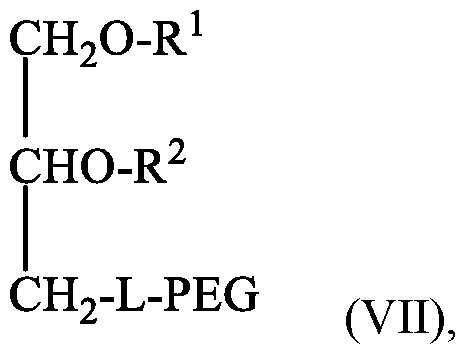WO2018232330A1 - Therapeutic compositions and methods for treating hepatitis b - Google Patents
Therapeutic compositions and methods for treating hepatitis b Download PDFInfo
- Publication number
- WO2018232330A1 WO2018232330A1 PCT/US2018/037890 US2018037890W WO2018232330A1 WO 2018232330 A1 WO2018232330 A1 WO 2018232330A1 US 2018037890 W US2018037890 W US 2018037890W WO 2018232330 A1 WO2018232330 A1 WO 2018232330A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- lipid
- mol
- sirna
- ifn
- peg
- Prior art date
Links
- 0 C1C2C1*C*2 Chemical compound C1C2C1*C*2 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/12—Viral antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/212—IFN-alpha
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/543—Lipids, e.g. triglycerides; Polyamines, e.g. spermine or spermidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6905—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion
- A61K47/6907—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion the form being a microemulsion, nanoemulsion or micelle
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Liposomes
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes, liposomes coated with polymers
- A61K9/1272—Non-conventional liposomes, e.g. PEGylated liposomes, liposomes coated with polymers with substantial amounts of non-phosphatidyl, i.e. non-acylglycerophosphate, surfactants as bilayer-forming substances, e.g. cationic lipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55555—Liposomes; Vesicles, e.g. nanoparticles; Spheres, e.g. nanospheres; Polymers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2730/00—Reverse transcribing DNA viruses
- C12N2730/00011—Details
- C12N2730/10011—Hepadnaviridae
- C12N2730/10111—Orthohepadnavirus, e.g. hepatitis B virus
- C12N2730/10134—Use of virus or viral component as vaccine, e.g. live-attenuated or inactivated virus, VLP, viral protein
Definitions
- Hepatitis B virus (abbreviated as "HBV") is a member of the Hepadnavirus family.
- the virus particle (sometimes referred to as a virion) includes an outer lipid envelope and an icosahedral nucleocapsid core composed of protein.
- the nucleocapsid encloses the viral DNA and a DNA polymerase that has reverse transcriptase activity.
- the outer envelope contains embedded proteins that are involved in viral binding of, and entry into, susceptible cells, typically liver hepatocytes.
- filamentous and spherical bodies lacking a core can be found in the serum of infected individuals. These particles are not infectious and are composed of the lipid and protein that forms part of the surface of the virion, which is called the surface antigen (HBsAg), and is produced in excess during the life cycle of the virus.
- HBsAg surface antigen
- the genome of HBV is made of circular DNA, but it is unusual because the DNA is not fully double-stranded. One end of the full length strand is linked to the viral DNA polymerase.
- the genome is 3020-3320 nucleotides long (for the full-length strand) and 1700-2800 nucleotides long (for the shorter strand).
- the negative-sense (non-coding) is complementary to the viral mRNA.
- the fully double stranded viral DNA (covalently closed circular DNA, or cccDNA) is found in the nucleus soon after infection of the cell.
- C covalently closed circular DNA
- P X
- S S.
- the core protein is coded for by gene C
- HBeAg HBcAg
- HBeAg is produced by proteolytic processing of the pre-core protein.
- the DNA polymerase is encoded by gene P.
- Gene S is the gene that codes for the surface antigen (HBsAg).
- the HBsAg gene is one long open reading frame but contains three in frame "start” (ATG) codons that divide the gene into three sections, pre-Sl, pre-S2, and S.
- the virus is transmitted by exposure to infectious blood or body fluids. Perinatal infection can also be a major route of infection.
- the acute illness causes liver inflammation, vomiting, jaundice, and possibly death.
- Chronic hepatitis B may eventually cause cirrhosis and liver cancer.
- Hepatitis D virus is a small circular enveloped RNA virus that can propagate only in the presence of the hepatitis B virus (HBV).
- HBV hepatitis B virus
- HDV requires the HBV surface antigen protein to propagate itself. Infection with both HBV and HDV results in more severe complications compared to infection with HBV alone. These complications include a greater likelihood of experiencing liver failure in acute infections and a rapid progression to liver cirrhosis, with an increased chance of developing liver cancer in chronic infections.
- hepatitis D In combination with hepatitis B virus, hepatitis D has the highest mortality rate of all the hepatitis infections.
- the routes of transmission of HDV are similar to those for HBV. Infection is largely restricted to persons at high risk of HBV infection, particularly injecting drug users and persons receiving clotting factor concentrates.
- compositions and methods for the treatment of HBV infection humans as well as for the treatment of HBV/HDV infection in humans.
- the present invention provides therapeutic combinations and therapeutic methods that are useful for treating viral infections such as HBV and/or HDV.
- certain embodiments of the invention provide a method for treating hepatitis B in a human, comprising administering to the human:
- siRNA 1 SEQ ID NO: l and 2
- siRNA 2 SEQ ID NO:3 and 4
- siRNA 3 SEQ ID NO:5 and 6
- IFN interferon
- Certain embodiments of the invention also provide a method for treating hepatitis D in a human, comprising administering to the human:
- siRNA 1 SEQ ID NO: l and 2
- siRNA 2 SEQ ID NO:3 and 4
- siRNA 3 SEQ ID NO:5 and 6
- IFN interferon
- Certain embodiments of the invention provide a lipid nanoparticle formulation in combination with an interferon for the prophylactic or therapeutic treatment of hepatitis B, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6).
- siRNA 1 SEQ ID NO: l and 2
- siRNA 2 SEQ ID NO:3 and 4
- siRNA 3 SEQ ID NO: 5 and 6
- Certain embodiments of the invention provide the use a lipid nanoparticle formulation in combination with an interferon (IFN) for the preparation of a medicament for treating hepatitis B in a human, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
- IFN interferon
- lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
- lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6);
- Certain embodiments of the invention provide a method for treating hepatitis B and/or hepatitis D in a human, comprising administering to the human:
- siRNA 1 SEQ ID NO: l and 2
- siRNA 2 SEQ ID NO:3 and 4
- siRNA 3 SEQ ID NO:5 and 6
- IFN interferon
- the lipid nanoparticle formulation described herein includes a cocktail of siRNAs (i.e., siRNA 1, siRNA 2 and siRNA3) encapsulated within lipid particles.
- the different siRNA molecules are co-encapsulated in the same lipid particle.
- each type of siRNA species present in the cocktail is encapsulated in its own particle.
- some siRNA species are co-encapsulated in the same particle while other siRNA species are encapsulated in different particles.
- the lipid nanoparticle formulation further comprises a cationic lipid and a non-cationic lipid. Accordingly, in certain embodiments, the lipid nanoparticle formulation comprises a population of lipid nanoparticles, wherein each lipid nanoparticle comprises a cationic lipid, a non-cationic lipid and at least one siRNA molecule selected from siRNA 1, siRNA 2 and siRNA 3. In certain embodiments, the lipid nanoparticle formulation comprises a population of lipid nanoparticles, wherein each lipid nanoparticle comprises a cationic lipid, a non-cationic lipid, siRNA 1, siRNA 2 and siRNA 3.
- the cationic lipid is selected from the group consisting of 1,2- dilinoleyloxy-N,N-dimethylaminopropane (DLinDMA), 1 ,2-dilinolenyloxy-N,N- dimethylaminopropane (DLenDMA), l,2-di-Y-linolenyloxy-N,N-dimethylaminopropane ( ⁇ - DLenDMA; Compound (15)) , 3-((6Z,9Z,28Z,31Z)-heptatriaconta-6,9,28,31-tetraen-19-yloxy)- N,N-dimethylpropan-l -amine (DLin-MP-DMA; Compound (8)), (6Z,9Z,28Z,31Z)- heptatriaconta-6,9,28,31-tetraen-19-yl 4-(dimethylamino)butanoate) (Compound (8)), (6Z
- the cationic lipid is: or a salt thereof.
- the non-cationic lipid is cholesterol or a derivative thereof. In certain embodiments, the non-cationic lipid is a phospholipid.
- the non-cationic lipid is a mixture of a phospholipid and cholesterol or a derivative thereof.
- the phospholipid is selected from the group consisting of dipalmitoylphosphatidylcholine (DPPC), distearoylphosphatidylcholine (DSPC), and a mixture thereof.
- DPPC dipalmitoylphosphatidylcholine
- DSPC distearoylphosphatidylcholine
- the phospholipid is DSPC.
- the lipid nanoparticle formulation further comprises a conjugated lipid that inhibits aggregation of particles.
- the conjugated lipid that inhibits aggregation of particles is a polyethyleneglycol (PEG)-lipid conjugate.
- PEG polyethyleneglycol
- the PEG-lipid conjugate is selected from the group consisting of a PEG-diacylglycerol (PEG-DAG) conjugate, a PEG-dialkyloxypropyl (PEG-DAA) conjugate, a PEG-phospholipid conjugate, a PEG-ceramide (PEG-Cer) conjugate, a PEG- dimyristyloxypropyl (PEG-DMA) conjugate and a mixture thereof.
- PEG-DAG PEG-diacylglycerol
- PEG-DAA PEG-dialkyloxypropyl
- PEG-phospholipid conjugate a PEG-ceramide (PEG-Cer) conjugate
- PEG-Cer PEG- dimyristyloxypropyl
- PEG-DMA PEG- dimyristyloxypropyl
- the PEG-lipid conjugate is a PEG-C-DMA conjugate.
- the lipid nanoparticle formulation comprises a population of lipid nanoparticles.
- the cationic lipid generally comprises from about 48 mol % to about 62 mol % of the total lipid present in each particle.
- the lipid nanoparticle formulation comprises a phospholipid and cholesterol or cholesterol derivative, wherein the phospholipid generally comprises from about 7 mol % to about 17 mol % of the total lipid present in each particle and the cholesterol or derivative thereof generally comprises from about 25 mol % to about 40 mol % of the total lipid present in each particle.
- the conjugated lipid that inhibits aggregation of particles generally comprises from about 0.5 mol % to about 3 mol % of the total lipid present in each particle.
- Interferons are a group of signaling proteins made and released by host cells in response to the presence of certain pathogens, such as, e.g., viruses, bacteria, and parasites, as well as to the presence of tumor cells. More than twenty distinct IFN genes and proteins have been identified in animals, including humans. Such interferons may be used to practice the claimed invention.
- the IFN is IFN alpha (IFN-a), IFN beta (IFN- ⁇ ) or IFN lambda (IFN- ⁇ ).
- the IFN is pegylated.
- the IFN is IFN-a.
- the IFN-a is pegylated IFN-a2a or peglyated IFN- a2b.
- the IFN is IFN- ⁇ .
- the IFN- ⁇ is pegylated IFN-beta (e.g., TRK-560; Toray Industries).
- the IFN is IFN lambda (IFN- ⁇ ).
- the lipid nanoparticle formulation is administered via injection.
- the IFN is administered via injection.
- the lipid nanoparticle formulation and the IFN are administered separately.
- the lipid nanoparticle formulation and the IFN are administered sequentially.
- the lipid nanoparticle formulation is administered first and the IFN is administered second.
- the IFN is administered first and the lipid nanoparticle formulation is administered second.
- the lipid nanoparticle formulation and the IFN are administered concurrently.
- the lipid nanoparticle formulation and/or the IFN are administered two or more times.
- a pharmaceutical composition comprising the lipid nanoparticle formulation, the IFN and a pharmaceutically acceptable carrier is administered to the human.
- methods of the invention further comprise administering at least one additional therapeutic agent.
- the at least one additional therapeutic agent is selected from the group consisting of:
- lipid nanoparticle formulation in combination with an interferon for the prophylactic or therapeutic treatment of hepatitis B, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6).
- the term "in combination with” refers to the simultaneous or sequential use of a lipid nanoparticle formulation and an interferon, as well as compositions comprising a lipid nanoparticle formulation and an interferon.
- Certain embodiments of the invention provide the use of a lipid nanoparticle formulation in combination with an interferon (IFN) for the preparation of a medicament for treating hepatitis B in a human, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
- IFN interferon
- lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
- the composition is a pharmaceutical composition further comprising a pharmaceutically acceptable carrier.
- the composition further comprises at least one additional therapeutic agent (e.g., a Category I, II and/or III agent discussed herein).
- at least one additional therapeutic agent e.g., a Category I, II and/or III agent discussed herein.
- lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO: 3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6); (b) an interferon; and
- the kit further comprises at least one additional therapeutic agent (e.g., a Category I, II and/or III agent discussed herein).
- at least one additional therapeutic agent e.g., a Category I, II and/or III agent discussed herein.
- Category I treatments are directed to the use of agents that control, e.g., inhibit, viral replication.
- the reverse transcriptase inhibitor is a nucleoside analog.
- the reverse transcriptase inhibitor is a nucleoside analog reverse-transcriptase inhibitor (NARTI or NRTI).
- the reverse transcriptase inhibitor is a nucleotide analog reverse- transcriptase inhibitor (NtARTI or NtRTI).
- reverse transcriptase inhibitor includes, but is not limited to: entecavir, clevudine, telbivudine, lamivudine, adefovir, and tenofovir, tenofovir disoproxil, tenofovir alafenamide, tenofovir disoproxil fumarate, adefovir dipivoxil, (lR,2R,3R,5R)-3-(6-amino-9H- 9-purinyl)-2-fluoro-5-(hydroxymethyl)-4-methylenecyclopentan-l-ol (described in U.S. Patent No.
- reverse transcriptase inhibitor includes, but is not limited to, entecavir, lamivudine, and (lR,2R,3R,5R)-3-(6-amino-9H-9-purinyl)-2-fluoro-5-(hydroxymethyl)-4- methylenecyclopentan- 1 -ol .
- reverse transcriptase inhibitor includes, but is not limited to a covalently bound phosphoramidate or phosphonamidate moiety of the above-mentioned reverse transcriptase inhibitors, or as described in, for example, U.S. Patent No. 8,816,074, US 2011/0245484 Al, and US 2008/0286230A1.
- reverse transcriptase inhibitor includes, but is not limited to, nucleotide analogs that comprise a phosphoramidate moiety, such as, methyl ((((lR,3R,4R,5R)-3-(6-amino-9H- purin-9-yl)-4-fluoro-5-hydroxy-2-methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate and methyl (((lR,2R,3R,4R)-3-fluoro-2-hydroxy-5-methylene-4-(6-oxo-l,6- dihydro-9H-purin-9-yl)cyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate.
- nucleotide analogs that comprise a phosphoramidate moiety, such as, methyl ((((lR,3R,4R,5R)-3-(6-amino-9H- purin-9-yl
- the individual diastereomers thereof which includes, for example, methyl ((R)- (((1 R, 3R,4R, 5R)-3 -(6-amino-9H-purin-9-yl)-4-fluoro-5 -hydroxy-2- methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate and methyl ((S)- ((( 1 R, 3R,4R, 5R)-3 -(6-amino-9H-purin-9-yl)-4-fluoro-5 -hydroxy-2- methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate.
- reverse transcriptase inhibitor includes, but is not limited to a phosphonamidate moiety, such as, tenofovir alafenamide, as well as those described in US 2008/0286230 Al .
- a phosphonamidate moiety such as, tenofovir alafenamide, as well as those described in US 2008/0286230 Al .
- Methods for preparing stereoselective phosphoramidate or phosphonamidate containing actives are described in, for example, U.S. Patent No. 8,816,074, as well as US 2011/0245484 Al and US 2008/0286230 Al .
- capsid inhibitor includes compounds that are capable inhibiting the expression and/or function of a capsid protein either directly or indirectly.
- a capsid inhibitor may include, but is not limited to, any compound that inhibits capsid assembly, induces formation of non-capsid polymers, promotes excess capsid assembly or misdirected capsid assembly, affects capsid stabilization, and/or inhibits encapsidation of RNA.
- Capsid inhibitors also include any compound that inhibits capsid function in a downstream event(s) within the replication process (e.g., viral DNA synthesis, transport of relaxed circular DNA (rcDNA) into the nucleus, covalently closed circular DNA (cccDNA) formation, virus maturation, budding and/or release, and the like).
- the inhibitor detectably inhibits the expression level or biological activity of the capsid protein as measured, e.g., using an assay described herein.
- the inhibitor inhibits the level of rcDNA and downstream products of viral life cycle by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, at least 90%, at least 95% or at least 99%.
- capsid inhibitor includes compounds described in International Patent
- capsid inhibitor also includes the compounds Bay-41-4109 (see International Patent Application Publication Number WO/2013/144129), AT-61 (see International Patent Application Publication Number WO/1998/33501; and King, RW, et al., Antimicrob Agents Chemother., 1998, 42, 12, 3179-3186), DVR-01 and DVR-23 (see International Patent Application Publication Number WO 2013/006394; and Campagna, MR, et al., J. of Virology, 2013, 87, 12, 6931, and pharmaceutically acceptable salts thereof:
- capsid inhibitor also includes the compounds Compound 3, GLS-4, and NVR
- cccDNA Covalently closed circular DNA
- cccDNA formation inhibitor includes compounds that are capable of inhibiting the formation and/or stability of cccDNA either directly or indirectly.
- a cccDNA formation inhibitor may include, but is not limited to, any compound that inhibits capsid disassembly, rcDNA entry into the nucleus, and/or the conversion of rcDNA into cccDNA.
- the inhibitor detectably inhibits the formation and/or stability of the cccDNA as measured, e.g., using an assay described herein.
- the inhibitor inhibits the formation and/or stability of cccDNA by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%.
- cccDNA formation inhibitor includes compounds described in International Patent Application Publication Number WO2013130703, including the following compound:
- cccDNA formation inhibitor includes, but is not limited to those generally and specifically described in United States Patent Application Publication Number US
- cccDNA formation inhibitor includes, but is not limited to, 1- (phenylsulfonyl)-N-(pyridin-4-ylmethyl)-lH-indole-2-carboxamide; 1-Benzenesulfonyl- pyrrolidine-2-carboxylic acid (pyridin-4-ylmethyl)-amide; 2-(2-chloro-N-(2-chloro-5- (trifluoromethyl)phenyl)-4-(trifluoromethyl)phenylsulfonamido)-N-(pyridin-4- ylmethyl)acetamide; 2-(4-chloro-N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N- (pyridin-4-ylmethyl)acetamide; 2-(N-(2-chloro-5-(trifluoromethyl)phenyl)-4- (trifluoromethyl)phenyl
- Certain embodiments of the invention are directed to the use of agents that are HBV entry inhibitors.
- Entry inhibitors include Myrcludex-B, NTCP inhibitor small molecules, and FXR agonist EYP001 (see, e.g., Gripon, P., Cannie, I. and Urban, S. Efficient Inhibition of Hepatitis B Virus Infection by Acylated Peptides Derived from the Large Viral Surface Protein.
- the hepatitis B virus uses its surface lipopeptide pre-Sl for docking to mature liver cells via their sodium/bile acid cotransporter (NTCP) and subsequently entering the cells.
- NTCP sodium/bile acid cotransporter
- Myrcludex B is a synthetic N-acylated pre-Sl that can also dock to NTCP, blocking the virus's entry mechanism.
- Category II treatments are directed to the use of agents that reduce viral antigens.
- the oligomeric nucleotides can be designed to target one or more genes and/or transcripts of the HBV genome.
- Examples of such siRNA molecules are the siRNA molecules set forth in Example 1.
- the term oligomeric nucleotide targeted to the Hepatitis B genome includes Arrowhead- ARC-520 (see United States Patent Number 8,809,293; and Wooddell CI, et al., Molecular Therapy, 2013, 21, 5, 973-985).
- oligomeric nucleotide targeted to the Hepatitis B genome also includes isolated, double stranded, siRNA molecules, that each include a sense strand and an antisense strand that is hybridized to the sense strand.
- the siRNA target one or more genes and/or transcripts of the HBV genome.
- this term includes siRNA molecules that target GalNAc and REP 2139, REP-2165 (see, e.g., WO 2016/077321, Al-Mathtab et al., PLoS ONE 1 l(6):e0156667. doi: 10.1371/journal.pone.0156667 and Guillot et al., Poster P0556, EASL, 2015).
- the term "sAg secretion inhibitor” includes compounds that are capable of inhibiting, either directly or indirectly, the secretion of sAg (S, M and/or L surface antigens) bearing subviral particles and/or DNA containing viral particles from HBV-infected cells.
- the inhibitor detectably inhibits the secretion of sAg as measured, e.g., using assays known in the art or described herein, e.g., ELISA assay or by Western Blot.
- the inhibitor inhibits the secretion of sAg by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%.
- the inhibitor reduces serum levels of sAg in a patient by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%.
- sAg secretion inhibitor includes compounds described in United States Patent Number 8,921,381, as well as compounds described in United States Patent Application Publication Numbers 2015/0087659 and 2013/0303552.
- the term includes the compounds PBHBV- le salts thereof:
- Certain aspects of the invention are directed to the use of anti-HBsAg antibodies, e.g., mAbs. Certain aspects of the invention are directed to the use of hepatitis B immune globulin (HBIG). III. Agents that Improve Immune Response
- Category III treatments are directed to the use of agents that improve the immune response against viral infection.
- at least one 'immune enhancer' agent is used in combination with at least one 'immune stimulant agent' .
- Such a combination can be used in further combination with at least one agent that controls viral replication and/or at least one agent that reduces the viral antigens.
- Certain aspects of the invention are directed to the use of agents that act to improve an immune response by reducing or eliminating immune exhaustion, e.g., by using checkpoint inhibitors, thereby enhancing the immune response.
- an immune enhancer is a PD-L1 inhibitor.
- PD-L1 inhibitors are a group of agents that act to inhibit the association of the programmed death-ligand 1 (PD-L1) with its receptor, programmed cell death protein 1 (PD-1).
- Immune enhancers include the following:
- anti-PD-1 mAbs e.g., Nivolumab, Pembrolizumab;
- anti-PD-Ll mAbs e.g., Atezolizumab, Avelumab
- anti-CTLA4 mAbs e.g., Ipilimumab
- anti-VISTA mAbs e.g., JNJ-61610588
- anti-LAG3 mAbs e.g., BMS-986016
- anti-TEVB mAbs e.g., TSR-022
- peptidomimetics e.g., AU P-12
- immune stimulant includes compounds that are capable of modulating an immune response (e.g., stimulating an innate and/or adaptive immune response (e.g., an adjuvant)).
- immune stimulant includes polyinosinic:polycytidylic acid (poly I:C) and interferons.
- immune stimulant includes agonists of stimulator of IFN genes (STING) and interleukins.
- the term also includes HBsAg release inhibitors, TLR-7 agonists (GS-9620, RG- 7795), T-cell and/or B-cell stimulators (GS-4774, OX-40 agonists (BMS 986178), anti-GITR agonists (BMS-986156)), RIG-1 inhibitors (SB-9200), and SMAC-mimetics (Birinapant).
- the term also includes the following:
- anti-HBV vaccines Engerix-B, RECOMBIVAX HB, GS-4744, Heplisav-B
- interferons Pegylated IFN-a2a, Peglyated IFN-a2b, IFN-a, IFN- ⁇
- IFN- ⁇ interferons
- STING agonists cGAMP, cGAMP bisphosphorothioate, ADU SI 00, and other small molecule compounds
- TLR9 agonists (CYT-009, CpG dinucleotides);
- TLR3 agonists (Ampligen/poly LC12U);
- IL-2 aldesleukin
- Hepatitis B virus refers to a virus species of the genus Orthohepadnavirus, which is a part of the Hepadnaviridae family of viruses, and that is capable of causing liver inflammation in humans.
- Hepatitis D virus refers to a virus species of the genus
- Deltaviridae which is capable of causing liver inflammation in humans.
- treatment refers to clinical intervention in an attempt to alter the typical disease course of the individual being treated. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- antibodies of the invention are used to delay development of a disease or to slow the progression of a disease.
- small -interfering RNA or "siRNA” as used herein refers to double stranded
- RNA i.e., duplex RNA
- the siRNA may have substantial or complete identity to the target gene or sequence, or may comprise a region of mismatch (i.e., a mismatch motif).
- the siRNAs may be about 19-25 (duplex) nucleotides in length, and is preferably about 20-24, 21-22, or 21-23 (duplex) nucleotides in length.
- siRNA duplexes may comprise 3' overhangs of about 1 to about 4 nucleotides or about 2 to about 3 nucleotides and 5' phosphate termini.
- Examples of siRNA include, without limitation, a double-stranded polynucleotide molecule assembled from two separate stranded molecules, wherein one strand is the sense strand and the other is the complementary antisense strand.
- siRNA are chemically synthesized.
- siRNA can also be generated by cleavage of longer dsRNA (e.g., dsRNA greater than about 25 nucleotides in length) with the E. coli RNase III or Dicer. These enzymes process the dsRNA into biologically active siRNA (see, e.g., Yang et al, Proc. Natl. Acad. Sci. USA, 99:9942-9947 (2002); Calegari et al, Proc. Natl. Acad. Sci.
- dsRNA are at least 50 nucleotides to about 100, 200, 300, 400, or 500 nucleotides in length.
- a dsRNA may be as long as 1000, 1500, 2000, 5000 nucleotides in length, or longer.
- the dsRNA can encode for an entire gene transcript or a partial gene transcript.
- siRNA may be encoded by a plasmid (e.g., transcribed as sequences that automatically fold into duplexes with hairpin loops).
- inhibiting expression of a target gene refers to the ability of a siRNA to silence, reduce, or inhibit expression of a target gene (e.g., a gene within the HBV genome).
- a test sample e.g., a biological sample from an organism of interest expressing the target gene or a sample of cells in culture expressing the target gene
- a siRNA that silences, reduces, or inhibits expression of the target gene.
- Expression of the target gene in the test sample is compared to expression of the target gene in a control sample (e.g., a biological sample from an organism of interest expressing the target gene or a sample of cells in culture expressing the target gene) that is not contacted with the siRNA.
- Control samples e.g., samples expressing the target gene
- silencing, inhibition, or reduction of expression of a target gene is achieved when the value of the test sample relative to the control sample (e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.) is about 100%, 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91%, 90%, 89%, 88%, 87%, 86%, 85%, 84%, 83%, 82%, 81%, 80%, 79%, 78%, 77%, 76%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, 5%, or 0%.
- the control sample e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.
- Suitable assays include, without limitation, examination of protein or mRNA levels using techniques known to those of skill in the art, such as, e.g., dot blots, Northern blots, in situ hybridization, ELISA, immunoprecipitation, enzyme function, as well as phenotypic assays known to those of skill in the art.
- An "effective amount” or “therapeutically effective amount” of a therapeutic nucleic acid such as an siRNA is an amount sufficient to produce the desired effect, e.g., an inhibition of expression of a target sequence in comparison to the normal expression level detected in the absence of a siRNA.
- inhibition of expression of a target gene or target sequence is achieved when the value obtained with a siRNA relative to the control (e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.) is about 100%, 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91%, 90%, 89%, 88%, 87%, 86%, 85%, 84%, 83%, 82%, 81%, 80%, 79%, 78%, 77%, 76%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%), 25%), 20%), 15%), 10%, 5%, or 0%.
- a siRNA relative to the control e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.
- Suitable assays for measuring the expression of a target gene or target sequence include, but are not limited to, examination of protein or mRNA levels using techniques known to those of skill in the art, such as, e.g., dot blots, Northern blots, in situ hybridization, ELISA, immunoprecipitation, enzyme function, as well as phenotypic assays known to those of skill in the art.
- nucleic acid refers to a polymer containing at least two nucleotides (i.e., deoxyribonucleotides or ribonucleotides) in either single- or double-stranded form and includes DNA and RNA.
- nucleotides contain a sugar deoxyribose (DNA) or ribose (RNA), a base, and a phosphate group. Nucleotides are linked together through the phosphate groups.
- Bases include purines and pyrimidines, which further include natural compounds adenine, thymine, guanine, cytosine, uracil, inosine, and natural analogs, and synthetic derivatives of purines and pyrimidines, which include, but are not limited to, modifications which place new reactive groups such as, but not limited to, amines, alcohols, thiols,
- Nucleic acids include nucleic acids containing known nucleotide analogs or modified backbone residues or linkages, which are synthetic, naturally occurring, and non-naturally occurring, and which have similar binding properties as the reference nucleic acid. Examples of such analogs and/or modified residues include, without limitation,
- nucleic acids can include one or more UNA moieties.
- nucleic acid includes any oligonucleotide or polynucleotide, with fragments containing up to 60 nucleotides generally termed oligonucleotides, and longer fragments termed polynucleotides.
- a deoxyribooligonucleotide consists of a 5-carbon sugar called deoxyribose joined covalently to phosphate at the 5' and 3' carbons of this sugar to form an alternating, unbranched polymer.
- DNA may be in the form of, e.g., antisense molecules, plasmid DNA, pre- condensed DNA, a PCR product, vectors, expression cassettes, chimeric sequences,
- RNA may be in the form, for example, of small interfering RNA (siRNA), Dicer- substrate dsRNA, small hairpin RNA (shRNA), asymmetrical interfering RNA (aiRNA), microRNA (miRNA), mRNA, tRNA, rRNA, tRNA, viral RNA (vRNA), and combinations thereof.
- polynucleotide and oligonucleotide refer to a polymer or oligomer of nucleotide or nucleoside monomers consisting of naturally-occurring bases, sugars and intersugar (backbone) linkages.
- polynucleotide and oligonucleotide also include polymers or oligomers comprising non-naturally occurring monomers, or portions thereof, which function similarly.
- modified or substituted oligonucleotides are often preferred over native forms because of properties such as, for example, enhanced cellular uptake, reduced immunogenicity, and increased stability in the presence of nucleases.
- degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res., 19:5081 (1991); Ohtsuka et al., J. Biol. Chem., 260:2605-2608 (1985); Rossolini et al., Mol. Cell. Probes, 8:91-98 (1994)).
- An “isolated” or “purified” DNA molecule or RNA molecule is a DNA molecule or
- RNA molecule that exists apart from its native environment.
- An isolated DNA molecule or RNA molecule may exist in a purified form or may exist in a non-native environment such as, for example, a transgenic host cell.
- an "isolated" or “purified” nucleic acid molecule or biologically active portion thereof is substantially free of other cellular material, or culture medium when produced by recombinant techniques, or substantially free of chemical precursors or other chemicals when chemically synthesized.
- an "isolated" nucleic acid is free of sequences that naturally flank the nucleic acid (i.e., sequences located at the 5' and 3' ends of the nucleic acid) in the genomic DNA of the organism from which the nucleic acid is derived.
- the isolated nucleic acid molecule can contain less than about 5 kb, 4 kb, 3 kb, 2 kb, 1 kb, 0.5 kb, or 0.1 kb of nucleotide sequences that naturally flank the nucleic acid molecule in genomic DNA of the cell from which the nucleic acid is derived.
- gene refers to a nucleic acid (e.g., DNA or RNA) sequence that comprises partial length or entire length coding sequences necessary for the production of a polypeptide or precursor polypeptide.
- Gene product refers to a product of a gene such as an RNA transcript or a polypeptide.
- unlocked nucleobase analogue refers to an acyclic nucleobase in which the C2' and C3' atoms of the ribose ring are not covalently linked.
- unlocked nucleobase analogue includes nucleobase analogues having the following structure identified as Structure A:
- Base is any natural or unnatural base such as, for example, adenine (A), cytosine (C), guanine (G) and thymine (T).
- UNA include the molecules identified as acyclic 2' -3 '-seco-nucleotide monomers in U.S. patent serial number 8,314,227.
- lipid refers to a group of organic compounds that include, but are not limited to, esters of fatty acids and are characterized by being insoluble in water, but soluble in many organic solvents. They are usually divided into at least three classes: (1) “simple lipids,” which include fats and oils as well as waxes; (2) “compound lipids,” which include phospholipids and glycolipids; and (3) “derived lipids” such as steroids.
- lipid particle includes a lipid formulation that can be used to deliver a therapeutic nucleic acid (e.g., siRNA) to a target site of interest (e.g., cell, tissue, organ, and the like).
- a therapeutic nucleic acid e.g., siRNA
- the lipid particle is typically formed from a cationic lipid, a non-cationic lipid, and optionally a conjugated lipid that prevents aggregation of the particle.
- a lipid particle that includes a nucleic acid molecule e.g., siRNA molecule
- the nucleic acid is fully encapsulated within the lipid particle, thereby protecting the nucleic acid from enzymatic degradation.
- nucleic acid-lipid particles are extremely useful for systemic applications, as they can exhibit extended circulation lifetimes following intravenous (i.v.) injection, they can accumulate at distal sites (e.g., sites physically separated from the
- the nucleic acid may be complexed with a condensing agent and encapsulated within a lipid particle as set forth in PCT Publication No. WO 00/03683, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- the lipid particles typically have a mean diameter of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 1 10 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, or about 30 nm, 35 nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125
- nucleic acids when present in the lipid particles, are resistant in aqueous solution to degradation with a nuclease.
- Nucleic acid-lipid particles and their method of preparation are disclosed in, e.g., U.S. Patent Publication Nos. 20040142025 and 20070042031, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
- lipid encapsulated can refer to a lipid particle that provides a therapeutic nucleic acid such as a siRNA, with full encapsulation, partial encapsulation, or both.
- the nucleic acid e.g., siRNA
- the nucleic acid is fully encapsulated in the lipid particle (e.g., to form a nucleic acid-lipid particle).
- lipid conjugate refers to a conjugated lipid that inhibits aggregation of lipid particles.
- lipid conjugates include, but are not limited to, PEG-lipid conjugates such as, e.g., PEG coupled to dialkyloxypropyls (e.g., PEG-DAA conjugates), PEG coupled to diacylglycerols (e.g., PEG-DAG conjugates), PEG coupled to cholesterol, PEG coupled to phosphatidylethanolamines, and PEG conjugated to ceramides (see, e.g., U.S. Patent No.
- cationic PEG lipids cationic PEG lipids, polyoxazoline (POZ)-lipid conjugates (e.g., POZ-DAA conjugates), polyamide oligomers (e.g., ATTA-lipid conjugates), and mixtures thereof.
- POZ polyoxazoline
- DAA polyoxazoline conjugates
- polyamide oligomers e.g., ATTA-lipid conjugates
- PEG or POZ can be conjugated directly to the lipid or may be linked to the lipid via a linker moiety.
- Any linker moiety suitable for coupling the PEG or the POZ to a lipid can be used including, e.g., non-ester containing linker moieties and ester-containing linker moieties.
- non-ester containing linker moieties such as amides or carbamates, are used.
- amphipathic lipid refers, in part, to any suitable material wherein the hydrophobic portion of the lipid material orients into a hydrophobic phase, while the hydrophilic portion orients toward the aqueous phase.
- Hydrophilic characteristics derive from the presence of polar or charged groups such as carbohydrates, phosphate, carboxylic, sulfato, amino, sulfhydryl, nitro, hydroxyl, and other like groups. Hydrophobicity can be conferred by the inclusion of apolar groups that include, but are not limited to, long-chain saturated and unsaturated aliphatic hydrocarbon groups and such groups substituted by one or more aromatic, cycloaliphatic, or heterocyclic group(s). Examples of amphipathic compounds include, but are not limited to, phospholipids, aminolipids, and sphingolipids.
- phospholipids include, but are not limited to,
- phosphatidylcholine phosphatidyl ethanolamine, phosphatidylserine, phosphatidylinositol, phosphatidic acid, palmitoyloleoyl phosphatidylcholine, lysophosphatidylcholine, lysophosphatidylethanolamine, dipalmitoylphosphatidylcholine, dioleoylphosphatidylcholine, distearoylphosphatidylcholine, and dilinoleoylphosphatidylcholine.
- amphipathic lipids Other compounds lacking in phosphorus, such as sphingolipid, glycosphingolipid families, diacylglycerols, and ⁇ - acyloxyacids, are also within the group designated as amphipathic lipids. Additionally, the amphipathic lipids described above can be mixed with other lipids including triglycerides and sterols.
- neutral lipid refers to any of a number of lipid species that exist either in an uncharged or neutral zwitterionic form at a selected pH.
- lipids include, for example, diacylphosphatidyl choline, diacylphosphatidylethanolamine, ceramide, sphingomyelin, cephalin, cholesterol, cerebrosides, and diacylglycerols.
- non-cationic lipid refers to any amphipathic lipid as well as any other neutral lipid or anionic lipid.
- anionic lipid refers to any lipid that is negatively charged at physiological pH. These lipids include, but are not limited to, phosphatidylglycerols, cardiolipins,
- diacylphosphatidylserines diacylphosphatidic acids, N-dodecanoyl phosphatidylethanolamines, N-succinyl phosphatidylethanolamines, N-glutarylphosphatidylethanolamines,
- lysylphosphatidylglycerols palmitoyloleyolphosphatidylglycerol (POPG), and other anionic modifying groups joined to neutral lipids.
- POPG palmitoyloleyolphosphatidylglycerol
- hydrophobic lipid refers to compounds having apolar groups that include, but are not limited to, long-chain saturated and unsaturated aliphatic hydrocarbon groups and such groups optionally substituted by one or more aromatic, cycloaliphatic, or heterocyclic group(s). Suitable examples include, but are not limited to, diacylglycerol, dialkylglycerol, N- N-dialkylamino, l,2-diacyloxy-3-aminopropane, and l,2-dialkyl-3-aminopropane.
- cationic lipid and “amino lipid” are used interchangeably herein to include those lipids and salts thereof having one, two, three, or more fatty acid or fatty alkyl chains and a pH-titratable amino head group (e.g., an alkylamino or dialkylamino head group).
- the cationic lipid is typically protonated (i.e., positively charged) at a pH below the pK a of the cationic lipid and is substantially neutral at a pH above the pK a .
- the cationic lipids may also be termed titratable cationic lipids.
- the cationic lipids comprise: a protonatable tertiary amine (e.g., pH-titratable) head group; C 18 alkyl chains, wherein each alkyl chain independently has 0 to 3 (e.g., 0, 1, 2, or 3) double bonds; and ether, ester, or ketal linkages between the head group and alkyl chains.
- a protonatable tertiary amine e.g., pH-titratable
- C 18 alkyl chains wherein each alkyl chain independently has 0 to 3 (e.g., 0, 1, 2, or 3) double bonds
- ether, ester, or ketal linkages between the head group and alkyl chains e.g., 1, 2, or 3
- Such cationic lipids include, but are not limited to, DSDMA, DODMA, DLinDMA, DLenDMA, ⁇ -DLenDMA, DLin-K-DMA, DLin-K-C2-DMA (also known as DLin-C2K-DMA, XTC2, and C2K), DLin-K-C3 -DMA, DLin-K-C4-DMA, DLen-C2K-DMA, y-DLen-C2K-DMA, DLin-M-C2-DMA (also known as MC2), and DLin-M- C3-DMA (also known as MC3).
- a compound as a pharmaceutically acceptable acid or base salt may be appropriate.
- pharmaceutically acceptable salts are organic acid addition salts formed with acids which form a physiologically acceptable anion, for example, tosylate, methanesulfonate, acetate, citrate, malonate, tartrate, succinate, benzoate, ascorbate, a- ketoglutarate, and a-glycerophosphate.
- Suitable inorganic salts may also be formed, including hydrochloride, sulfate, nitrate, bicarbonate, and carbonate salts.
- salts may be obtained using standard procedures well known in the art, for example by reacting a sufficiently basic compound such as an amine with a suitable acid affording a physiologically acceptable anion.
- a sufficiently basic compound such as an amine
- a suitable acid affording a physiologically acceptable anion.
- Alkali metal (for example, sodium, potassium or lithium) or alkaline earth metal (for example calcium) salts of carboxylic acids can also be made.
- salts includes any anionic and cationic complex, such as the complex formed between a cationic lipid and one or more anions.
- anions include inorganic and organic anions, e.g., hydride, fluoride, chloride, bromide, iodide, oxalate (e.g., hemioxalate), phosphate, phosphonate, hydrogen phosphate, dihydrogen phosphate, oxide, carbonate, bicarbonate, nitrate, nitrite, nitride, bisulfite, sulfide, sulfite, bisulfate, sulfate, thiosulfate, hydrogen sulfate, borate, formate, acetate, benzoate, citrate, tartrate, lactate, acrylate, polyacrylate, fumarate, maleate, itaconate, glycolate, gluconate, malate, mandelate, tiglate, ascorbate,
- alkyl includes a straight chain or branched, noncyclic or cyclic, saturated aliphatic hydrocarbon containing from 1 to 24 carbon atoms.
- Representative saturated straight chain alkyls include, but are not limited to, methyl, ethyl, ⁇ -propyl, «-butyl, «-pentyl, «-hexyl, and the like, while saturated branched alkyls include, without limitation, isopropyl, sec-butyl, isobutyl, tert-butyl, isopentyl, and the like.
- saturated cyclic alkyls include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and the like, while unsaturated cyclic alkyls include, without limitation, cyclopentenyl, cyclohexenyl, and the like.
- alkenyl includes an alkyl, as defined above, containing at least one double bond between adjacent carbon atoms. Alkenyls include both cis and trans isomers.
- Representative straight chain and branched alkenyls include, but are not limited to, ethyl enyl, propylenyl, 1-butenyl, 2-butenyl, isobutylenyl, 1-pentenyl, 2-pentenyl, 3 -methyl- 1-butenyl, 2- methyl-2-butenyl, 2,3 -dimethyl -2-butenyl, and the like.
- alkynyl includes any alkyl or alkenyl, as defined above, which additionally contains at least one triple bond between adjacent carbons.
- Representative straight chain and branched alkynyls include, without limitation, acetyl enyl, propynyl, 1-butynyl, 2-butynyl, 1- pentynyl, 2-pentynyl, 3 -methyl- 1 butynyl, and the like.
- acyl includes any alkyl, alkenyl, or alkynyl wherein the carbon at the point of attachment is substituted with an oxo group, as defined below.
- heterocycle includes a 5- to 7-membered monocyclic, or 7- to 10- membered bicyclic, heterocyclic ring which is either saturated, unsaturated, or aromatic, and which contains from 1 or 2 heteroatoms independently selected from nitrogen, oxygen and sulfur, and wherein the nitrogen and sulfur heteroatoms may be optionally oxidized, and the nitrogen heteroatom may be optionally quaternized, including bicyclic rings in which any of the above heterocycles are fused to a benzene ring.
- the heterocycle may be attached via any heteroatom or carbon atom.
- Heterocycles include, but are not limited to, heteroaryls as defined below, as well as morpholinyl, pyrrolidinonyl, pyrrolidinyl, piperidinyl, piperizynyl,
- hydantoinyl valerolactamyl, oxiranyl, oxetanyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydropyridinyl, tetrahydroprimidinyl, tetrahydrothiophenyl, tetrahydrothiopyranyl, tetrahydropyrimidinyl, tetrahydrothiophenyl, tetrahydrothiopyranyl, and the like.
- halogen includes fluoro, chloro, bromo, and iodo.
- fuusogenic refers to the ability of a lipid particle to fuse with the membranes of a cell.
- the membranes can be either the plasma membrane or membranes surrounding organelles, e.g., endosome, nucleus, etc.
- aqueous solution refers to a composition comprising in whole, or in part, water.
- organic lipid solution refers to a composition comprising in whole, or in part, an organic solvent having a lipid.
- electrophilic core when used to describe a lipid particle, refers to the dark appearance of the interior portion of a lipid particle when visualized using cryo
- lipid particles have an electron dense core and lack a lipid bilayer structure.
- Some lipid particles have an elctron dense core, lack a lipid bilayer structure, and have an inverse Hexagonal or Cubic phase structure. While not wishing to be bound by theory, it is thought that the non-bilayer lipid packing provides a 3- dimensional network of lipid cylinders with water and nucleic acid on the inside, i.e., essentially a lipid droplet interpenetrated with aqueous channels containing the nucleic acid.
- Distal site refers to a physically separated site, which is not limited to an adjacent capillary bed, but includes sites broadly distributed throughout an organism.
- “Serum-stable” in relation to nucleic acid-lipid particles means that the particle is not significantly degraded after exposure to a serum or nuclease assay that would significantly degrade free DNA or RNA.
- Suitable assays include, for example, a standard serum assay, a DNAse assay, or an RNAse assay.
- Systemic delivery refers to delivery of lipid particles that leads to a broad biodistribution of an active agent such as a siRNA within an organism. Some techniques of administration can lead to the systemic delivery of certain agents, but not others. Systemic delivery means that a useful, preferably therapeutic, amount of an agent is exposed to most parts of the body. To obtain broad biodistribution generally requires a blood lifetime such that the agent is not rapidly degraded or cleared (such as by first pass organs (liver, lung, etc.) or by rapid, nonspecific cell binding) before reaching a disease site distal to the site of administration.
- Systemic delivery of lipid particles can be by any means known in the art including, for example, intravenous, subcutaneous, and intraperitoneal. In a preferred embodiment, systemic delivery of lipid particles is by intravenous delivery.
- “Local delivery,” as used herein, refers to delivery of an active agent such as a siRNA directly to a target site within an organism.
- an agent can be locally delivered by direct injection into a disease site, other target site, or a target organ such as the liver, heart, pancreas, kidney, and the like.
- virus particle load refers to a measure of the number of virus particles (e.g., HBV and/or HDV) present in a bodily fluid, such as blood.
- particle load may be expressed as the number of virus particles per milliliter of, e.g., blood.
- Particle load testing may be performed using nucleic acid amplification based tests, as well as non-nucleic acid-based tests (see, e.g., Puren et al., The Journal of Infectious Diseases, 201 :S27- 36 (2010)).
- oligonucleotides such as the sense and antisense RNA strands set forth in the Example specifically hybridize to or are complementary to a target polynucleotide sequence.
- the terms "specifically hybridizable” and “complementary” as used herein indicate a sufficient degree of complementarity such that stable and specific binding occurs between the DNA or RNA target and the oligonucleotide. It is understood that an oligonucleotide need not be 100% complementary to its target nucleic acid sequence to be specifically hybridizable. In preferred embodiments, an oligonucleotide is specifically hybridizable when binding of the
- oligonucleotide to the target sequence interferes with the normal function of the target sequence to cause a loss of utility or expression therefrom, and there is a sufficient degree of
- the oligonucleotide may include 1, 2, 3, or more base substitutions as compared to the region of a gene or mRNA sequence that it is targeting or to which it specifically hybridizes.
- siRNA can be provided in several forms including, e.g., as one or more isolated small- interfering RNA (siRNA) duplexes, as longer double-stranded RNA (dsRNA), or as siRNA or dsRNA transcribed from a transcriptional cassette in a DNA plasmid.
- siRNA may be produced enzymatically or by partial/total organic synthesis, and modified ribonucleotides can be introduced by in vitro enzymatic or organic synthesis.
- each strand is prepared chemically. Methods of synthesizing RNA molecules are known in the art, e.g., the chemical synthesis methods as described in Verma and Eckstein (1998) or as described herein.
- RNA, synthesizing RNA, hybridizing nucleic acids, making and screening cDNA libraries, and performing PCR are well known in the art (see, e.g., Gubler and Hoffman, Gene, 25:263-269 (1983); Sambrook et al, supra; Ausubel et al, supra), as are PCR methods (see, U.S. Patent Nos. 4,683, 195 and 4,683,202; PCR Protocols: A Guide toMethods and Applications (Innis et al., eds, 1990)).
- Expression libraries are also well known to those of skill in the art. Additional basic texts disclosing the general methods include Sambrook et al., Molecular Cloning, A Laboratory Manual (2nd ed. 1989); Kriegler, Gene Transfer and
- siRNA are chemically synthesized.
- the oligonucleotides that comprise the siRNA molecules can be synthesized using any of a variety of techniques known in the art, such as those described in Usman et al, J. Am. Chem. Soc, 109:7845 (1987); Scaringe et al, Nucl. Acids Res., 18:5433 (1990); Wincott et al, Nucl. Acids Res., 23 :2677-2684 (1995); and Wincott et al., Methods Mol. Bio., 74:59 (1997).
- oligonucleotides makes use of common nucleic acid protecting and coupling groups, such as dimethoxytrityl at the 5 '-end and phosphoramidites at the 3 '-end.
- small scale syntheses can be conducted on an Applied Biosystems synthesizer using a 0.2 ⁇ scale protocol.
- syntheses at the 0.2 ⁇ scale can be performed on a 96-well plate synthesizer from Protogene (Palo Alto, CA).
- Protogene Protogene
- Suitable reagents for oligonucleotide synthesis, methods for RNA deprotection, and methods for RNA purification are known to those of skill in the art.
- siRNA molecules can be assembled from two distinct oligonucleotides, wherein one oligonucleotide comprises the sense strand and the other comprises the antisense strand of the siRNA.
- each strand can be synthesized separately and joined together by hybridization or ligation following synthesis and/or deprotection.
- Lipid particles may be used to deliver siRNA molecules described herein (e.g., a combination comprising the three siRNA molecules described in Example 1).
- Lipid particles may comprise one or more siRNA ⁇ e.g., the three siRNA molecules described in Example 1), a cationic lipid, a non-cationic lipid, and a conjugated lipid that inhibits aggregation of particles.
- the siRNA molecule(s) is fully encapsulated within the lipid portion of the lipid particle such that the siRNA molecule(s) in the lipid particle is resistant in aqueous solution to nuclease degradation.
- the lipid particles described herein are substantially non-toxic to humans.
- the lipid particles typically have a mean diameter of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 110 nm, or from about 70 to about 90 nm. In certain embodiments, the lipid particles have a median diameter of from about 30 nm to about 150 nm.
- the lipid particles also typically have a lipid:nucleic acid ratio (e.g., a lipid: siRNA ratio) (mass/mass ratio) of from about 1 : 1 to about 100: 1, from about 1 : 1 to about 50: 1, from about 2: 1 to about 25 : 1, from about 3 : 1 to about 20: 1, from about 5 : 1 to about 15 : 1, or from about 5 : 1 to about 10: 1.
- the nucleic acid-lipid particle has a lipid: siRNA mass ratio of from about 5 : 1 to about 15 : 1.
- the lipid particles include serum-stable nucleic acid-lipid particles which comprise one or more siRNA molecules (e.g., a combination comprising the three siRNA molecules described in Example 1), a cationic lipid (e.g., one or more cationic lipids of Formula I-III or salts thereof as set forth herein), a non-cationic lipid (e.g., mixtures of one or more phospholipids and cholesterol), and a conjugated lipid that inhibits aggregation of the particles (e.g., one or more PEG-lipid conjugates).
- siRNA molecules e.g., a combination comprising the three siRNA molecules described in Example 1
- a cationic lipid e.g., one or more cationic lipids of Formula I-III or salts thereof as set forth herein
- a non-cationic lipid e.g., mixtures of one or more phospholipids and cholesterol
- a conjugated lipid that inhibits aggregation of the particles e
- the lipid particle may comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more siRNA molecules (e.g., a combination comprising the three siRNA molecules described in Example 1) that target one or more of the genes described herein.
- siRNA molecules e.g., a combination comprising the three siRNA molecules described in Example 1
- Nucleic acid-lipid particles and their method of preparation are described in, e.g., U.S. Patent Nos. 5,753,613; 5,785,992; 5,705,385; 5,976,567; 5,981,501 ; 6, 1 10,745; and 6,320,017; and PCT Publication No. WO 96/40964, the disclosures of which are each herein incorporated by reference in their entirety for all purposes.
- the one or more siRNA molecules e.g., a
- combination comprising the three siRNA molecules described in Example 1) may be fully encapsulated within the lipid portion of the particle, thereby protecting the siRNA from nuclease degradation.
- the siRNA in the nucleic acid-lipid particle is not substantially degraded after exposure of the particle to a nuclease at 37°C for at least about 20, 30, 45, or 60 minutes.
- the siRNA in the nucleic acid-lipid particle is not substantially degraded after incubation of the particle in serum at 37°C for at least about 30, 45, or 60 minutes or at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32,
- the siRNA is complexed with the lipid portion of the particle.
- the nucleic acid-lipid particle is that the nucleic acid-lipid particle
- compositions are substantially non-toxic to humans.
- siRNA e.g., a combination
- nucleic acid-lipid particle comprising the three siRNA molecules described in Example 1) in the nucleic acid-lipid particle is not significantly degraded after exposure to serum or a nuclease assay that would significantly degrade free DNA or RNA.
- a fully encapsulated system preferably less than about 25% of the siRNA in the particle is degraded in a treatment that would normally degrade 100% of free siRNA, more preferably less than about 10%, and most preferably less than about 5% of the siRNA in the particle is degraded.
- “Fully encapsulated” also indicates that the nucleic acid-lipid particles are serum-stable, that is, that they do not rapidly decompose into their component parts upon in vivo administration.
- full encapsulation may be determined by performing a membrane-impermeable fluorescent dye exclusion assay, which uses a dye that has enhanced fluorescence when associated with nucleic acid.
- fluorescent dye exclusion assay which uses a dye that has enhanced fluorescence when associated with nucleic acid.
- Specific dyes such as OliGreen ® and
- RiboGreen ® (Invitrogen Corp.; Carlsbad, CA) are available for the quantitative determination of plasmid DNA, single-stranded deoxyribonucleotides, and/or single- or double-stranded ribonucleotides. Encapsulation is determined by adding the dye to a liposomal formulation, measuring the resulting fluorescence, and comparing it to the fluorescence observed upon addition of a small amount of nonionic detergent. Detergent-mediated disruption of the liposomal bilayer releases the encapsulated nucleic acid, allowing it to interact with the membrane-impermeable dye.
- the nucleic acid-lipid particle composition comprises a siRNA molecule that is fully encapsulated within the lipid portion of the particles, such that from about 30% to about 100%, from about 40% to about 100%, from about 50% to about 100%, from about 60% to about 100%, from about 70% to about 100%, from about 80% to about 100%, from about 90% to about 100%, from about 30% to about 95%, from about 40% to about 95%, from about 50% to about 95%, from about 60%> to about 95%, from about 70% to about 95%, from about 80% to about 95%, from about 85% to about 95%, from about 90% to about 95%, from about 30% to about 90%, from about 40% to about 90%, from about 50% to about 90%, from about 60% to about 90%, from about 70% to about 90%, from about 80% to about 90%, or at least about 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%
- the nucleic acid-lipid particle composition comprises siRNA that is fully encapsulated within the lipid portion of the particles, such that from about 30% to about 100%, from about 40% to about 100%, from about 50% to about 100%, from about 60% to about 100%, from about 70% to about 100%, from about 80% to about 100%, from about 90% to about 100%, from about 30% to about 95%, from about 40% to about 95%, from about 50% to about 95%), from about 60% to about 95%, from about 70% to about 95%, from about 80% to about 95%, from about 85% to about 95%, from about 90% to about 95%, from about 30% to about 90%, from about 40% to about 90%, from about 50% to about 90%, from about 60% to about 90%, from about 70% to about 90%, from about 80% to about 90%, or at least about 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%., 96%., 97%., 9
- the proportions of the components can be varied and the delivery efficiency of a particular formulation can be measured using, e.g., an endosomal release parameter (ERP) assay.
- ERP endosomal release parameter
- cationic lipids or salts thereof may be used in the lipid particles either alone or in combination with one or more other cationic lipid species or non-cationic lipid species.
- the cationic lipids include the (R) and/or (S) enantiomers thereof.
- the cationic lipid is a dialkyl lipid.
- dialkyl lipids may include lipids that comprise two saturated or unsaturated alkyl chains, wherein each of the alkyl chains may be substituted or unsubstituted.
- each of the two alkyl chains comprise at least, e.g., 8 carbon atoms, 10 carbon atoms, 12 carbon atoms, 14 carbon atoms, 16 carbon atoms, 18 carbon atoms, 20 carbon atoms, 22 carbon atoms or 24 carbon atoms.
- the cationic lipid is a trialkyl lipid.
- trialkyl lipids may include lipids that comprise three saturated or unsaturated alkyl chains, wherein each of the alkyl chains may be substituted or unsubstituted.
- each of the three alkyl chains comprise at least, e.g., 8 carbon atoms, 10 carbon atoms, 12 carbon atoms, 14 carbon atoms, 16 carbon atoms, 18 carbon atoms, 20 carbon atoms, 22 carbon atoms or 24 carbon atoms.
- cationic li ids of Formula I having the following structure are useful:
- R 1 and R 2 are either the same or different and are independently hydrogen (H) or an optionally substituted Ci-C 6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R 1 and R 2 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms selected from the group consisting of nitrogen (N), oxygen (O), and mixtures thereof;
- R 3 is either absent or is hydrogen (H) or a Ci-C 6 alkyl to provide a quaternary amine
- R 4 and R 5 are either the same or different and are independently an optionally substituted C10-C24 alkyl, C10-C24 alkenyl, C10-C24 alkynyl, or C10-C24 acyl, wherein at least one of R 4 and R 5 comprises at least two sites of unsaturation;
- n 0, 1, 2, 3, or 4.
- R 1 and R 2 are independently an optionally substituted C1-C4 alkyl, C2-C4 alkenyl, or C2-C4 alkynyl. In one preferred embodiment, R 1 and R 2 are both methyl groups. In other preferred embodiments, n is 1 or 2. In other embodiments, R 3 is absent when the pH is above the pK a of the cationic lipid and R 3 is hydrogen when the pH is below the pK a of the cationic lipid such that the amino head group is protonated. In an alternative embodiment, R 3 is an optionally substituted C1-C4 alkyl to provide a quaternary amine. In further, R 3 is absent when the pH is above the pK a of the cationic lipid and R 3 is hydrogen when the pH is below the pK a of the cationic lipid such that the amino head group is protonated. In an alternative embodiment, R 3 is an optionally substituted C1-C4 alkyl to provide a quaternary amine. In
- R 4 and R 5 are independently an optionally substituted C12-C20 or C14-C22 alkyl, C12-C20 or C14-C22 alkenyl, C12-C20 or C14-C22 alkynyl, or C12-C20 or C14-C22 acyl, wherein at least one of R 4 and R 5 comprises at least two sites of unsaturation.
- R 4 and R 5 are independently selected from the group consisting of a dodecadienyl moiety, a tetradecadienyl moiety, a hexadecadienyl moiety, an octadecadienyl moiety, an icosadienyl moiety, a dodecatrienyl moiety, a tetradectrienyl moiety, a hexadecatrienyl moiety, an octadecatrienyl moiety, an icosatrienyl moiety, an arachidonyl moiety, and a docosahexaenoyl moiety, as well as acyl derivatives thereof (e.g., linoleoyl, linolenoyl, ⁇ -linolenoyl, etc.).
- acyl derivatives thereof e.g., linoleoyl, linolenoyl,
- one of R 4 and R 5 comprises a branched alkyl group (e.g., a phytanyl moiety) or an acyl derivative thereof (e.g., a phytanoyl moiety).
- the octadecadienyl moiety is a linoleyl moiety.
- the octadecatrienyl moiety is a linolenyl moiety or a ⁇ -linolenyl moiety.
- R 4 and R 5 are both linoleyl moieties, linolenyl moieties, or ⁇ -linolenyl moieties.
- the cationic lipid of Formula I is l,2-dilinoleyloxy-N,N-dimethylaminopropane (DLinDMA), l,2-dilinolenyloxy-N,N-dimethylaminopropane (DLenDMA), 1,2-dilinoleyloxy- (N,N-dimethyl)-butyl-4-amine (C2-DLinDMA), 1 ,2-dilinoleoyloxy-(N,N-dimethyl)-butyl-4- amine (C2-DLinDAP), or mixtures thereof.
- DLinDMA l,2-dilinoleyloxy-N,N-dimethylaminopropane
- DLenDMA 1,2-dilinoleyloxy- (N,N-dimethyl)-butyl-4-amine
- C2-DLinDAP 1,2-dilinoleoyloxy-(N,N-dimethyl)-butyl-4- amine
- the cationic lipid of Formula I forms a salt (preferably a crystalline salt) with one or more anions.
- the cationic lipid of Formula I is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
- cationic lipids such as DLinDMA and DLenDMA, as well as additional cationic lipids
- U.S. Patent Publication No. 20060083780 the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- the synthesis of cationic lipids such as C2-DLinDMA and C2-DLinDAP, as well as additional cationic lipids, is described in international patent application number WO201 1/000106 the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- cationic lipids of Formula II having the following structure (or salts thereof) are useful:
- R 1 and R 2 are either the same or different and are independently an optionally substituted C12-C24 alkyl, C12-C24 alkenyl, C12-C24 alkynyl, or C12-C24 acyl;
- R 3 and R 4 are either the same or different and are independently an optionally substituted Ci-C 6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R 3 and R 4 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms chosen from nitrogen and oxygen;
- R 5 is either absent or is hydrogen (H) or a Ci-C 6 alkyl to provide a quaternary amine;
- m, n, and p are either the same or different and are independently either 0, 1, or 2, with the proviso that m, n, and p are not simultaneously 0;
- q is 0, 1, 2, 3, or 4;
- Y and Z are either the same or different and
- the cationic lipid of Formula II is 2,2-dilinoleyl-4-(2- dimethylaminoethyl)-[l,3]-dioxolane (DLin-K-C2-DMA; "XTC2" or “C2K”), 2,2-dilinoleyl-4- (3-dimethylaminopropyl)-[l,3]-dioxolane (DLin-K-C3 -DMA; "C3K”), 2,2-dilinoleyl-4-(4- dimethylaminobutyl)-[l,3]-dioxolane (DLin-K-C4-DMA; "C4K”), 2,2-dilinoleyl-5- dimethylaminomethyl-[l,3]-dioxane (DLin-K6-DMA), 2,2-dilinoleyl-4-N-methylpepiazino- [l,3]-dioxolane (DLin-N-methylpe
- the cationic lipid of Formula II forms a salt (preferably a crystalline salt) with one or more anions.
- the cationic lipid of Formula II is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
- cationic lipids such as DLin-K-DMA, as well as additional cationic lipids, is described in PCT Publication No. WO 09/086558, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- cationic lipids such as DLin-K-C2-DMA, DLin-K-C3-DMA, DLin-K-C4-DMA, DLin-K6-DMA, DLin-K-MPZ, DO- K-DMA, DS-K-DMA, DLin-K-MA, DLin-K-TMA.Cl, DLin-K 2 -DMA, and D-Lin-K-N- methylpiperzine, as well as additional cationic lipids, is described in PCT Application No.
- cationic lipids of Formula III having the following structure are useful:
- R 1 and R 2 are either the same or different and are independently an optionally substituted Ci-C 6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R 1 and R 2 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms selected from the group consisting of nitrogen (N), oxygen (O), and mixtures thereof;
- R 3 is either absent or is hydrogen (H) or a Ci-C 6 alkyl to provide a quaternary amine;
- R 4 and R 5 are either absent or present and when present are either the same or different and are independently an optionally substituted C1-C10 alkyl or C2-C10 alkenyl; and n is 0, 1, 2, 3, or 4.
- R 1 and R 2 are independently an optionally substituted C1-C4 alkyl, C2-C4 alkenyl, or C2-C4 alkynyl.
- R 1 and R 2 are both methyl groups.
- R 4 and R 5 are both butyl groups.
- n is 1.
- R 3 is absent when the pH is above the pK a of the cationic lipid and R 3 is hydrogen when the pH is below the pK a of the cationic lipid such that the amino head group is protonated.
- R 3 is an optionally substituted C1-C4 alkyl to provide a quaternary amine.
- R 4 and R 5 are independently an optionally substituted C2-C6 or C2-C4 alkyl or C2-C6 or C2-C4 alkenyl.
- the cationic lipid of Formula III comprises ester linkages between the amino head group and one or both of the alkyl chains.
- the cationic lipid of Formula III forms a salt (preferably a crystalline salt) with one or more anions.
- the cationic lipid of Formula III is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
- each of the alkyl chains in Formula III contains cis double bonds at positions 6, 9, and 12 (i.e., cis,cis,cis-A 6 , 9 , u ), in an alternative embodiment, one, two, or three of these double bonds in one or both alkyl chains may be in the trans configuration.
- the cationic lipid of Formula III has the structure:
- MC3 DLin-M-C3 -DMA
- additional cationic lipids e.g., certain analogs of MC3
- cationic lipids or salts thereof which may be included in the lipid particles include, but are not limited to, cationic lipids such as those described in
- WO2011/000106 the disclosure of which is herein incorporated by reference in its entirety for all purposes, as well as cationic lipids such as N,N-dioleyl-N,N-dimethylammonium chloride (DODAC), l,2-dioleyloxy-N,N-dimethylaminopropane (DODMA), l,2-distearyloxy-N,N- dimethylaminopropane (DSDMA), N-(l -(2,3-dioleyloxy)propyl)-N,N,N-trimethylammonium chloride (DOTMA), N,N-distearyl-N,N-dimethylammonium bromide (DDAB), N-(l-(2,3- dioleoyloxy)propyl)-N,N,N-trimethylammonium chloride (DOTAP), 3 -( ⁇ -( ⁇ ', ⁇ '- dimethylaminoethane)-carbamoyl)chol
- dioctadecylamidoglycyl spermine DOGS
- 3-dimethylamino-2-(cholest-5-en-3-beta-oxybutan- 4-oxy)-l-(cis,cis-9, 12-octadecadienoxy)propane CLinDMA
- 2-[5'-(cholest-5-en-3-beta-oxy)- 3 ' -oxapentoxy)-3 -dimethy- 1 -(cis,cis-9 ' , 1 -2' -octadecadienoxy)propane CpLinDMA
- DMOBA N,N- dimethyl-3,4-dioleyloxybenzylamine
- DOcarbDAP l,2-N,N'-dilinoleylcarbamyl-3-dimethylaminopropane
- cationic lipids such as CLinDMA, as well as additional cationic lipids
- cationic lipids such as DLin-C-DAP, DLinDAC, DLinMA, DLinDAP, DLin-S-DMA, DLin-2-DMAP, DLinTMA.Cl, DLinTAP.Cl, DLinMPZ, DLinAP, DOAP, and DLin-EG-DMA, as well as additional cationic lipids, is described in PCT Publication No.
- WO 09/086558 the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- cationic lipids such as DO-C-DAP, DMDAP, DOTAP.Cl, DLin-M-C2-DMA, as well as additional cationic lipids, is described in PCT Application No. PCT/US2009/060251, entitled “Improved Amino Lipids and Methods for the Delivery of Nucleic Acids," filed October 9, 2009, the disclosure of which is incorporated herein by reference in its entirety for all purposes.
- the synthesis of a number of other cationic lipids and related analogs has been described in U.S. Patent Nos. 5,208,036;
- LIPOFECTAMINE ® including DOSPA and DOPE, available from Invitrogen
- TRANSFECTAM ® including DOGS, available from Promega Corp.
- the cationic lipid comprises from about 50 mol % to about 90 mol %, from about 50 mol % to about 85 mol %, from about 50 mol % to about 80 mol %, from about 50 mol % to about 75 mol %, from about 50 mol % to about 70 mol %, from about 50 mol % to about 65 mol %, from about 50 mol % to about 60 mol %, from about 55 mol % to about 65 mol %, or from about 55 mol % to about 70 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the cationic lipid comprises about 50 mol %, 51 mol %, 52 mol %, 53 mol %, 54 mol %, 55 mol %, 56 mol %, 57 mol %, 58 mol %, 59 mol %, 60 mol %, 61 mol %, 62 mol %, 63 mol %, 64 mol %, or 65 mol % (or any fraction thereof) of the total lipid present in the particle.
- the cationic lipid comprises from about 2 mol % to about 60 mol %, from about 5 mol % to about 50 mol %, from about 10 mol % to about 50 mol %, from about 20 mol % to about 50 mol %, from about 20 mol % to about 40 mol %, from about 30 mol % to about 40 mol %, or about 40 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the percentage of cationic lipid present in the lipid particles is a target amount, and that the actual amount of cationic lipid present in the formulation may vary, for example, by ⁇ 5 mol %.
- the target amount of cationic lipid is 57.1 mol %, but the actual amount of cationic lipid may be ⁇ 5 mol %, ⁇ 4 mol %, ⁇ 3 mol %, ⁇ 2 mol %, ⁇ 1 mol %, ⁇ 0.75 mol %, ⁇ 0.5 mol %, ⁇ 0.25 mol %, or ⁇ 0.1 mol % of that target amount, with the balance of the formulation being made up of other lipid components (adding up to 100 mol % of total lipids present in the particle; however, one skilled in the art will understand that the total mol % may deviate slightly from 100% due to rounding, for example, 99.9 mol % or 100.1 mol %
- cationic lipids useful for inclusion in lipid particles are shown below:
- the non-cationic lipids used in the lipid particles can be any of a variety of neutral uncharged, zwitterionic, or anionic lipids capable of producing a stable complex.
- Non-limiting examples of non-cationic lipids include phospholipids such as lecithin, phosphatidylethanolamine, lysolecithin, lysophosphatidylethanolamine, phosphatidylserine, phosphatidylinositol, sphingomyelin, egg sphingomyelin (ESM), cephalin, cardiolipin, phosphatidic acid, cerebrosides, dicetylphosphate, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC),
- phospholipids such as lecithin, phosphatidylethanolamine, lysolecithin, lysophosphatidylethanolamine, phosphatidylserine, phosphatidylinositol, sphingomyelin, egg sphingomyelin (
- DOPG dioleoylphosphatidylglycerol
- DPPG dipalmitoylphosphatidylglycerol
- dioleoylphosphatidylethanolamine DOPE
- palmitoyloleoyl-phosphatidylcholine POPC
- palmitoyloleoyl-phosphatidylethanolamine POPE
- palmitoyloleyol-phosphatidylglycerol POPG
- dioleoylphosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane-l-carboxylate DOPE-mal
- dipalmitoyl-phosphatidylethanolamine DPPE
- dimyristoyl- phosphatidylethanolamine DMPE
- distearoyl-phosphatidylethanolamine DSPE
- monomethyl-phosphatidylethanolamine dimethyl -phosphatidyl ethanolamine, dielaidoyl- phosphatidylethanolamine (DEPE), stearoyloleoyl-phosphatidylethanolamine (SOPE), lysophosphatidylcholine, dilinoleoylphosphatidylcholine, and mixtures thereof.
- DEPE dielaidoyl- phosphatidylethanolamine
- SOPE stearoyloleoyl-phosphatidylethanolamine
- lysophosphatidylcholine dilinoleoylphosphatidylcholine
- Other diacylphosphatidylcholine and diacylphosphatidylethanolamine phospholipids can also be used.
- acyl groups in these lipids are preferably acyl groups derived from fatty acids having C 10 - C24 carbon chains, e.g., lauroyl, myristoyl, palmitoyl, stearoyl, or oleoyl.
- non-cationic lipids include sterols such as cholesterol and derivatives thereof.
- cholesterol derivatives include polar analogues such as 5a-cholestanol, 5P-coprostanol, cholesteryl-(2'-hydroxy)-ethyl ether, cholesteryl-(4'- hydroxy)-butyl ether, and 6-ketocholestanol; non-polar analogues such as 5a-cholestane, cholestenone, 5a-cholestanone, 5P-cholestanone, and cholesteryl decanoate; and mixtures thereof.
- the cholesterol derivative is a polar analogue such as cholesteryl-(4'-hydroxy)-butyl ether.
- cholesteryl-(2'-hydroxy)-ethyl ether is described in PCT Publication No. WO 09/127060, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- the non-cationic lipid present in the lipid particles comprises or consists of a mixture of one or more phospholipids and cholesterol or a derivative thereof. In other embodiments, the non-cationic lipid present in the lipid particles comprises or consists of one or more phospholipids, e.g., a cholesterol -free lipid particle formulation. In yet other embodiments, the non-cationic lipid present in the lipid particles comprises or consists of cholesterol or a derivative thereof, e.g., a phospholipid-free lipid particle formulation.
- non-cationic lipids suitable for use include nonphosphorous containing lipids such as, e.g., stearylamine, dodecylamine, hexadecylamine, acetyl palmitate, glycerolricinoleate, hexadecyl stereate, isopropyl myristate, amphoteric acrylic polymers, triethanolamine-lauryl sulfate, alkyl-aryl sulfate poly ethyl oxylated fatty acid amides, dioctadecyl dimethyl ammonium bromide, ceramide, sphingomyelin, and the like.
- nonphosphorous containing lipids such as, e.g., stearylamine, dodecylamine, hexadecylamine, acetyl palmitate, glycerolricinoleate, hexadecyl stereate, isoprop
- the non-cationic lipid comprises from about 10 mol % to about 60 mol %, from about 20 mol % to about 55 mol %, from about 20 mol % to about 45 mol %, from about 20 mol % to about 40 mol %, from about 25 mol % to about 50 mol %, from about 25 mol % to about 45 mol %, from about 30 mol % to about 50 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 35 mol % to about 45 mol %, from about 37 mol % to about 45 mol %, or about 35 mol %, 36 mol %, 37 mol %, 38 mol %, 39 mol %, 40 mol %, 41 mol %, 42 mol %, 43 mol %, 44 mol %, or 45 mol % (or any fraction thereof or range therein
- the lipid particles contain a mixture of phospholipid and cholesterol or a cholesterol derivative
- the mixture may comprise up to about 40 mol %, 45 mol %, 50 mol %, 55 mol %, or 60 mol % of the total lipid present in the particle.
- the phospholipid component in the mixture may comprise from about 2 mol % to about 20 mol %, from about 2 mol % to about 15 mol %, from about 2 mol % to about 12 mol %, from about 4 mol % to about 15 mol %, or from about 4 mol % to about 10 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the phospholipid component in the mixture comprises from about 5 mol % to about 17 mol %, from about 7 mol % to about 17 mol %, from about 7 mol % to about 15 mol %, from about 8 mol % to about 15 mol %, or about 8 mol %, 9 mol %, 10 mol %, 1 1 mol %, 12 mol %, 13 mol %, 14 mol %, or 15 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- a lipid particle formulation comprising a mixture of phospholipid and cholesterol may comprise a phospholipid such as DPPC or DSPC at about 7 mol % (or any fraction thereof), e.g., in a mixture with cholesterol or a cholesterol derivative at about 34 mol % (or any fraction thereof) of the total lipid present in the particle.
- a lipid particle formulation comprising a mixture of phospholipid and cholesterol may comprise a phospholipid such as DPPC or DSPC at about 7 mol % (or any fraction thereof), e.g., in a mixture with cholesterol or a cholesterol derivative at about 32 mol % (or any fraction thereof) of the total lipid present in the particle.
- a lipid formulation useful has a lipid to drug (e.g., siRNA) ratio of about 10: 1 (e.g., a lipid:drug ratio of from 9.5 : 1 to 1 1 : 1, or from 9.9: 1 to 1 1 : 1, or from 10: 1 to 10.9: 1).
- a lipid to drug e.g., siRNA
- a lipid:drug ratio of from 9.5 : 1 to 1 1 : 1, or from 9.9: 1 to 1 1 : 1, or from 10: 1 to 10.9: 1).
- a lipid formulation useful has a lipid to drug (e.g., siRNA) ratio of about 9: 1 (e.g., a lipid:drug ratio of from 8.5 : 1 to 10: 1, or from 8.9: 1 to 10: 1, or from 9: 1 to 9.9: 1, including 9.1 : 1, 9.2: 1, 9.3 : 1, 9.4: 1, 9.5 : 1, 9.6: 1, 9.7: 1, and 9.8: 1).
- a lipid to drug e.g., siRNA
- a lipid to drug e.g., siRNA ratio of about 9: 1
- a lipid:drug ratio of from 8.5 : 1 to 10: 1, or from 8.9: 1 to 10: 1, or from 9: 1 to 9.9: 1, including 9.1 : 1, 9.2: 1, 9.3 : 1, 9.4: 1, 9.5 : 1, 9.6: 1, 9.7: 1, and 9.8: 1).
- the cholesterol component in the mixture may comprise from about 25 mol % to about 45 mol %, from about 25 mol % to about 40 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 27 mol % to about 37 mol %, from about 25 mol % to about 30 mol %, or from about 35 mol % to about 40 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the cholesterol component in the mixture comprises from about 25 mol % to about 35 mol %, from about 27 mol % to about 35 mol %, from about 29 mol % to about 35 mol %, from about 30 mol % to about 35 mol %, from about 30 mol % to about 34 mol %, from about 31 mol % to about 33 mol %, or about 30 mol %, 31 mol %, 32 mol %, 33 mol %, 34 mol %, or 35 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the cholesterol or derivative thereof may comprise up to about 25 mol %, 30 mol %, 35 mol %, 40 mol %, 45 mol %, 50 mol %, 55 mol %, or 60 mol % of the total lipid present in the particle.
- the cholesterol or derivative thereof in the phospholipid-free lipid particle formulation may comprise from about 25 mol % to about 45 mol %, from about 25 mol % to about 40 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 31 mol % to about 39 mol %, from about 32 mol % to about 38 mol %, from about 33 mol % to about 37 mol %, from about 35 mol % to about 45 mol %, from about 30 mol % to about 35 mol %, from about 35 mol % to about 40 mol %, or about 30 mol %, 31 mol %, 32 mol %, 33 mol %, 34 mol %, 35 mol %, 36 mol %, 37 mol %, 38 mol %, 39 mol %, or 40 mol % (or any fraction thereof or range therein) of the total mol %,
- a lipid particle formulation may comprise cholesterol at about 37 mol % (or any fraction thereof) of the total lipid present in the particle.
- a lipid particle formulation may comprise cholesterol at about 35 mol % (or any fraction thereof) of the total lipid present in the particle.
- the non-cationic lipid comprises from about 5 mol % to about 90 mol %, from about 10 mol % to about 85 mol %, from about 20 mol % to about 80 mol %, about 10 mol % (e.g., phospholipid only), or about 60 mol % (e.g., phospholipid and cholesterol or derivative thereof) (or any fraction thereof or range therein) of the total lipid present in the particle.
- the percentage of non-cationic lipid present in the lipid particles is a target amount, and that the actual amount of non-cationic lipid present in the formulation may vary, for example, by ⁇ 5 mol %, ⁇ 4 mol %, ⁇ 3 mol %, ⁇ 2 mol %, ⁇ 1 mol %, ⁇ 0.75 mol %, ⁇ 0.5 mol %, ⁇ 0.25 mol %, or ⁇ 0.1 mol %.
- the lipid particles may further comprise a lipid conjugate.
- the conjugated lipid is useful in that it prevents the aggregation of particles.
- Suitable conjugated lipids include, but are not limited to, PEG-lipid conjugates, POZ-lipid conjugates, ATTA-lipid conjugates, cationic-polymer-lipid conjugates (CPLs), and mixtures thereof.
- the particles comprise either a PEG-lipid conjugate or an ATTA-lipid conjugate together with a CPL.
- the lipid conjugate is a PEG-lipid.
- PEG-lipids include, but are not limited to, PEG coupled to dialkyloxypropyls (PEG-DAA) as described in, e.g., PCT Publication No. WO 05/026372, PEG coupled to diacylglycerol (PEG-DAG) as described in, e.g., U.S. Patent Publication Nos. 20030077829 and 2005008689, PEG coupled to phospholipids such as phosphatidylethanolamine (PEG-PE), PEG conjugated to ceramides as described in, e.g., U.S. Patent No. 5,885,613, PEG conjugated to cholesterol or a derivative thereof, and mixtures thereof.
- PEG-lipids include, but are not limited to, PEG coupled to dialkyloxypropyls (PEG-DAA) as described in, e.g., PCT Publication No. WO 05/026372, PEG
- PEG-lipids suitable for use include, without limitation, mPEG2000-l,2-di- 0-alkyl-s «3-carbomoylglyceride (PEG-C-DOMG).
- PEG-C-DOMG The synthesis of PEG-C-DOMG is described in PCT Publication No. WO 09/086558, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- Yet additional suitable PEG-lipid conjugates include, without limitation, l-[8'-(l,2-dimyristoyl-3-propanoxy)-carboxamido-3',6'- dioxaoctanyl]carbamoyl-ro-methyl-poly(ethylene glycol) (2KPEG-DMG).
- 2KPEG-DMG The synthesis of 2KPEG-DMG is described in U.S. Patent No. 7,404,969, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- PEG is a linear, water-soluble polymer of ethylene PEG repeating units with two terminal hydroxyl groups. PEGs are classified by their molecular weights; for example, PEG 2000 has an average molecular weight of about 2,000 daltons, and PEG 5000 has an average molecular weight of about 5,000 daltons. PEGs are commercially available from Sigma
- MePEG-S monomethoxypolyethylene glycol-succinimidyl succinate
- MePEG-NEh monomethoxypolyethylene glycol-amine
- MePEG-TRES monomethoxypolyethylene glycol-tresylate
- MePEG- EVI monomethoxypolyethylene glycol-imidazolyl-carbonyl
- Other PEGs such as those described in U. S. Patent Nos.
- MePEG-CH 2 COOH monomethoxypolyethyleneglycol-acetic acid
- the PEG moiety of the PEG-lipid conjugates described herein may comprise an average molecular weight ranging from about 550 daltons to about 10,000 daltons. In certain instances, the PEG moiety has an average molecular weight of from about 750 daltons to about 5,000 daltons (e.g., from about 1,000 daltons to about 5,000 daltons, from about 1 ,500 daltons to about 3,000 daltons, from about 750 daltons to about 3,000 daltons, from about 750 daltons to about 2,000 daltons, etc.). In preferred embodiments, the PEG moiety has an average molecular weight of about 2,000 daltons or about 750 daltons.
- the PEG can be optionally substituted by an alkyl, alkoxy, acyl, or aryl group.
- the PEG can be conjugated directly to the lipid or may be linked to the lipid via a linker moiety.
- Any linker moiety suitable for coupling the PEG to a lipid can be used including, e.g., non-ester containing linker moieties and ester-containing linker moieties.
- the linker moiety is a non-ester containing linker moiety.
- non-ester containing linker moiety refers to a linker moiety that does not contain a carboxylic ester bond (-OC(O)-).
- Suitable non-ester containing linker moieties include, but are not limited to, amido (-C(O) H-), amino (- R-), carbonyl (-C(O)-), carbamate (- HC(O)O-), urea (- HC(O) H-), disulphide (-S-S-), ether (-0-), succinyl (-(0)CCH 2 CH 2 C(0)-), succinamidyl (- HC(0)CH 2 CH 2 C(0) H-), ether, disulphide, as well as combinations thereof (such as a linker containing both a carbamate linker moiety and an amido linker moiety).
- a carbamate linker is used to couple the PEG to the lipid.
- an ester containing linker moiety is used to couple the PEG to the lipid.
- Suitable ester containing linker moieties include, e.g., carbonate (-OC(O)O-), succinoyl, phosphate esters (-O-(O)POH-O-), sulfonate esters, and combinations thereof.
- Phosphatidylethanolamines having a variety of acyl chain groups of varying chain lengths and degrees of saturation can be conjugated to PEG to form the lipid conjugate.
- Such phosphatidylethanolamines are commercially available, or can be isolated or synthesized using conventional techniques known to those of skill in the art.
- Phosphatidylethanolamines with mono- or diunsaturated fatty acids and mixtures of saturated and unsaturated fatty acids can also be used.
- Suitable phosphatidylethanolamines include, but are not limited to, dimyristoyl- phosphatidylethanolamine (DMPE), dipalmitoyl-phosphatidylethanolamine (DPPE), dioleoylphosphatidylethanolamine (DOPE), and distearoyl-phosphatidylethanolamine (DSPE).
- ATTA or "polyamide” includes, without limitation, compounds described in U.S. Patent Nos. 6,320,017 and 6,586,559, the disclosures of which are herein incorporated by reference in their entirety for all purposes. These compounds include a compound having the formula:
- R is a member selected from the group consisting of hydrogen, alkyl and acyl
- R 1 is a member selected from the group consisting of hydrogen and alkyl; or optionally, R and R 1 and the nitrogen to which they are bound form an azido moiety
- R 2 is a member of the group selected from hydrogen, optionally substituted alkyl, optionally substituted aryl and a side chain of an amino acid
- R 3 is a member selected from the group consisting of hydrogen, halogen, hydroxy, alkoxy, mercapto, hydrazino, amino and R 4 R 5 , wherein R 4 and R 5 are independently hydrogen or alkyl
- n is 4 to 80
- m is 2 to 6
- p is 1 to 4
- q is 0 or 1. It will be apparent to those of skill in the art that other polyamides can be.
- diacylglycerol or “DAG” includes a compound having 2 fatty acyl chains, R 1 and R 2 , both of which have independently between 2 and 30 carbons bonded to the 1- and 2- position of glycerol by ester linkages.
- the acyl groups can be saturated or have varying degrees of unsaturation. Suitable acyl groups include, but are not limited to, lauroyl (C 12 ), myristoyl (C 14 ), palmitoyl (C 16 ), stearoyl (C 18 ), and icosoyl (C 20 ).
- R 1 and R 2 are the same, i.e., R 1 and R 2 are both myristoyl ⁇ i.e., dimyristoyl), R 1 and R 2 are both stearoyl ⁇ i.e., distearoyl), etc.
- Diacylglycerols have the following general formula:
- dialkyloxypropyl or “DAA” includes a compound having 2 alkyl chains, R 1 and R 2 , both of which have independently between 2 and 30 carbons.
- the alkyl groups can be saturated or have varying degrees of unsaturation
- Dialkyloxypropyls have the following general formula:
- the PEG-lipid is a PEG-DAA conjugate having the following formula:
- R 1 and R 2 are independently selected and are long-chain alkyl groups having from about 10 to about 22 carbon atoms; PEG is a polyethyleneglycol; and L is a non-ester containing linker moiety or an ester containing linker moiety as described above.
- the long-chain alkyl groups can be saturated or unsaturated. Suitable alkyl groups include, but are not limited to, decyl (Cio), lauryl (C 12 ), myristyl (C 14 ), palmityl (C 16 ), stearyl (C 18 ), and icosyl (C 20 ).
- R 1 and R 2 are the same, i.e., R 1 and R 2 are both myristyl ⁇ i.e., dimyristyl), R 1 and R 2 are both stearyl ⁇ i.e., distearyl), etc.
- the PEG has an average molecular weight ranging from about 550 daltons to about 10,000 daltons. In certain instances, the PEG has an average molecular weight of from about 750 daltons to about 5,000 daltons ⁇ e.g., from about 1 ,000 daltons to about 5,000 daltons, from about 1,500 daltons to about 3,000 daltons, from about 750 daltons to about 3,000 daltons, from about 750 daltons to about 2,000 daltons, etc.). In preferred embodiments, the PEG has an average molecular weight of about 2,000 daltons or about 750 daltons.
- the PEG can be optionally substituted with alkyl, alkoxy, acyl, or aryl groups. In certain embodiments, the terminal hydroxyl group is substituted with a methoxy or methyl group.
- L is a non-ester containing linker moiety.
- Suitable non- ester containing linkers include, but are not limited to, an amido linker moiety, an amino linker moiety, a carbonyl linker moiety, a carbamate linker moiety, a urea linker moiety, an ether linker moiety, a disulphide linker moiety, a succinamidyl linker moiety, and combinations thereof.
- the non-ester containing linker moiety is a carbamate linker moiety ⁇ i.e. , a PEG-C-DAA conjugate).
- the non-ester containing linker moiety is an amido linker moiety ⁇ i.e. , a PEG- 4-DAA conjugate). In yet another preferred embodiment, the non-ester containing linker moiety is a succinamidyl linker moiety (i.e., a PEG- ⁇ -DAA conjugate).
- the PEG-lipid conjugate is selected from:
- the PEG-DAA conjugates are synthesized using standard techniques and reagents known to those of skill in the art. It will be recognized that the PEG-DAA conjugates will contain various amide, amine, ether, thio, carbamate, and urea linkages. Those of skill in the art will recognize that methods and reagents for forming these bonds are well known and readily available. See, e.g., March, ADVANCED ORGANIC CHEMISTRY (Wiley 1992); Larock,
- the PEG-DAA conjugate is a PEG-didecyloxypropyl (Cio) conjugate, a PEG-dilauryloxypropyl (C12) conjugate, a PEG-dimyristyloxypropyl (C14) conjugate, a PEG- dipalmityloxypropyl (Ci 6 ) conjugate, or a PEG-distearyloxypropyl (Cis) conjugate.
- the PEG preferably has an average molecular weight of about 750 or about 2,000 daltons.
- the PEG-lipid conjugate comprises
- the PEG-lipid conjugate comprises PEG750-C- DMA, wherein the "750” denotes the average molecular weight of the PEG, the “C” denotes a carbamate linker moiety, and the "DMA” denotes dimyristyloxypropyl.
- the terminal hydroxyl group of the PEG is substituted with a methyl group.
- dialkyloxypropyls can be used in the PEG-DAA conjugates.
- hydrophilic polymers can be used in place of PEG. Examples of suitable polymers that can be used in place of PEG include, but are not limited to, polyvinylpyrrolidone,
- polymethyloxazoline polyethyloxazoline, polyhydroxypropyl methacrylamide,
- polymethacrylamide and polydimethylacrylamide polylactic acid, polyglycolic acid, and derivatized celluloses such as hydroxymethyl cellulose or hydroxyethylcellulose.
- the lipid particles can further comprise cationic poly(ethylene glycol) (PEG) lipids or CPLs ⁇ see, e.g., Chen et al., Bioconj. Chem., 11 :433-437 (2000); U.S. Patent No. 6,852,334; PCT Publication No. WO 00/62813, the disclosures of which are herein incorporated by reference in their entirety for all purposes).
- PEG poly(ethylene glycol)
- Suitable CPLs include compounds of Formula VIII:
- A is a lipid moiety such as an amphipathic lipid, a neutral lipid, or a hydrophobic lipid that acts as a lipid anchor.
- Suitable lipid examples include, but are not limited to, diacylglycerolyls, dialkylglycerolyls, N-N-dialkylaminos, 1,2-diacyloxy- 3-aminopropanes, and l,2-dialkyl-3-aminopropanes.
- W is a polymer or an oligomer such as a hydrophilic polymer or oligomer.
- the hydrophilic polymer is a biocompatable polymer that is nonimmunogenic or possesses low inherent immunogenicity.
- the hydrophilic polymer can be weakly antigenic if used with appropriate adjuvants.
- Suitable nonimmunogenic polymers include, but are not limited to, PEG, polyamides, polylactic acid, polyglycolic acid, polylactic
- the polymer has a molecular weight of from about 250 to about 7,000 daltons.
- Y is a polycationic moiety.
- polycationic moiety refers to a compound, derivative, or functional group having a positive charge, preferably at least 2 positive charges at a selected pH, preferably physiological pH.
- Suitable polycationic moieties include basic amino acids and their derivatives such as arginine, asparagine, glutamine, lysine, and histidine;
- the polycationic moieties can be linear, such as linear tetralysine, branched or dendrimeric in structure.
- Polycationic moieties have between about 2 to about 15 positive charges, preferably between about 2 to about 12 positive charges, and more preferably between about 2 to about 8 positive charges at selected pH values.
- the selection of which polycationic moiety to employ may be determined by the type of particle application which is desired.
- the charges on the polycationic moieties can be either distributed around the entire particle moiety, or alternatively, they can be a discrete concentration of charge density in one particular area of the particle moiety e.g., a charge spike. If the charge density is distributed on the particle, the charge density can be equally distributed or unequally distributed. All variations of charge distribution of the polycationic moiety are encompassed.
- the lipid "A” and the nonimmunogenic polymer “W” can be attached by various methods and preferably by covalent attachment. Methods known to those of skill in the art can be used for the covalent attachment of "A” and “W.” Suitable linkages include, but are not limited to, amide, amine, carboxyl, carbonate, carbamate, ester, and hydrazone linkages. It will be apparent to those skilled in the art that "A” and “W” must have complementary functional groups to effectuate the linkage. The reaction of these two groups, one on the lipid and the other on the polymer, will provide the desired linkage.
- the lipid is a diacylglycerol and the terminal hydroxyl is activated, for instance with NHS and DCC, to form an active ester, and is then reacted with a polymer which contains an amino group, such as with a polyamide (see, e.g., U.S. Patent Nos. 6,320,017 and 6,586,559, the disclosures of which are herein incorporated by reference in their entirety for all purposes), an amide bond will form between the two groups.
- a polyamide see, e.g., U.S. Patent Nos. 6,320,017 and 6,586,559, the disclosures of which are herein incorporated by reference in their entirety for all purposes
- the polycationic moiety can have a ligand attached, such as a targeting ligand or a chelating moiety for complexing calcium.
- a ligand attached such as a targeting ligand or a chelating moiety for complexing calcium.
- the cationic moiety maintains a positive charge.
- the ligand that is attached has a positive charge.
- Suitable ligands include, but are not limited to, a compound or device with a reactive functional group and include lipids, amphipathic lipids, carrier
- bioaffinity compounds biomaterials, biopolymers, biomedical devices, analytically detectable compounds, therapeutically active compounds, enzymes, peptides, proteins, antibodies, immune stimulators, radiolabels, fluorogens, biotin, drugs, haptens, DNA, RNA, polysaccharides, liposomes, virosomes, micelles, immunoglobulins, functional groups, other targeting moieties, or toxins.
- the lipid conjugate (e.g., PEG-lipid) comprises from about 0.1 mol % to about 3 mol %, from about 0.5 mol % to about 3 mol %, or about 0.6 mol %, 0.7 mol %, 0.8 mol %, 0.9 mol %, 1.0 mol %, 1.1 mol %, 1.2 mol %, 1.3 mol %, 1.4 mol %, 1.5 mol %, 1.6 mol %, 1.7 mol %, 1.8 mol %, 1.9 mol %, 2.0 mol %, 2.1 mol%, 2.2 mol%, 2.3 mol %, 2.4 mol %, 2.5 mol %, 2.6 mol %, 2.7 mol %, 2.8 mol %, 2.9 mol % or 3 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- the lipid conjugate (e.g., PEG-lipid) comprises from about 0 mol % to about 20 mol %, from about 0.5 mol % to about 20 mol %, from about 2 mol % to about 20 mol %, from about 1.5 mol % to about 18 mol %, from about 2 mol % to about 15 mol %, from about 4 mol % to about 15 mol %, from about 2 mol % to about 12 mol %, from about 5 mol % to about 12 mol %, or about 2 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- PEG-lipid comprises from about 0 mol % to about 20 mol %, from about 0.5 mol % to about 20 mol %, from about 2 mol % to about 20 mol %, from about 1.5 mol % to about 18 mol %, from about 2 mol % to about 15 mol %, from about 4
- the lipid conjugate (e.g., PEG-lipid) comprises from about 4 mol % to about 10 mol %, from about 5 mol % to about 10 mol %, from about 5 mol % to about 9 mol %, from about 5 mol % to about 8 mol %, from about 6 mol % to about 9 mol %, from about 6 mol % to about 8 mol %, or about 5 mol %, 6 mol %, 7 mol %, 8 mol %, 9 mol %, or 10 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
- PEG-lipid comprises from about 4 mol % to about 10 mol %, from about 5 mol % to about 10 mol %, from about 5 mol % to about 9 mol %, from about 5 mol % to about 8 mol %, from about 6 mol % to about 9 mol %, from about 6 mol %
- the percentage of lipid conjugate present in the lipid particles is a target amount, and that the actual amount of lipid conjugate present in the formulation may vary, for example, by ⁇ 5 mol %, ⁇ 4 mol %, ⁇ 3 mol %, ⁇ 2 mol %, ⁇ 1 mol %, ⁇ 0.75 mol %, ⁇ 0.5 mol %, ⁇ 0.25 mol %, or ⁇ 0.1 mol %.
- concentration of the lipid conjugate can be varied depending on the lipid conjugate employed and the rate at which the lipid particle is to become fusogenic.
- the rate at which the lipid conjugate exchanges out of the lipid particle and, in turn, the rate at which the lipid particle becomes fusogenic can be varied, for example, by varying the concentration of the lipid conjugate, by varying the molecular weight of the PEG, or by varying the chain length and degree of saturation of the alkyl groups on the PEG-DAA conjugate.
- other variables including, for example, pH, temperature, ionic strength, etc. can be used to vary and/or control the rate at which the lipid particle becomes fusogenic. Other methods which can be used to control the rate at which the lipid particle becomes fusogenic will become apparent to those of skill in the art upon reading this disclosure.
- the composition and concentration of the lipid conjugate one can control the lipid particle size. Additional Carrier Systems
- Non-limiting examples of additional lipid-based carrier systems suitable for use include lipoplexes ⁇ see, e.g., U.S. Patent Publication No. 20030203865; and Zhang et al, J. Control Release, 100: 165-180 (2004)), pH-sensitive lipoplexes (see, e.g., U.S. Patent
- WO 03/057190 and WO 03/059322 lipid- encapsulated nucleic acid (see, e.g., U.S. Patent Publication No. 20030129221; and U.S. Patent No. 5,756,122), other liposomal compositions (see, e.g., U.S. Patent Publication Nos.
- polymer-based carrier systems suitable for use include, but are not limited to, cationic polymer-nucleic acid complexes (i.e., polyplexes).
- a nucleic acid e.g., a siRNA molecule described herein or a combination thereof
- a cationic polymer having a linear, branched, star, or dendritic polymeric structure that condenses the nucleic acid into positively charged particles capable of interacting with anionic proteoglycans at the cell surface and entering cells by endocytosis.
- the polyplex comprises nucleic acid (e.g., a combination comprising the three siRNA molecules described in Example 1) complexed with a cationic polymer such as polyethylenimine (PEI)
- CA as In v/vojetPEITM, a linear form of PEI), polypropylenimine (PPI), polyvinylpyrrolidone
- PVP poly-L-lysine
- DEAE diethylaminoethyl
- PAE poly(P-amino ester)
- PAMAM polyamidoamine dendrimers
- PAMAM polyamidoamine dendrimers
- porphyrin see, e.g., U.S. Patent No.
- polyvinylether see, e.g., U.S. Patent Publication No. 20040156909
- polycyclic amidinium see, e.g., U.S. Patent Publication No. 20030220289
- other polymers comprising primary amine, imine, guanidine, and/or imidazole groups (see, e.g., U.S. Patent No. 6,013,240; PCT
- the polyplex comprises cationic polymer-nucleic acid complexes as described in U.S. Patent Publication Nos. 20060211643, 20050222064, 20030125281, and 20030185890, and PCT Publication No. WO 03/066069; biodegradable poly( -amino ester) polymer-nucleic acid complexes as described in U.S. Patent Publication No. 20040071654; microparticles containing polymeric matrices as described in U.S. Patent Publication No.
- the siRNA may be complexed with cyclodextrin or a polymer thereof.
- cyclodextrin-based carrier systems include the cyclodextrin- modified polymer-nucleic acid complexes described in U.S. Patent Publication No.
- siRNA may be complexed with a peptide or polypeptide.
- a protein-based carrier system includes, but is not limited to, the cationic oligopeptide-nucleic acid complex described in PCT Publication No. W095/21931.
- nucleic acid-lipid particles in which a nucleic acid (e.g., a combination
- Example 1 comprising the three siRNA molecules described in Example 1) is entrapped within the lipid portion of the particle and is protected from degradation, can be formed by any method known in the art including, but not limited to, a continuous mixing method, a direct dilution process, and an in-line dilution process.
- the cationic lipids may comprise lipids of Formula I-III or salts thereof, alone or in combination with other cationic lipids.
- the non- cationic lipids are egg sphingomyelin (ESM), distearoylphosphatidylcholine (DSPC), dioleoylphosphatidyl choline (DOPC), l-palmitoyl-2-oleoyl-phosphatidyl choline (POPC), dipalmitoyl-phosphatidyl choline (DPPC), monomethyl-phosphatidylethanolamine, dimethyl - phosphatidylethanolamine, 14:0 PE (1,2-dimyristoyl-phosphatidylethanolamine (DMPE)), 16:0 PE (1,2-dipalmitoyl-phosphatidylethanolamine (DPPE)), 18:0 PE (1,2-distearoyl- phosphatidylethanolamine (DSPE)), 18: 1
- the nucleic acid-lipid particles produced via a continuous mixing method e.g., a process that includes providing an aqueous solution comprising a siRNA in a first reservoir, providing an organic lipid solution in a second reservoir (wherein the lipids present in the organic lipid solution are solubilized in an organic solvent, e.g., a lower alkanol such as ethanol), and mixing the aqueous solution with the organic lipid solution such that the organic lipid solution mixes with the aqueous solution so as to substantially instantaneously produce a lipid vesicle (e.g., liposome) encapsulating the siRNA within the lipid vesicle.
- a lipid vesicle e.g., liposome
- the organic lipid solution undergoes a continuous stepwise dilution in the presence of the buffer solution (i.e., aqueous solution) to produce a nucleic acid-lipid particle.
- the buffer solution i.e., aqueous solution
- the nucleic acid-lipid particles formed using the continuous mixing method typically have a size of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 110 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, less than about 120 nm, 110 nm, 100 nm, 90 nm, or 80 nm, or about 30 nm, 35 nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 n
- the nucleic acid-lipid particles produced via a direct dilution process that includes forming a lipid vesicle (e.g., liposome) solution and immediately and directly introducing the lipid vesicle solution into a collection vessel containing a controlled amount of dilution buffer.
- the collection vessel includes one or more elements configured to stir the contents of the collection vessel to facilitate dilution.
- the amount of dilution buffer present in the collection vessel is substantially equal to the volume of lipid vesicle solution introduced thereto.
- a lipid vesicle solution in 45% ethanol when introduced into the collection vessel containing an equal volume of dilution buffer will advantageously yield smaller particles.
- the nucleic acid-lipid particles produced via an in-line dilution process in which a third reservoir containing dilution buffer is fluidly coupled to a second mixing region.
- the lipid vesicle (e.g., liposome) solution formed in a first mixing region is immediately and directly mixed with dilution buffer in the second mixing region.
- the second mixing region includes a T-connector arranged so that the lipid vesicle solution and the dilution buffer flows meet as opposing 180° flows; however, connectors providing shallower angles can be used, e.g., from about 27° to about 180° (e.g., about 90°).
- a pump mechanism delivers a controllable flow of buffer to the second mixing region.
- the flow rate of dilution buffer provided to the second mixing region is controlled to be substantially equal to the flow rate of lipid vesicle solution introduced thereto from the first mixing region.
- This embodiment advantageously allows for more control of the flow of dilution buffer mixing with the lipid vesicle solution in the second mixing region, and therefore also the concentration of lipid vesicle solution in buffer throughout the second mixing process.
- Such control of the dilution buffer flow rate advantageously allows for small particle size formation at reduced concentrations.
- the nucleic acid-lipid particles formed using the direct dilution and in-line dilution processes typically have a size of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 1 10 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, less than about 120 nm, 1 10 nm, 100 nm, 90 nm, or 80 nm, or about 30 nm, 35 nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm,
- the lipid particles can be sized by any of the methods available for sizing liposomes.
- the sizing may be conducted in order to achieve a desired size range and relatively narrow distribution of particle sizes.
- Extrusion of the particles through a small-pore polycarbonate membrane or an asymmetric ceramic membrane is also an effective method for reducing particle sizes to a relatively well-defined size distribution.
- the suspension is cycled through the membrane one or more times until the desired particle size distribution is achieved.
- the particles may be extruded through successively smaller-pore membranes, to achieve a gradual reduction in size.
- the nucleic acids present in the particles are precondensed as described in, e.g., U. S. Patent Application No. 09/744, 103, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
- the methods may further comprise adding non-lipid polycations which are useful to effect the lipofection of cells using the present compositions.
- suitable non-lipid polycations include, hexadimethrine bromide (sold under the brand name POLYBRENE®, from Aldrich Chemical Co., Milwaukee, Wisconsin, USA) or other salts of hexadimethrine.
- suitable polycations include, for example, salts of poly-L-ornithine, poly-L-arginine, poly-L-lysine, poly-D-lysine, polyallylamine, and polyethyleneimine. Addition of these salts is preferably after the particles have been formed.
- the nucleic acid (e.g., siRNA) to lipid ratios (mass/mass ratios) in a formed nucleic acid-lipid particle will range from about 0.01 to about 0.2, from about 0.05 to about 0.2, from about 0.02 to about 0.1, from about 0.03 to about 0.1, or from about 0.01 to about 0.08.
- the ratio of the starting materials (input) also falls within this range.
- the particle preparation uses about 400 ⁇ g nucleic acid per 10 mg total lipid or a nucleic acid to lipid mass ratio of about 0.01 to about 0.08 and, more preferably, about 0.04, which corresponds to 1.25 mg of total lipid per 50 ⁇ g of nucleic acid.
- the particle has a nucleic acid:lipid mass ratio of about 0.08.
- the lipid to nucleic acid (e.g., siRNA) ratios (mass/mass ratios) in a formed nucleic acid-lipid particle will range from about 1 (1 : 1) to about 100 (100: 1), from about 5 (5: 1) to about 100 (100: 1), from about 1 (1 : 1) to about 50 (50: 1), from about 2 (2: 1) to about 50 (50: 1), from about 3 (3 : 1) to about 50 (50: 1), from about 4 (4: 1) to about 50 (50: 1), from about 5 (5: 1) to about 50 (50: 1), from about 1 (1 : 1) to about 25 (25: 1), from about 2 (2: 1) to about 25 (25 : 1), from about 3 (3 : 1) to about 25 (25 : 1), from about 4 (4: 1) to about 25 (25 : 1), from about 5 (5: 1) to about 25 (25: 1), from about 5 (5: 1) to about 20 (20: 1), from about 5 (5: 1) to about 15 (15: 1), from about 5 (5: 1) to
- the conjugated lipid may further include a CPL.
- CPL-containing lipid particles A variety of general methods for making lipid particle-CPLs (CPL-containing lipid particles) are discussed herein. Two general techniques include the "post-insertion” technique, that is, insertion of a CPL into, for example, a pre-formed lipid particle, and the "standard” technique, wherein the CPL is included in the lipid mixture during, for example, the lipid particle formation steps.
- the post-insertion technique results in lipid particles having CPLs mainly in the external face of the lipid particle bilayer membrane, whereas standard techniques provide lipid particles having CPLs on both internal and external faces.
- the method is especially useful for vesicles made from phospholipids (which can contain cholesterol) and also for vesicles containing PEG-lipids (such as PEG-DAAs and PEG-DAGs).
- PEG-lipids such as PEG-DAAs and PEG-DAGs.
- Methods of making lipid particle-CPLs are taught, for example, in U.S. Patent Nos. 5,705,385; 6,586,410; 5,981,501; 6,534,484; and 6,852,334; U.S. Patent Publication No. 20020072121; and PCT Publication No. WO 00/62813, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
- the lipid particles can be adsorbed to almost any cell type with which they are mixed or contacted. Once adsorbed, the particles can either be endocytosed by a portion of the cells, exchange lipids with cell membranes, or fuse with the cells. Transfer or incorporation of the siRNA portion of the particle can take place via any one of these pathways. In particular, when fusion takes place, the particle membrane is integrated into the cell membrane and the contents of the particle combine with the intracellular fluid.
- the lipid particles can be administered either alone or in a mixture with a pharmaceutically acceptable carrier (e.g., physiological saline or phosphate buffer) selected in accordance with the route of administration and standard pharmaceutical practice.
- a pharmaceutically acceptable carrier e.g., physiological saline or phosphate buffer
- physiological saline or phosphate buffer e.g., physiological saline or phosphate buffer
- suitable carriers include, e.g., water, buffered water, 0.4% saline, 0.3% glycine, and the like, including glycoproteins for enhanced stability, such as albumin, lipoprotein, globulin, etc. Additional suitable carriers are described in, e.g.,
- carrier includes any and all solvents, dispersion media, vehicles, coatings, diluents, antibacterial and antifungal agents, isotonic and absorption delaying agents, buffers, carrier solutions, suspensions, colloids, and the like.
- pharmaceutically acceptable refers to molecular entities and compositions that do not produce an allergic or similar untoward reaction when administered to a human.
- the pharmaceutically acceptable carrier is generally added following lipid particle formation.
- the particle can be diluted into
- pharmaceutically acceptable carriers such as normal buffered saline.
- the concentration of particles in the pharmaceutical formulations can vary widely, i.e., from less than about 0.05%, usually at or at least about 2 to 5%, to as much as about 10 to 90% by weight, and will be selected primarily by fluid volumes, viscosities, etc., in accordance with the particular mode of administration selected.
- the concentration may be increased to lower the fluid load associated with treatment. This may be particularly desirable in patients having atherosclerosis-associated congestive heart failure or severe hypertension.
- particles composed of irritating lipids may be diluted to low concentrations to lessen
- compositions may be sterilized by conventional, well-known sterilization techniques.
- Aqueous solutions can be packaged for use or filtered under aseptic conditions and lyophilized, the lyophilized preparation being combined with a sterile aqueous solution prior to administration.
- the compositions can contain pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents and the like, for example, sodium acetate, sodium lactate, sodium chloride, potassium chloride, and calcium chloride.
- the particle suspension may include lipid-protective agents which protect lipids against free-radical and lipid-peroxidative damages on storage. Lipophilic free-radical quenchers, such as alphatocopherol, and water-soluble iron-specific chelators, such as ferrioxamine, are suitable.
- Systemic delivery for in vivo therapy e.g., delivery of a siRNA molecule described herein, such as a combination comprising the three siRNA molecules described in Example 1, to a distal target cell via body systems such as the circulation, may be achieved using nucleic acid- lipid particles, such as those described in PCT Publication Nos. WO 05/007196, WO 05/121348, WO 05/120152, and WO 04/002453, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
- administration can be in any manner known in the art, e.g., by injection, oral administration, inhalation ⁇ e.g., intransal or intratracheal), transdermal application, or rectal administration.
- Administration can be accomplished via single or divided doses.
- the pharmaceutical compositions can be administered parenterally, i.e., intraarticularly, intravenously, intraperitoneally, subcutaneously, or intramuscularly.
- the pharmaceutical compositions are administered intravenously or intraperitoneally by a bolus injection ⁇ see, e.g., U.S. Patent No. 5,286,634).
- Intracellular nucleic acid delivery has also been discussed in Straubringer et al. , Methods Enzymol. , 101 :512 (1983); Mannino et al,
- the lipid particles can be administered by direct injection at the site of disease or by injection at a site distal from the site of disease ⁇ see, e.g., Culver, HUMAN GENE THERAPY, Mary Ann Liebert, Inc., Publishers, New York. pp.70-71(1994)).
- Culver HUMAN GENE THERAPY
- Mary Ann Liebert, Inc. Publishers, New York. pp.70-71(1994)
- the disclosures of the above-described references are herein incorporated by reference in their entirety for all purposes.
- the lipid particles are administered intravenously, at least about 5%, 10%, 15%), 20%), or 25% of the total injected dose of the particles is present in plasma about 8, 12, 24, 36, or 48 hours after injection. In other embodiments, more than about 20%, 30%, 40%) and as much as about 60%, 70% or 80% of the total injected dose of the lipid particles is present in plasma about 8, 12, 24, 36, or 48 hours after injection. In certain instances, more than about 10%) of a plurality of the particles is present in the plasma of a human about 1 hour after administration. In certain other instances, the presence of the lipid particles is detectable at least about 1 hour after administration of the particle.
- the presence of a siRNA molecule is detectable in cells at about 8, 12, 24, 36, 48, 60, 72 or 96 hours after administration.
- downregulation of expression of a target sequence, such as a viral or host sequence, by a siRNA molecule is detectable at about 8, 12, 24, 36, 48, 60, 72 or 96 hours after administration.
- downregulation of expression of a target sequence, such as a viral or host sequence, by a siRNA molecule occurs preferentially in infected cells and/or cells capable of being infected.
- the presence or effect of a siRNA molecule in cells at a site proximal or distal to the site of administration is detectable at about 12, 24, 48, 72, or 96 hours, or at about 6, 8, 10, 12, 14, 16, 18, 19, 20, 22, 24, 26, or 28 days after administration.
- the lipid particles are
- compositions can be made into aerosol formulations (i.e., they can be "nebulized") to be administered via inhalation (e.g., intranasally or intratracheally) (see, Brigham et al, Am. J. Sci., 298:278 (1989)). Aerosol formulations can be placed into pressurized acceptable propellants, such as
- the pharmaceutical compositions may be delivered by intranasal sprays, inhalation, and/or other aerosol delivery vehicles.
- Methods for delivering nucleic acid compositions directly to the lungs via nasal aerosol sprays have been described, e.g., in U.S. Patent Nos. 5,756,353 and 5,804,212.
- delivery of drugs using intranasal microparticle resins and lysophosphatidyl -glycerol compounds U.S. Patent
- Formulations suitable for parenteral administration include aqueous and non-aqueous, isotonic sterile injection solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic with the blood of the intended recipient, and aqueous and non-aqueous sterile suspensions that can include suspending agents, solubilizers, thickening agents, stabilizers, and preservatives.
- aqueous and non-aqueous, isotonic sterile injection solutions which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic with the blood of the intended recipient
- aqueous and non-aqueous sterile suspensions that can include suspending agents, solubilizers, thickening agents, stabilizers, and preservatives.
- the lipid particle formulations are formulated with a suitable pharmaceutical carrier.
- suitable formulations are found, for example, in REMINGTON' S PHARMACEUTICAL SCIENCES, Mack Publishing Company,
- aqueous carriers may be used, for example, water, buffered water, 0.4% saline, 0.3% glycine, and the like, and may include glycoproteins for enhanced stability, such as albumin, lipoprotein, globulin, etc.
- glycoproteins for enhanced stability such as albumin, lipoprotein, globulin, etc.
- normal buffered saline (135-150 mM NaCl) will be employed as the pharmaceutically acceptable carrier, but other suitable carriers will suffice.
- These compositions can be sterilized by conventional liposomal sterilization techniques, such as filtration.
- the compositions may contain
- auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents, wetting agents and the like, for example, sodium acetate, sodium lactate, sodium chloride, potassium chloride, calcium chloride, sorbitan monolaurate, triethanolamine oleate, etc.
- These compositions can be sterilized using the techniques referred to above or, alternatively, they can be produced under sterile conditions.
- the resulting aqueous solutions may be packaged for use or filtered under aseptic conditions and lyophilized, the lyophilized preparation being combined with a sterile aqueous solution prior to administration.
- the lipid particles disclosed herein may be delivered via oral administration to the individual.
- the particles may be incorporated with excipients and used in the form of ingestible tablets, buccal tablets, troches, capsules, pills, lozenges, elixirs, mouthwash, suspensions, oral sprays, syrups, wafers, and the like ⁇ see, e.g., U.S. Patent Nos. 5,641,515, 5,580,579, and 5,792,451, the disclosures of which are herein incorporated by reference in their entirety for all purposes).
- These oral dosage forms may also contain the following: binders, gelatin; excipients, lubricants, and/or flavoring agents.
- the unit dosage form When the unit dosage form is a capsule, it may contain, in addition to the materials described above, a liquid carrier. Various other materials may be present as coatings or to otherwise modify the physical form of the dosage unit. Of course, any material used in preparing any unit dosage form should be pharmaceutically pure and substantially non-toxic in the amounts employed.
- these oral formulations may contain at least about 0.1% of the lipid particles or more, although the percentage of the particles may, of course, be varied and may
- each therapeutically useful composition may be prepared is such a way that a suitable dosage will be obtained in any given unit dose of the compound.
- Factors such as solubility, bioavailability, biological half-life, route of administration, product shelf life, as well as other pharmacological considerations will be contemplated by one skilled in the art of preparing such pharmaceutical formulations, and as such, a variety of dosages and treatment regimens may be desirable.
- Formulations suitable for oral administration can consist of: (a) liquid solutions, such as an effective amount of a packaged siRNA molecule(s) ⁇ e.g., a combination comprising the three siRNA molecules described in Example 1) suspended in diluents such as water, saline, or PEG 400; (b) capsules, sachets, or tablets, each containing a predetermined amount of a siRNA molecule(s), as liquids, solids, granules, or gelatin; (c) suspensions in an appropriate liquid; and (d) suitable emulsions.
- liquid solutions such as an effective amount of a packaged siRNA molecule(s) ⁇ e.g., a combination comprising the three siRNA molecules described in Example 1) suspended in diluents such as water, saline, or PEG 400
- capsules, sachets, or tablets each containing a predetermined amount of a siRNA molecule(s), as liquids, solids, granules, or ge
- Tablet forms can include one or more of lactose, sucrose, mannitol, sorbitol, calcium phosphates, corn starch, potato starch, microcrystalline cellulose, gelatin, colloidal silicon dioxide, talc, magnesium stearate, stearic acid, and other excipients, colorants, fillers, binders, diluents, buffering agents, moistening agents, preservatives, flavoring agents, dyes, disintegrating agents, and pharmaceutically compatible carriers.
- Lozenge forms can comprise a siRNA molecule in a flavor, e.g., sucrose, as well as pastilles comprising the therapeutic nucleic acid in an inert base, such as gelatin and glycerin or sucrose and acacia emulsions, gels, and the like containing, in addition to the siRNA molecule(s), carriers known in the art.
- a flavor e.g., sucrose
- pastilles comprising the therapeutic nucleic acid in an inert base, such as gelatin and glycerin or sucrose and acacia emulsions, gels, and the like containing, in addition to the siRNA molecule(s), carriers known in the art.
- lipid particles can be incorporated into a broad range of topical dosage forms.
- a suspension containing nucleic acid-lipid particles can be formulated and administered as gels, oils, emulsions, topical creams, pastes, ointments, lotions, foams, mousses, and the like.
- the amount of particles administered will depend upon the ratio of siRNA molecules to lipid, the particular siRNA used, the strain of HBV being treated, the age, weight, and condition of the patient, and the judgment of the clinician, but will generally be between about
- 0.01 and about 50 mg per kilogram of body weight preferably between about 0.1 and about 5 mg/kg of body weight, or about 10 8 -10 10 particles per administration (e.g., injection).
- the term "combination” means that the combined siRNA molecules are present together in the same composition of matter (e.g., dissolved together within the same solution; or present together within the same lipid particle; or present together in the same pharmaceutical formulation of lipid particles, although each lipid particle within the same composition of matter (e.g., dissolved together within the same solution; or present together within the same lipid particle; or present together in the same pharmaceutical formulation of lipid particles, although each lipid particle within the
- composition may or may not include each different siRNA of the siRNA combination).
- the combined siRNA molecules usually are not covalently linked together.
- siRNA 1 SEQ ID NO: 1 and 2
- siRNA 2 siRNA 2
- siRNA 3 SEQ ID NO: 5 and 6 are used in methods of the invention.
- the siRNA is administered via nucleic acid lipid particle.
- the different siRNA molecules are co-encapsulated in the same lipid particle.
- each type of siRNA species present in the cocktail is encapsulated in its own particle.
- siRNA species are co-encapsulated in the same particle while other siRNA species are encapsulated in different particles.
- methods of the invention include the administration of a lipid nanoparticle formulation, an interferon, and optionally, at least one additional therapeutic agent.
- these agents can be formulated together in a single preparation or that they can be formulated separately and, thus, administered separately, either simultaneously or sequentially.
- the agents when the agents are administered sequentially (e.g. at different times), the agents may be administered so that their biological effects overlap (i.e. each agent is producing a biological effect at a single given time).
- the lipid nanoparticle formulation is administered first and the IFN is administered second.
- the IFN is administered first and the lipid nanoparticle formulation is administered second.
- the administration of the lipid nanoparticle formulation begins, e.g., about 30 weeks to about 1 week, about 24 weeks to about 6 weeks, or about 12 weeks to about 6 weeks before the administration of the IFN (i.e., the lipid nanoparticle is administered in a monotherapy phase). In certain embodiments, the lipid nanoparticle is administered two or more times prior to the administration of the IFN.
- such a monotherapy phase is followed by a co-therapy phase.
- the lipid nanoparticle formulation and the IFN are administered to the human concurrently (e.g., within the same week, day or hour) during the co-therapy phase.
- the IFN and the lipid nanoparticle formulation are administered to the human concurrently (e.g., within the same week, day or hour) during the co-therapy phase.
- the IFN and the lipid nanoparticle formulation are administered to the human concurrently (e.g., within the same week, day or hour) during the co-therapy phase.
- the IFN and the lipid nanoparticle formulation are administered to the human concurrently (e.g., within the same week, day or hour) during the co-therapy phase.
- the administration of the lipid nanoparticle formulation and the IFN begin on the same day.
- the IFN and the lipid nanoparticle are administered within about 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 hour of each other.
- the administration of the IFN begins about 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 days before the
- the administration of the lipid nanoparticle formulation begins about 3 days before the administration of the lipid nanoparticle formulation.
- the administration of the lipid nanoparticle formulation and the IFN begin on the same day. In certain embodiments, the administration of the IFN begins about 12,
- the lipid nanoparticle formulation and/or the IFN are administered two or more times during the co-therapy phase.
- the agents can be formulated for and administered using any acceptable route of administration depending on the agent selected.
- suitable routes include, but are not limited to, oral, sublingual, buccal, topical, transdermal, parenteral, subcutaneous, intraperitoneal, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration.
- the small molecule agents identified herein can be administered orally.
- the oligomeric nucleotides can be administered by injection (e.g., into a blood vessel, such as a vein), or subcutaneously.
- a subject in need thereof is administered one or more agents orally (e.g., in pill form), and also one or more oligomeric nucleotides by injection or subcutaneously.
- a subject in need thereof is administered the interferon and the lipid nanoparticle formulation by injection or subcutaneously.
- the oligomeric nucleotides targeted to the Hepatitis B genome are
- the present invention is not limited to intravenous formulations comprising the oligomeric nucleotides or to treatment methods wherein an oligomeric nucleotide is administered intravenously.
- the agents can be individually formulated by mixing at ambient temperature at the appropriate pH, and at the desired degree of purity, with physiologically acceptable carriers, i.e., carriers that are non-toxic to recipients at the dosages and concentrations employed.
- physiologically acceptable carriers i.e., carriers that are non-toxic to recipients at the dosages and concentrations employed.
- the pH of the formulation depends mainly on the particular use and the concentration of compound, but may typically range anywhere from about 3 to about 8.
- the agents ordinarily will be stored as a solid composition, although lyophilized formulations or aqueous solutions are acceptable.
- compositions comprising the agents can be formulated, dosed, and administered in a fashion consistent with good medical practice.
- Factors for consideration in this context include the particular disorder being treated, the particular human being treated, the clinical condition of the individual patient, the cause of the disorder, the site of administration, the method of administration, the scheduling of administration, and other factors known to medical
- the agents may be administered in any convenient administrative form, e.g., tablets, powders, capsules, solutions, dispersions, suspensions, syrups, sprays, suppositories, gels, emulsions, patches, etc.
- Such compositions may contain components conventional in
- compositions e.g., diluents, carriers, pH modifiers, sweeteners, bulking agents, and further active agents. If parenteral administration is desired, the compositions will be sterile and in a solution or suspension form suitable for injection or infusion.
- Suitable carriers and excipients are well known to those skilled in the art and are described in detail in, e.g., Ansel, Howard C, et al., Ansel's Pharmaceutical Dosage Forms and Drug Delivery Systems. Philadelphia: Lippincott, Williams & Wilkins, 2004; Gennaro, Alfonso R., et al. Remington: The Science and Practice of Pharmacy. Philadelphia: Lippincott, Williams & Wilkins, 2000; and Rowe, Raymond C. Handbook of Pharmaceutical Excipients. Chicago, Pharmaceutical Press, 2005.
- the formulations may also include one or more buffers, stabilizing agents, surfactants, wetting agents, lubricating agents, emulsifiers, suspending agents, preservatives, antioxidants, opaquing agents, glidants, processing aids, colorants, sweeteners, perfuming agents, flavoring agents, diluents and other known additives to provide an elegant presentation of the drug or aid in the manufacturing of the pharmaceutical product (i.e., medicament).
- buffers stabilizing agents, surfactants, wetting agents, lubricating agents, emulsifiers, suspending agents, preservatives, antioxidants, opaquing agents, glidants, processing aids, colorants, sweeteners, perfuming agents, flavoring agents, diluents and other known additives to provide an elegant presentation of the drug or aid in the manufacturing of the pharmaceutical product (i.e., medicament).
- an effective dosing regimen will dose at least a minimum amount that reaches the desired biological effect, or biologically effective dose, however, the dose should not be so high as to outweigh the benefit of the biological effect with unacceptable side effects. Therefore, an effective dosing regimen will dose no more than the maximum tolerated dose ("MTD").
- the maximum tolerated dose is defined as the highest dose that produces an acceptable incidence of dose-limiting toxicities (“DLT"). Doses that cause an unacceptable rate of DLT are considered non-tolerated.
- the MTD for a particular schedule is established in phase 1 clinical trials.
- the amount of the agents administered will depend upon the particular agent used, the strain of HBV being treated, the age, weight, and condition of the patient, and the judgment of the clinician, but will generally be between about 0.2 to 2.0 grams per day.
- the kit may comprise a container comprising the combination.
- Suitable containers include, for example, bottles, vials, syringes, blister pack, etc.
- the container may be formed from a variety of materials such as glass or plastic.
- the container may hold the combination which is effective for treating the condition and may have a sterile access port (for example, the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
- the kit may further comprise a label or package-insert on or associated with the container.
- package-insert is used to refer to instructions customarily included in commercial packages of therapeutic agents that contain information about the indications, usage, dosage, administration, contraindications and/or warnings concerning the use of such therapeutic agents.
- the label or package inserts indicates that the therapeutic agents can be used to treat a viral infection, such as Hepatitis B.
- kits are suitable for the delivery of solid oral forms of the therapeutic agents, such as tablets or capsules.
- a kit preferably includes a number of unit dosages.
- Such kits can include a card having the dosages oriented in the order of their intended use.
- An example of such a kit is a "blister pack".
- Blister packs are well known in the packaging industry and are widely used for packaging pharmaceutical unit dosage forms.
- a memory aid can be provided, for example in the form of numbers, letters, or other markings or with a calendar insert, designating the days in the treatment schedule in which the dosages can be administered.
- a kit may comprise (a) a first container with one agent contained therein; and (b) a second container with a second agent contained therein.
- the kit may further comprise a third container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
- BWFI bacteriostatic water for injection
- phosphate-buffered saline such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution.
- BWFI bacteriostatic water for injection
- phosphate-buffered saline such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution.
- BWFI bacteriostatic water for injection
- Ringer's solution such as phosphate-buffered s
- the kit may further comprise directions for the administration of the therapeutic agents.
- the kit may further comprise directions for the simultaneous, sequential or separate administration of the therapeutic agents to a patient in need thereof.
- the kit may comprise a container for containing separate compositions such as a divided bottle or a divided foil packet, however, the separate
- compositions may also be contained within a single, undivided container.
- the kit comprises directions for the administration of the separate therapeutic agents.
- the kit form is particularly advantageous when the separate therapeutic agents are preferably administered in different dosage forms (e.g., oral and parenteral), are administered at different dosage intervals, or when titration of the individual therapeutic agents of the combination is desired by the prescribing physician.
- the ability of a combination of therapeutic agents to treat Hepatitis B may be determined using pharmacological models which are well known to the art.
- siRNA molecules a lipid nanoparticle formulation of a mixture of three siRNAs targeting the HBV genome and that inhibits production of HBV DNA, HBsAg and HBeAg), and pegylated interferon alpha 2a (IFNa2a, an antiviral cytokine that activates innate immunity pathways in hepatocytes, and is used clinically for treatment of chronic hepatitis B
- IFNa2a pegylated interferon alpha 2a
- the lipid nanoparticle formulation comprises a mixture of three siRNAs targeting the HBV genome. Specifically, the following lipid nanoparticle (LNP) formulation was used to deliver the HBV siRNAs in the experiments reported herein. This formulation is referred to as siRNA-NP2 throughout this Example. The values shown in the table are mole percentages.
- the abbreviation DSPC means distearoylphosphatidylcholine.
- the cationic lipid had the following structure:
- a mixture of three siRNAs targeting the HBV genome were used in this experiment.
- the sequences of the three siRNAs are shown below.
- IFNa2a concentration range of 25.0 IU/mL to 0.31 IU/mL in a 3-fold dilution series and 5 point titration
- siRNA-NP2 concentration range of 5.0 ng/mL to 0.06 ng/mL in a 3-fold dilution series and 5 point titration
- IFNa2a or siRNA-NP2 treatments alone or in combination are shown in Tables, 1, 2, and 3 as indicated below.
- the EC50 values of IFNa2a and siRNA-NP2 were determined in an earlier experiment and are shown in Table 4; some variance was observed from different lots of PHH cells.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Virology (AREA)
- Dispersion Chemistry (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Nanotechnology (AREA)
- Communicable Diseases (AREA)
- Dermatology (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention provides therapeutic combinations and therapeutic methods that are useful for treating viral infections such as Hepatitis B virus and/or Hepatitis D virus. In certain embodiments of the invention provide a method for treating hepatitis B in a human, comprising administering to the human: a lipid nanoparticle formulation comprising siRNAs; and an interferon. Certain embodiments of the invention provide a kit comprising a lipid nanoparticle formulation comprising siRNAs; an interferon; and instructions for the simultaneous, sequential or separate administration of the lipid nanoparticle formulation and the interferon.
Description
THERAPEUTIC COMPOSITIONS AND METHODS FOR TREATING HEPATITIS B
CROSS-REFERENCE TO RELATED APPLICATION(S) This patent application claims the benefit of priority of U.S. application serial No.
62/520,981, filed June 16, 2017, which application is herein incorporated by reference.
BACKGROUND
Hepatitis B virus (abbreviated as "HBV") is a member of the Hepadnavirus family. The virus particle (sometimes referred to as a virion) includes an outer lipid envelope and an icosahedral nucleocapsid core composed of protein. The nucleocapsid encloses the viral DNA and a DNA polymerase that has reverse transcriptase activity. The outer envelope contains embedded proteins that are involved in viral binding of, and entry into, susceptible cells, typically liver hepatocytes. In addition to the infectious viral particles, filamentous and spherical bodies lacking a core can be found in the serum of infected individuals. These particles are not infectious and are composed of the lipid and protein that forms part of the surface of the virion, which is called the surface antigen (HBsAg), and is produced in excess during the life cycle of the virus.
The genome of HBV is made of circular DNA, but it is unusual because the DNA is not fully double-stranded. One end of the full length strand is linked to the viral DNA polymerase.
The genome is 3020-3320 nucleotides long (for the full-length strand) and 1700-2800 nucleotides long (for the shorter strand). The negative-sense (non-coding) is complementary to the viral mRNA. The fully double stranded viral DNA (covalently closed circular DNA, or cccDNA) is found in the nucleus soon after infection of the cell. There are four known genes encoded by the genome, called C, X, P, and S. The core protein is coded for by gene C
(HBcAg), and its start codon is preceded by an upstream in-frame AUG start codon from which the pre-core protein is produced. HBeAg is produced by proteolytic processing of the pre-core protein. The DNA polymerase is encoded by gene P. Gene S is the gene that codes for the surface antigen (HBsAg). The HBsAg gene is one long open reading frame but contains three in frame "start" (ATG) codons that divide the gene into three sections, pre-Sl, pre-S2, and S.
Because of the multiple start codons, polypeptides of three different sizes called large, middle, and small are produced. The function of the protein coded for by gene X is not fully understood, but it is required for viral gene expression and is associated with the development of liver cancer. Replication of HBV is a complex process. Although replication takes place in the liver, the virus spreads to the blood where viral proteins and antibodies against them are found in infected people. The structure, replication and biology of HBV is reviewed in D. Glebe and
C.M.Bremer, Seminars in Liver Disease, Vol. 33, No. 2, pages 103-112 (2013).
Infection of humans with HBV can cause an infectious inflammatory illness of the liver. Infected individuals may not exhibit symptoms for many years. It is estimated that about a third of the world population has been infected at one point in their lives, including 350 million who are chronic carriers.
The virus is transmitted by exposure to infectious blood or body fluids. Perinatal infection can also be a major route of infection. The acute illness causes liver inflammation, vomiting, jaundice, and possibly death. Chronic hepatitis B may eventually cause cirrhosis and liver cancer.
Although most people who are infected with HBV clear the infection through the action of their immune system, some infected people suffer an aggressive course of infection
(fulminant hepatitis); while others are chronically infected thereby increasing their chance of liver disease. Several medications are currently approved for treatment of HBV infection, but infected individuals respond with various degrees of success to these medications, and none of these medications clear the virus from the infected person.
Hepatitis D virus (HDV) is a small circular enveloped RNA virus that can propagate only in the presence of the hepatitis B virus (HBV). In particular, HDV requires the HBV surface antigen protein to propagate itself. Infection with both HBV and HDV results in more severe complications compared to infection with HBV alone. These complications include a greater likelihood of experiencing liver failure in acute infections and a rapid progression to liver cirrhosis, with an increased chance of developing liver cancer in chronic infections. In combination with hepatitis B virus, hepatitis D has the highest mortality rate of all the hepatitis infections. The routes of transmission of HDV are similar to those for HBV. Infection is largely restricted to persons at high risk of HBV infection, particularly injecting drug users and persons receiving clotting factor concentrates.
Thus, there is a continuing need for compositions and methods for the treatment of HBV infection humans, as well as for the treatment of HBV/HDV infection in humans.
SUMMARY
The present invention provides therapeutic combinations and therapeutic methods that are useful for treating viral infections such as HBV and/or HDV.
Thus, certain embodiments of the invention provide a method for treating hepatitis B in a human, comprising administering to the human:
1) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
2) an interferon (IFN).
Certain embodiments of the invention also provide a method for treating hepatitis D in a
human, comprising administering to the human:
1) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
2) an interferon (IFN).
Certain embodiments of the invention provide a lipid nanoparticle formulation in combination with an interferon for the prophylactic or therapeutic treatment of hepatitis B, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6).
Certain embodiments of the invention provide the use a lipid nanoparticle formulation in combination with an interferon (IFN) for the preparation of a medicament for treating hepatitis B in a human, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
Certain embodiments of the invention provide a composition comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
(b) an interferon.
Certain embodiments of the invention provide a kit comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6);
(b) an interferon; and
(c) instructions for the simultaneous, sequential or separate administration of the lipid nanoparticle formulation and the interferon.
DETAILED DESCRIPTION
Certain embodiments of the invention provide a method for treating hepatitis B and/or hepatitis D in a human, comprising administering to the human:
1) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
2) an interferon (IFN).
The lipid nanoparticle formulation described herein includes a cocktail of siRNAs (i.e., siRNA 1, siRNA 2 and siRNA3) encapsulated within lipid particles. In certain embodiments, the different siRNA molecules are co-encapsulated in the same lipid particle. In certain embodiments, each type of siRNA species present in the cocktail is encapsulated in its own particle. In certain embodiments, some siRNA species are co-encapsulated in the same particle while other siRNA species are encapsulated in different particles.
In certain embodiments, the lipid nanoparticle formulation further comprises a cationic
lipid and a non-cationic lipid. Accordingly, in certain embodiments, the lipid nanoparticle formulation comprises a population of lipid nanoparticles, wherein each lipid nanoparticle comprises a cationic lipid, a non-cationic lipid and at least one siRNA molecule selected from siRNA 1, siRNA 2 and siRNA 3. In certain embodiments, the lipid nanoparticle formulation comprises a population of lipid nanoparticles, wherein each lipid nanoparticle comprises a cationic lipid, a non-cationic lipid, siRNA 1, siRNA 2 and siRNA 3.
In certain embodiments, the cationic lipid is selected from the group consisting of 1,2- dilinoleyloxy-N,N-dimethylaminopropane (DLinDMA), 1 ,2-dilinolenyloxy-N,N- dimethylaminopropane (DLenDMA), l,2-di-Y-linolenyloxy-N,N-dimethylaminopropane (γ- DLenDMA; Compound (15)) , 3-((6Z,9Z,28Z,31Z)-heptatriaconta-6,9,28,31-tetraen-19-yloxy)- N,N-dimethylpropan-l -amine (DLin-MP-DMA; Compound (8)), (6Z,9Z,28Z,31Z)- heptatriaconta-6,9,28,31-tetraen-19-yl 4-(dimethylamino)butanoate) (Compound (7)), (6Z, 16Z)- 12-((Z)-dec-4-enyl)docosa-6,16-dien-l 1-yl 5-(dimethylamino)pentanoate (Compound (13)), a salt thereof, and a mixture thereof.
In certain embodiments, the non-cationic lipid is cholesterol or a derivative thereof. In certain embodiments, the non-cationic lipid is a phospholipid.
In certain embodiments, the non-cationic lipid is a mixture of a phospholipid and cholesterol or a derivative thereof.
In certain embodiments, the phospholipid is selected from the group consisting of dipalmitoylphosphatidylcholine (DPPC), distearoylphosphatidylcholine (DSPC), and a mixture thereof.
In certain embodiments, the phospholipid is DSPC.
In certain embodiments, the lipid nanoparticle formulation further comprises a conjugated lipid that inhibits aggregation of particles.
In certain embodiments, the conjugated lipid that inhibits aggregation of particles is a polyethyleneglycol (PEG)-lipid conjugate.
In certain embodiments, the PEG-lipid conjugate is selected from the group consisting of a PEG-diacylglycerol (PEG-DAG) conjugate, a PEG-dialkyloxypropyl (PEG-DAA) conjugate, a
PEG-phospholipid conjugate, a PEG-ceramide (PEG-Cer) conjugate, a PEG- dimyristyloxypropyl (PEG-DMA) conjugate and a mixture thereof.
In certain embodiments, the PEG-lipid conjugate is a PEG-C-DMA conjugate.
As described herein, the lipid nanoparticle formulation comprises a population of lipid nanoparticles. In certain embodiments, the cationic lipid generally comprises from about 48 mol % to about 62 mol % of the total lipid present in each particle. In certain embodiments, the lipid nanoparticle formulation comprises a phospholipid and cholesterol or cholesterol derivative, wherein the phospholipid generally comprises from about 7 mol % to about 17 mol % of the total lipid present in each particle and the cholesterol or derivative thereof generally comprises from about 25 mol % to about 40 mol % of the total lipid present in each particle. In certain embodiments, the conjugated lipid that inhibits aggregation of particles generally comprises from about 0.5 mol % to about 3 mol % of the total lipid present in each particle.
Interferons (IFNs) are a group of signaling proteins made and released by host cells in response to the presence of certain pathogens, such as, e.g., viruses, bacteria, and parasites, as well as to the presence of tumor cells. More than twenty distinct IFN genes and proteins have been identified in animals, including humans. Such interferons may be used to practice the claimed invention. In certain embodiments, the IFN is IFN alpha (IFN-a), IFN beta (IFN-β) or IFN lambda (IFN- λ). In certain embodiments, the IFN is pegylated. In certain embodiments, the IFN is IFN-a. In certain embodiments, the IFN-a is pegylated IFN-a2a or peglyated IFN- a2b. In certain embodiments, the IFN is IFN-β. In certain embodiments, the IFN-β is pegylated IFN-beta (e.g., TRK-560; Toray Industries). In certain embodiments, the IFN is IFN lambda (IFN- λ).
In certain embodiments, the lipid nanoparticle formulation is administered via injection. In certain embodiments, the IFN is administered via injection.
In certain embodiments, the lipid nanoparticle formulation and the IFN are administered separately.
In certain embodiments, the lipid nanoparticle formulation and the IFN are administered sequentially.
In certain embodiments, the lipid nanoparticle formulation is administered first and the IFN is administered second.
In certain embodiments, the IFN is administered first and the lipid nanoparticle formulation is administered second.
In certain embodiments, the lipid nanoparticle formulation and the IFN are administered concurrently.
In certain embodiments, the lipid nanoparticle formulation and/or the IFN are
administered two or more times.
In certain embodiments, a pharmaceutical composition comprising the lipid nanoparticle formulation, the IFN and a pharmaceutically acceptable carrier is administered to the human.
In certain embodiments, methods of the invention further comprise administering at least one additional therapeutic agent.
In certain embodiments, the at least one additional therapeutic agent is selected from the group consisting of:
(A) an agent that controls viral replication;
(B) an agent that reduces viral Ags;
(C) an immune enhancer; and
(D) an immune stimulant.
These classes of additional therapeutic agents are further described below as Category I, II and III agents.
Certain embodiments of the invention provide, a lipid nanoparticle formulation in combination with an interferon for the prophylactic or therapeutic treatment of hepatitis B, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6).
As used herein, the term "in combination with" refers to the simultaneous or sequential use of a lipid nanoparticle formulation and an interferon, as well as compositions comprising a lipid nanoparticle formulation and an interferon.
Certain embodiments of the invention provide the use of a lipid nanoparticle formulation in combination with an interferon (IFN) for the preparation of a medicament for treating hepatitis B in a human, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
Certain embodiments of the invention provide a composition comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
(b) an interferon.
In certain embodiments, the composition is a pharmaceutical composition further comprising a pharmaceutically acceptable carrier.
In certain embodiments, the composition further comprises at least one additional therapeutic agent (e.g., a Category I, II and/or III agent discussed herein).
Certain embodiments of the invention provide a kit comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO: 3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6);
(b) an interferon; and
(c) instructions for the simultaneous, sequential or separate administration of the lipid nanoparticle formulation and the interferon.
In certain embodiments, the kit further comprises at least one additional therapeutic agent (e.g., a Category I, II and/or III agent discussed herein).
I. Agents that Control Viral Replication
Category I treatments are directed to the use of agents that control, e.g., inhibit, viral replication.
A. Reverse Transcriptase Inhibitors
In certain embodiments, the reverse transcriptase inhibitor is a nucleoside analog.
In certain embodiments, the reverse transcriptase inhibitor is a nucleoside analog reverse-transcriptase inhibitor (NARTI or NRTI).
In certain embodiments, the reverse transcriptase inhibitor is a nucleotide analog reverse- transcriptase inhibitor (NtARTI or NtRTI).
The term reverse transcriptase inhibitor includes, but is not limited to: entecavir, clevudine, telbivudine, lamivudine, adefovir, and tenofovir, tenofovir disoproxil, tenofovir alafenamide, tenofovir disoproxil fumarate, adefovir dipivoxil, (lR,2R,3R,5R)-3-(6-amino-9H- 9-purinyl)-2-fluoro-5-(hydroxymethyl)-4-methylenecyclopentan-l-ol (described in U.S. Patent No. 8,816,074), emtricitabine, abacavir, elvucitabine, ganciclovir, lobucavir, famciclovir, penciclovir, amdoxovir and CMX157 (tenofovir exalidex).
The term reverse transcriptase inhibitor includes, but is not limited to, entecavir, lamivudine, and (lR,2R,3R,5R)-3-(6-amino-9H-9-purinyl)-2-fluoro-5-(hydroxymethyl)-4- methylenecyclopentan- 1 -ol .
The term reverse transcriptase inhibitor includes, but is not limited to a covalently bound phosphoramidate or phosphonamidate moiety of the above-mentioned reverse transcriptase inhibitors, or as described in, for example, U.S. Patent No. 8,816,074, US 2011/0245484 Al, and US 2008/0286230A1.
The term reverse transcriptase inhibitor includes, but is not limited to, nucleotide analogs that comprise a phosphoramidate moiety, such as, methyl ((((lR,3R,4R,5R)-3-(6-amino-9H- purin-9-yl)-4-fluoro-5-hydroxy-2-methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate and methyl ((((lR,2R,3R,4R)-3-fluoro-2-hydroxy-5-methylene-4-(6-oxo-l,6- dihydro-9H-purin-9-yl)cyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate. Also included are the individual diastereomers thereof, which includes, for example, methyl ((R)- (((1 R, 3R,4R, 5R)-3 -(6-amino-9H-purin-9-yl)-4-fluoro-5 -hydroxy-2-
methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate and methyl ((S)- ((( 1 R, 3R,4R, 5R)-3 -(6-amino-9H-purin-9-yl)-4-fluoro-5 -hydroxy-2- methylenecyclopentyl)methoxy)(phenoxy)phosphoryl)-(D or L)-alaninate.
The term reverse transcriptase inhibitor includes, but is not limited to a phosphonamidate moiety, such as, tenofovir alafenamide, as well as those described in US 2008/0286230 Al . Methods for preparing stereoselective phosphoramidate or phosphonamidate containing actives are described in, for example, U.S. Patent No. 8,816,074, as well as US 2011/0245484 Al and US 2008/0286230 Al .
B. Capsid Inhibitors
As described herein the term "capsid inhibitor" includes compounds that are capable inhibiting the expression and/or function of a capsid protein either directly or indirectly. For example, a capsid inhibitor may include, but is not limited to, any compound that inhibits capsid assembly, induces formation of non-capsid polymers, promotes excess capsid assembly or misdirected capsid assembly, affects capsid stabilization, and/or inhibits encapsidation of RNA. Capsid inhibitors also include any compound that inhibits capsid function in a downstream event(s) within the replication process (e.g., viral DNA synthesis, transport of relaxed circular DNA (rcDNA) into the nucleus, covalently closed circular DNA (cccDNA) formation, virus maturation, budding and/or release, and the like). For example, in certain embodiments, the inhibitor detectably inhibits the expression level or biological activity of the capsid protein as measured, e.g., using an assay described herein. In certain embodiments, the inhibitor inhibits the level of rcDNA and downstream products of viral life cycle by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, at least 90%, at least 95% or at least 99%.
The term capsid inhibitor includes compounds described in International Patent
Applications Publication Numbers WO2013006394, WO2014106019, and WO2014089296, including the following compounds:
The term capsid inhibitor also includes the compounds Bay-41-4109 (see International Patent Application Publication Number WO/2013/144129), AT-61 (see International Patent Application Publication Number WO/1998/33501; and King, RW, et al., Antimicrob Agents Chemother., 1998, 42, 12, 3179-3186), DVR-01 and DVR-23 (see International Patent
Application Publication Number WO 2013/006394; and Campagna, MR, et al., J. of Virology, 2013, 87, 12, 6931, and pharmaceutically acceptable salts thereof:
DVR-01 The term capsid inhibitor also includes the compounds Compound 3, GLS-4, and NVR
3-778.
C. cccDNA Formation Inhibitors
Covalently closed circular DNA (cccDNA) is generated in the cell nucleus from viral rcDNA and serves as the transcription template for viral mRNAs. As described herein, the term "cccDNA formation inhibitor" includes compounds that are capable of inhibiting the formation and/or stability of cccDNA either directly or indirectly. For example, a cccDNA formation inhibitor may include, but is not limited to, any compound that inhibits capsid disassembly, rcDNA entry into the nucleus, and/or the conversion of rcDNA into cccDNA. For example, in certain embodiments, the inhibitor detectably inhibits the formation and/or stability of the cccDNA as measured, e.g., using an assay described herein. In certain embodiments, the inhibitor inhibits the formation and/or stability of cccDNA by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%.
The term cccDNA formation inhibitor includes compounds described in International Patent Application Publication Number WO2013130703, including the following compound:
The term cccDNA formation inhibitor includes, but is not limited to those generally and specifically described in United States Patent Application Publication Number US
2015/0038515 Al . The term cccDNA formation inhibitor includes, but is not limited to, 1- (phenylsulfonyl)-N-(pyridin-4-ylmethyl)-lH-indole-2-carboxamide; 1-Benzenesulfonyl- pyrrolidine-2-carboxylic acid (pyridin-4-ylmethyl)-amide; 2-(2-chloro-N-(2-chloro-5- (trifluoromethyl)phenyl)-4-(trifluoromethyl)phenylsulfonamido)-N-(pyridin-4- ylmethyl)acetamide; 2-(4-chloro-N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N- (pyridin-4-ylmethyl)acetamide; 2-(N-(2-chloro-5-(trifluoromethyl)phenyl)-4- (trifluoromethyl)phenylsulfonamido)-N-(pyridin-4-ylmethyl)acetamide; 2-(N-(2-chloro-5- (trifluoromethyl)phenyl)-4-methoxyphenylsulfonamido)-N-(pyridin-4-ylmethyl)acetamide; 2- (N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N-((l-methylpiperidin-4- yl)methyl)acetamide; 2-(N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N- (piperidin-4-ylmethyl)acetamide; 2-(N-(2-chloro-5-
(trifluoromethyl)phenyl)phenylsulfonamido)-N-(pyridin-4-ylmethyl)propanamide; 2-(N-(2- chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N-(pyridin-3-ylmethyl)acetamide; 2-(N- (2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N-(pyrimidin-5-ylmethyl)acetamide; 2-(N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)-N-(pyrimidin-4- ylmethyl)acetamide; 2-(N-(5-chloro-2-fluorophenyl)phenylsulfonamido)-N-(pyridin-4- ylmethyl)acetamide; 2-[(2-chloro-5-trifluoromethyl-phenyl)-(4-fluoro-benzenesulfonyl)-amino] N-pyridin-4-ylmethyl-acetamide; 2-[(2-chloro-5-trifluoromethyl-phenyl)-(toluene-4-sulfonyl)- amino]-N-pyridin-4-ylmethyl-acetamide; 2-[benzenesulfonyl-(2-bromo-5-trifluoromethyl- phenyl)-amino]-N-pyridin-4-ylmethyl-acetamide; 2-[benzenesulfonyl-(2-chloro-5- trifluoromethyl-phenyl)-amino]-N-(2-methyl-benzothiazol-5-yl)-acetamide; 2-[benzenesulfonyl (2-chloro-5-trifluoromethyl-phenyl)-amino]-N-[4-(4-methyl-piperazin-l-yl)-benzyl]-acetamide; 2-[benzenesulfonyl-(2-chloro-5-trifluoromethyl-phenyl)-amino]-N-[3-(4-methyl-piperazin-l-yl) benzylj-acetamide; 2-[benzenesulfonyl-(2-chloro-5-trifluoromethyl-phenyl)-amino]-N-benzyl- acetamide; 2-[benzenesulfonyl-(2-chloro-5-trifluoromethyl-phenyl)-amino]-N-pyridin-4- ylmethyl-acetamide; 2-[benzenesulfonyl-(2-chloro-5-trifluoromethyl-phenyl)-amino]-N-pyridin 4-ylmethyl-propionamide; 2-[benzenesulfonyl-(2-fluoro-5-trifluoromethyl-phenyl)-amino]-N- pyridin-4-ylmethyl-acetamide; 4 (N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)- N-(pyridin-4-yl- methyl)butanamide; 4-((2-(N-(2-chloro-5-
(trifluoromethyl)phenyl)phenylsulfonamido)-acetamido)-methyl)- 1 , 1 -dimethylpiperidin- 1 -ium chloride; 4-(benzyl-methyl-sulfamoyl)-N-(2-chloro-5-trifluoromethyl-phenyl)-benzamide; 4-
(benzyl -methyl-sulfamoyl)-N-(2 -methyl- lH-indol-5-yl)-benzamide; 4-(benzyl-methyl- sulfamoyl)-N-(2 -methyl- lH-indol-5-yl)-benzamide; 4-(benzyl-methyl-sulfamoyl)-N-(2-methyl-
benzothiazol-5-yl)-benzamide; 4-(benzyl-methyl-sulfamoyl)-N-(2-methyl-benzothiazol-6-yl)- benzamide; 4-(benzyl-methyl-sulfamoyl)-N-(2-methyl-benzothiazol-6-yl)-benzamide; 4- (benzyl-methyl-sulfamoyl)-N-pyridin-4-ylmethyl-benzamide; N-(2-aminoethyl)-2-(N-(2-chloro- 5-(trifluoromethyl)phenyl)phenylsulfonamido)-acetamide; N-(2-chloro-5- (trifluoromethyl)phenyl)-N-(2-(3,4-dihydro-2,6-naphthyridin-2(lH)-yl)-2- oxoethyl)benzenesulfonamide; N-benzothiazol-6-yl-4-(benzyl-methyl-sulfamoyl)-benzamide; N-benzothiazol-6-yl-4-(benzyl-methyl-sulfamoyl)-benzamide; tert-butyl (2-(2-(N-(2-chloro-5- (trifluoromethyl)phenyl)phenylsulfonamido)acetamido)-ethyl)carbamate; and tert-butyl 4-((2- (N-(2-chloro-5-(trifluoromethyl)phenyl)phenylsulfonamido)- acetamido)-methyl)piperidine-l- carboxylate, and optionally, combinations thereof.
D. Entry Inhibitors
Certain embodiments of the invention are directed to the use of agents that are HBV entry inhibitors. Entry inhibitors include Myrcludex-B, NTCP inhibitor small molecules, and FXR agonist EYP001 (see, e.g., Gripon, P., Cannie, I. and Urban, S. Efficient Inhibition of Hepatitis B Virus Infection by Acylated Peptides Derived from the Large Viral Surface Protein. Journal of Virology, 79(3): 1613-1622; Volz, T., Allweiss, L., MBarek, M, Warlich, M, Lohse, A., Pollok, J., Alexandrov, A., Urban, S., Petersen, J., Lutgehetmann, M., Dandri, M. The entry inhibitor Myrcludex-B efficiently blocks intrahepatic virus spreading in humanized mice previously infected with hepatitis B virus. Journal of Hepatology, 58(5): 861-867; Radreau, P., Procherot, M., Vonderscher, J., Lotteau, V., Andre, P. Effect of a novel synthetic FXR agonist EYP001 on hepatitis B virus replication in HepaRG cell line and primary human hepatocytes. AASLD LiverLearning, Abstract 1652, November 16, 2015; WO 2015/036442; WO 00/37077; US2007/0015796). For example, the hepatitis B virus uses its surface lipopeptide pre-Sl for docking to mature liver cells via their sodium/bile acid cotransporter (NTCP) and subsequently entering the cells. Myrcludex B is a synthetic N-acylated pre-Sl that can also dock to NTCP, blocking the virus's entry mechanism.
II. Agents that Reduce Viral Ags
Category II treatments are directed to the use of agents that reduce viral antigens.
A. Oligomeric Nucleotides
The oligomeric nucleotides can be designed to target one or more genes and/or transcripts of the HBV genome. Examples of such siRNA molecules are the siRNA molecules set forth in Example 1.
The term oligomeric nucleotide targeted to the Hepatitis B genome includes Arrowhead- ARC-520 (see United States Patent Number 8,809,293; and Wooddell CI, et al., Molecular Therapy, 2013, 21, 5, 973-985).
The term oligomeric nucleotide targeted to the Hepatitis B genome also includes isolated, double stranded, siRNA molecules, that each include a sense strand and an antisense strand that is hybridized to the sense strand. The siRNA target one or more genes and/or transcripts of the HBV genome.
In another aspect, this term includes siRNA molecules that target GalNAc and REP 2139, REP-2165 (see, e.g., WO 2016/077321, Al-Mathtab et al., PLoS ONE 1 l(6):e0156667. doi: 10.1371/journal.pone.0156667 and Guillot et al., Poster P0556, EASL, 2015).
B . sAg Secretion Inhibitors
As described herein the term "sAg secretion inhibitor" includes compounds that are capable of inhibiting, either directly or indirectly, the secretion of sAg (S, M and/or L surface antigens) bearing subviral particles and/or DNA containing viral particles from HBV-infected cells. For example, in certain embodiments, the inhibitor detectably inhibits the secretion of sAg as measured, e.g., using assays known in the art or described herein, e.g., ELISA assay or by Western Blot. In certain embodiments, the inhibitor inhibits the secretion of sAg by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%. In certain embodiments, the inhibitor reduces serum levels of sAg in a patient by at least 5%, at least 10%, at least 20%, at least 50%, at least 75%, or at least 90%.
The term sAg secretion inhibitor includes compounds described in United States Patent Number 8,921,381, as well as compounds described in United States Patent Application Publication Numbers 2015/0087659 and 2013/0303552. For example, the term includes the compounds PBHBV- le salts thereof:
PBHBV-001 PBHBV-2-15
C. Anti-HBsAg agents
Certain aspects of the invention are directed to the use of anti-HBsAg antibodies, e.g., mAbs. Certain aspects of the invention are directed to the use of hepatitis B immune globulin (HBIG).
III. Agents that Improve Immune Response
Category III treatments are directed to the use of agents that improve the immune response against viral infection. In certain embodiments, at least one 'immune enhancer' agent is used in combination with at least one 'immune stimulant agent' . Such a combination can be used in further combination with at least one agent that controls viral replication and/or at least one agent that reduces the viral antigens.
A. Immune Enhancers
Certain aspects of the invention are directed to the use of agents that act to improve an immune response by reducing or eliminating immune exhaustion, e.g., by using checkpoint inhibitors, thereby enhancing the immune response.
In certain embodiments, an immune enhancer is a PD-L1 inhibitor. PD-L1 inhibitors are a group of agents that act to inhibit the association of the programmed death-ligand 1 (PD-L1) with its receptor, programmed cell death protein 1 (PD-1).
Immune enhancers include the following:
anti-PD-1 mAbs (e.g., Nivolumab, Pembrolizumab;
anti-PD-Ll mAbs (e.g., Atezolizumab, Avelumab);
anti-PD-L2 mAbs;
anti-CTLA4 mAbs (e.g., Ipilimumab);
anti-VISTA mAbs (e.g., JNJ-61610588);
anti-LAG3 mAbs (e.g., BMS-986016);
anti-TEVB mAbs (e.g., TSR-022);
peptidomimetics (e.g., AU P-12); and
small molecule compounds (see, e.g., Zak et al., Oncotarget, 2016, 7(21):30323-35)
B. Immune Stimulants
The term "immune stimulant" includes compounds that are capable of modulating an immune response (e.g., stimulating an innate and/or adaptive immune response (e.g., an adjuvant)). The term immune stimulant includes polyinosinic:polycytidylic acid (poly I:C) and interferons.
The term immune stimulant includes agonists of stimulator of IFN genes (STING) and interleukins. The term also includes HBsAg release inhibitors, TLR-7 agonists (GS-9620, RG- 7795), T-cell and/or B-cell stimulators (GS-4774, OX-40 agonists (BMS 986178), anti-GITR agonists (BMS-986156)), RIG-1 inhibitors (SB-9200), and SMAC-mimetics (Birinapant).
The term also includes the following:
anti-HBV vaccines (Engerix-B, RECOMBIVAX HB, GS-4744, Heplisav-B);
interