US6475903B1 (en) * | 1993-12-28 | 2002-11-05 | Intel Corporation | Copper reflow process |
| US20040155268A1 (en) * | 2003-02-06 | 2004-08-12 | Infineon Technologies North America Corp. | Method and apparatus for improving the electrical resistance of conductive paths |
Family Cites Families (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7A (en) * | 1836-08-10 | Thomas blanchard | ||
| US62A (en) * | 1836-10-20 | Cooking-stove | ||
| US11A (en) * | 1836-08-10 | |||
| US1116912A (en) * | 1914-05-14 | 1914-11-10 | Lawson Mfg Company | Gas-burner. |
| US2155862A (en) * | 1936-01-15 | 1939-04-25 | Manifold Supplies Company | Duplicating ink |
| US2290166A (en) * | 1941-02-17 | 1942-07-21 | Edward C Craig | Close fitting, insulating, watertight underwear |
| US2319532A (en) * | 1941-04-29 | 1943-05-18 | Willard Storage Battery Co | Liquid dispensing apparatus |
| US2319533A (en) * | 1941-08-15 | 1943-05-18 | Willard Storage Battery Co | Charging cover |
| US4072724A (en) * | 1975-11-03 | 1978-02-07 | The Goodyear Tire & Rubber Company | Process for preparing hindered alkenyl phenols |
| DE2815074C2 (en) * | 1978-04-07 | 1983-03-03 | Max-Planck-Gesellschaft zur Förderung der Wissenschaften e.V., 3400 Göttingen | Spectrophotometers for medical purposes |
| DE3574997D1 (en) | 1984-03-03 | 1990-02-01 | Stc Plc | PULSATING PLASMA PROCESS. |
| JPS61579A (en) | 1984-06-14 | 1986-01-06 | Ricoh Co Ltd | Manufacture of thin film |
| JPS619579A (en) | 1984-06-25 | 1986-01-17 | Nippon Steel Corp | Corrosion protecting method of steel structure |
| JPS62158859A (en) | 1986-01-07 | 1987-07-14 | Sumitomo Electric Ind Ltd | Pretreatment |
| JPH01116912A (en) | 1987-10-29 | 1989-05-09 | Sony Corp | Magneto-resistance effect type magnetic head |
| JPH0338953A (en) | 1989-07-05 | 1991-02-20 | Nec Corp | Dialer integrated circuit for telephone set |
| DE69025244T2 (en) | 1989-08-25 | 1996-06-27 | Applied Materials Inc | Cleaning process for a system for treating semiconductor wafers |
| JPH0445534A (en) | 1990-06-12 | 1992-02-14 | Sony Corp | Selective etching method of thin copper film |
| JP2646811B2 (en) | 1990-07-13 | 1997-08-27 | ソニー株式会社 | Dry etching method |
| JP3038953B2 (en) | 1991-02-28 | 2000-05-08 | ソニー株式会社 | Wiring formation method |
| JPH05144811A (en) | 1991-11-22 | 1993-06-11 | Hitachi Ltd | Thin film semiconductor device and manufacture thereof |
| AU3726593A (en) | 1992-02-26 | 1993-09-13 | Materials Research Corporation | Ammonia plasma treatment of silicide contact surfaces in semiconductor devices |
| JPH0697111A (en) | 1992-09-11 | 1994-04-08 | Sony Corp | Formation of barrier metal |
| JPH06204191A (en) | 1992-11-10 | 1994-07-22 | Sony Corp | Surface processing method after formation of metallic plug |
| JP3399583B2 (en) | 1993-04-06 | 2003-04-21 | 東京エレクトロン株式会社 | Method for manufacturing semiconductor device |
| JPH07142416A (en) | 1993-06-21 | 1995-06-02 | Applied Materials Inc | Plasma chemical vapor deposition method of layer with improved interface |
| JPH0793276B2 (en) | 1993-12-14 | 1995-10-09 | アプライド マテリアルズ インコーポレイテッド | Thin film forming pretreatment method and thin film forming method |
| JP2837087B2 (en) | 1993-12-28 | 1998-12-14 | アプライド マテリアルズ インコーポレイテッド | Thin film formation method |
| US5738956A (en) * | 1994-01-28 | 1998-04-14 | Kyowa Hakko Kogyo | Agent for maintaining and recovering the function of lead storage battery and electrolyte for lead storage battery using the same |
| IT1271636B (en) | 1994-05-04 | 1997-06-04 | Alcatel Italia | METHOD FOR THE PREPARATION AND PASSIVATION OF THE HIGH-EMISSION SEMICONDUCTOR LASER TERMINAL MIRRORS AND RELATED DEVICE |
| KR0144956B1 (en) | 1994-06-10 | 1998-08-17 | 김광호 | Forming method of wiring |
| BE1009839A3 (en) | 1995-12-20 | 1997-10-07 | Cockerill Rech & Dev | Method and device for cleaning substrate metal. |
| JPH1019244A (en) | 1996-06-27 | 1998-01-23 | Nkk Corp | Drain exhaust device, alternating combustion furnace having drain exhaust device and drain exhaust method for alternating combustion furnace |
| GB2319533B (en) | 1996-11-22 | 2001-06-06 | Trikon Equip Ltd | Methods of forming a barrier layer |
| GB2319532B (en) | 1996-11-22 | 2001-01-31 | Trikon Equip Ltd | Method and apparatus for treating a semiconductor wafer |
| KR19980064441A (en) | 1996-12-20 | 1998-10-07 | 윌리엄비.켐플러 | Selective bonding of conductive material to semiconductor device surface |
| DE19702124A1 (en) | 1997-01-22 | 1998-07-23 | Linde Ag | Workpiece surface cleaning, activating, wetting and/or coating |
| DE19717698A1 (en) | 1997-04-26 | 1998-10-29 | Fraunhofer Ges Forschung | Method and device for cleaning activation of electrical conductor tracks and circuit board surfaces |
| JPH1116912A (en) | 1997-06-25 | 1999-01-22 | Hitachi Ltd | Manufacture of semiconductor integrated circuit device and manufacture device of semiconductor integrated circuit device |
| US6872429B1 (en) * | 1997-06-30 | 2005-03-29 | Applied Materials, Inc. | Deposition of tungsten nitride using plasma pretreatment in a chemical vapor deposition chamber |
| TW507015B (en) * | 1997-12-02 | 2002-10-21 | Applied Materials Inc | In-situ, preclean of wafers prior to a chemical vapor deposition titanium deposition step |
| JP3501937B2 (en) | 1998-01-30 | 2004-03-02 | 富士通株式会社 | Method for manufacturing semiconductor device |
| FR2775986B1 (en) | 1998-03-10 | 2000-05-05 | Air Liquide | PROCESS AND INSTALLATION FOR SURFACE TREATMENT OF A METAL PART |
| KR100290835B1 (en) * | 1998-06-23 | 2001-07-12 | 윤종용 | Manufacturing method of semiconductor device |
| US6492266B1 (en) * | 1998-07-09 | 2002-12-10 | Advanced Micro Devices, Inc. | Method of forming reliable capped copper interconnects |
| US6097111A (en) * | 1998-12-29 | 2000-08-01 | Anrod; Stephen C. | Mode selectable power receptacle |
-
1999
- 1999-07-30 US US09/365,129 patent/US6355571B1/en not_active Expired - Lifetime
-
2000
- 2000-05-05 TW TW089108656A patent/TW459304B/en not_active IP Right Cessation
- 2000-05-08 EP EP00109718A patent/EP1073106B1/en not_active Revoked
- 2000-05-08 DE DE60040507T patent/DE60040507D1/en not_active Expired - Fee Related
- 2000-07-29 KR KR1020000043933A patent/KR100773188B1/en active IP Right Grant
- 2000-07-31 JP JP2000231957A patent/JP2001093902A/en not_active Withdrawn
-
2001
- 2001-12-07 US US10/013,182 patent/US6700202B2/en not_active Expired - Lifetime
-
2003
- 2003-09-04 US US10/655,438 patent/US6946401B2/en not_active Expired - Fee Related
-
2005
- 2005-06-29 US US11/169,337 patent/US20050263900A1/en not_active Abandoned
-
2008
- 2008-10-24 US US12/257,806 patent/US8183150B2/en not_active Expired - Fee Related
Patent Citations (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4308089A (en) * | 1979-05-28 | 1981-12-29 | Hitachi, Ltd. | Method for preventing corrosion of Al and Al alloys |
| US4436761A (en) * | 1981-07-30 | 1984-03-13 | Agency Of Industrial Science & Technology | Method for treatment of metal substrate for growth of hydrogen-containing semiconductor film |
| US4420386A (en) * | 1983-04-22 | 1983-12-13 | White Engineering Corporation | Method for pure ion plating using magnetic fields |
| US5000113A (en) * | 1986-12-19 | 1991-03-19 | Applied Materials, Inc. | Thermal CVD/PECVD reactor and use for thermal chemical vapor deposition of silicon dioxide and in-situ multi-step planarized process |
| US4849797A (en) * | 1987-01-23 | 1989-07-18 | Hosiden Electronics Co., Ltd. | Thin film transistor |
| US5121706A (en) * | 1987-10-16 | 1992-06-16 | The Curators Of The University Of Missouri | Apparatus for applying a composite insulative coating to a substrate |
| US4994410A (en) * | 1988-04-04 | 1991-02-19 | Motorola, Inc. | Method for device metallization by forming a contact plug and interconnect using a silicide/nitride process |
| US5232872A (en) * | 1989-05-09 | 1993-08-03 | Fujitsu Limited | Method for manufacturing semiconductor device |
| US4980196A (en) * | 1990-02-14 | 1990-12-25 | E. I. Du Pont De Nemours And Company | Method of coating steel substrate using low temperature plasma processes and priming |
| US5288527A (en) * | 1990-09-03 | 1994-02-22 | Saint Gobain Vitrage International C/O Saint Gobain Recherche | Silicon nitride thin films with improved properties |
| US5135775A (en) * | 1990-11-02 | 1992-08-04 | Thyssen Edelstalhwerke Ag | Process for plasma-chemical cleaning prior to pvd or pecvd coating |
| US5232871A (en) * | 1990-12-27 | 1993-08-03 | Intel Corporation | Method for forming a titanium nitride barrier layer |
| US5420044A (en) * | 1991-05-21 | 1995-05-30 | Canon Kabushiki Kaisha | Method for producing non-monocrystalline semiconductor device |
| US5643834A (en) * | 1991-07-01 | 1997-07-01 | Sumitomo Electric Industries, Ltd. | Process for manufacturing a semiconductor substrate comprising laminated copper, silicon oxide and silicon nitride layers |
| US5409543A (en) * | 1992-12-22 | 1995-04-25 | Sandia Corporation | Dry soldering with hot filament produced atomic hydrogen |
| US5458907A (en) * | 1993-02-12 | 1995-10-17 | Nec Corporation | Method of manufacturing printed circuit boards having an oxidation proof coating on a copper or copper alloy circuit pattern |
| US6475903B1 (en) * | 1993-12-28 | 2002-11-05 | Intel Corporation | Copper reflow process |
| US5451263A (en) * | 1994-02-03 | 1995-09-19 | Harris Corporation | Plasma cleaning method for improved ink brand permanency on IC packages with metallic parts |
| US5441768A (en) * | 1994-02-08 | 1995-08-15 | Applied Materials, Inc. | Multi-step chemical vapor deposition method for thin film transistors |
| US5975912A (en) * | 1994-06-03 | 1999-11-02 | Materials Research Corporation | Low temperature plasma-enhanced formation of integrated circuits |
| US5866213A (en) * | 1994-06-03 | 1999-02-02 | Tokyo Electron Limited | Method for producing thin films by low temperature plasma-enhanced chemical vapor deposition using a rotating susceptor reactor |
| US5726097A (en) * | 1994-07-28 | 1998-03-10 | Sony Corporation | Method of forming multilevel interconnections using high density plasma metal clean |
| US5736002A (en) * | 1994-08-22 | 1998-04-07 | Sharp Microelectronics Technology, Inc. | Methods and equipment for anisotropic, patterned conversion of copper into selectively removable compounds and for removal of same |
| US5725938A (en) * | 1994-08-23 | 1998-03-10 | Lucent Technologies Inc. | Metallization of ceramic through application of an adherent reducible layer |
| US5950083A (en) * | 1994-09-29 | 1999-09-07 | Nec Corporation | Method for fabricating CMOS transistor with self-aligned silicide (salicide) structure |
| US5899720A (en) * | 1994-12-28 | 1999-05-04 | Nec Corporation | Process of fabricating salicide structure from high-purity reproducible cobalt layer without sacrifice of leakage current and breakdown voltage of P-N junction |
| US5599736A (en) * | 1995-06-28 | 1997-02-04 | Vanguard International Semiconductor Corporation | Fabrication method for polysilicon contact plugs |
| US6013574A (en) * | 1996-01-30 | 2000-01-11 | Advanced Micro Devices, Inc. | Method of forming low resistance contact structures in vias arranged between two levels of interconnect lines |
| US5843847A (en) * | 1996-04-29 | 1998-12-01 | Applied Materials, Inc. | Method for etching dielectric layers with high selectivity and low microloading |
| US5780163A (en) * | 1996-06-05 | 1998-07-14 | Dow Corning Corporation | Multilayer coating for microelectronic devices |
| US5801098A (en) * | 1996-09-03 | 1998-09-01 | Motorola, Inc. | Method of decreasing resistivity in an electrically conductive layer |
| US5970378A (en) * | 1996-09-03 | 1999-10-19 | Taiwan Semiconductor Manufacturing Company, Ltd. | Multi-step plasma treatment process for forming low resistance titanium nitride layer |
| US6033537A (en) * | 1996-12-26 | 2000-03-07 | Kabushiki Kaisha Toshiba | Sputtering target and method of manufacturing a semiconductor device |
| US5834371A (en) * | 1997-01-31 | 1998-11-10 | Tokyo Electron Limited | Method and apparatus for preparing and metallizing high aspect ratio silicon semiconductor device contacts to reduce the resistivity thereof |
| US6143128A (en) * | 1997-01-31 | 2000-11-07 | Tokyo Electron Limited | Apparatus for preparing and metallizing high aspect ratio silicon semiconductor device contacts to reduce the resistivity thereof |
| US5821168A (en) * | 1997-07-16 | 1998-10-13 | Motorola, Inc. | Process for forming a semiconductor device |
| US6100184A (en) * | 1997-08-20 | 2000-08-08 | Sematech, Inc. | Method of making a dual damascene interconnect structure using low dielectric constant material for an inter-level dielectric layer |
| US6143640A (en) * | 1997-09-23 | 2000-11-07 | International Business Machines Corporation | Method of fabricating a stacked via in copper/polyimide beol |
| US6071813A (en) * | 1997-10-20 | 2000-06-06 | Advanced Micro Devices, Inc. | Method and system for electrical coupling to copper interconnects |
| US5893752A (en) * | 1997-12-22 | 1999-04-13 | Motorola, Inc. | Process for forming a semiconductor device |
| US6033584A (en) * | 1997-12-22 | 2000-03-07 | Advanced Micro Devices, Inc. | Process for reducing copper oxide during integrated circuit fabrication |
| US6174810B1 (en) * | 1998-04-06 | 2001-01-16 | Motorola, Inc. | Copper interconnect structure and method of formation |
| US6319728B1 (en) * | 1998-06-05 | 2001-11-20 | Applied Materials, Inc. | Method for treating a deposited film for resistivity reduction |
| US6303505B1 (en) * | 1998-07-09 | 2001-10-16 | Advanced Micro Devices, Inc. | Copper interconnect with improved electromigration resistance |
| US6287977B1 (en) * | 1998-07-31 | 2001-09-11 | Applied Materials, Inc. | Method and apparatus for forming improved metal interconnects |
| US6172421B1 (en) * | 1998-08-11 | 2001-01-09 | Advanced Micro Devices, Inc. | Semiconductor device having an intermetallic layer on metal interconnects |
| US6355571B1 (en) * | 1998-11-17 | 2002-03-12 | Applied Materials, Inc. | Method and apparatus for reducing copper oxidation and contamination in a semiconductor device |
| US6700202B2 (en) * | 1998-11-17 | 2004-03-02 | Applied Materials, Inc. | Semiconductor device having reduced oxidation interface |
| US6251775B1 (en) * | 1999-04-23 | 2001-06-26 | International Business Machines Corporation | Self-aligned copper silicide formation for improved adhesion/electromigration |
| US6365527B1 (en) * | 2000-10-06 | 2002-04-02 | United Microelectronics Corp. | Method for depositing silicon carbide in semiconductor devices |
| US20040155268A1 (en) * | 2003-02-06 | 2004-08-12 | Infineon Technologies North America Corp. | Method and apparatus for improving the electrical resistance of conductive paths |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150001728A1 (en) * | 2013-06-26 | 2015-01-01 | Taiwan Semiconductor Manufacturing Company, Ltd. | Pre-treatment method for metal-oxide reduction and device formed |
Also Published As
| Publication number | Publication date |
|---|---|
| US6355571B1 (en) | 2002-03-12 |
| EP1073106A3 (en) | 2005-08-24 |
| US6700202B2 (en) | 2004-03-02 |
| US20020081856A1 (en) | 2002-06-27 |
| US6946401B2 (en) | 2005-09-20 |
| US20040046260A1 (en) | 2004-03-11 |
| KR20010039772A (en) | 2001-05-15 |
| US8183150B2 (en) | 2012-05-22 |
| JP2001093902A (en) | 2001-04-06 |
| US20090050902A1 (en) | 2009-02-26 |
| TW459304B (en) | 2001-10-11 |
| DE60040507D1 (en) | 2008-11-27 |
| EP1073106B1 (en) | 2008-10-15 |
| EP1073106A2 (en) | 2001-01-31 |
| KR100773188B1 (en) | 2007-11-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6946401B2 (en) | Plasma treatment for copper oxide reduction | |
| US6734102B2 (en) | Plasma treatment for copper oxide reduction | |
| US10770346B2 (en) | Selective cobalt removal for bottom up gapfill | |
| US10049891B1 (en) | Selective in situ cobalt residue removal | |
| US6949450B2 (en) | Method for integrated in-situ cleaning and subsequent atomic layer deposition within a single processing chamber | |
| US7026238B2 (en) | Reliability barrier integration for Cu application | |
| US9418889B2 (en) | Selective formation of dielectric barriers for metal interconnects in semiconductor devices | |
| US6297147B1 (en) | Plasma treatment for ex-situ contact fill | |
| KR100652334B1 (en) | A method for depositing a metal layer on a semiconductor interconnect structure having a capping layer | |
| TW473869B (en) | Plasma preclean with argon, helium, and hydrogen gases | |
| EP1081751A2 (en) | Methods of pre-cleaning dielectric layers of substrates | |
| EP0799903A2 (en) | Methods of sputtering a metal onto a substrate and semiconductor processing apparatus | |
| WO2001004936A1 (en) | Method of cleaning a semiconductor device processing chamber after a copper etch process | |
| US7071095B2 (en) | Barrier metal re-distribution process for resistivity reduction | |
| US11024537B2 (en) | Methods and apparatus for hybrid feature metallization | |
| KR100670618B1 (en) | Sequential sputter and reactive precleans of vias and contacts | |
| WO2002046489A1 (en) | Method for integrated in-situ cleaning and subsequent atomic layer deposition within a single processing chamber | |
| TWI853995B (en) | Methods and apparatus for hybrid feature metallization | |
| JP3639142B2 (en) | Manufacturing method of semiconductor device | |
| JPH09181171A (en) | Multilayered wiring forming method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: APPLIED MATERIALS, INC., CALIFORNIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:HUANG, JUDY H.;BENCHER, CHRISTOPHER DENNIS;RATHI, SUDHA;AND OTHERS;REEL/FRAME:016861/0566;SIGNING DATES FROM 19990913 TO 19990914 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |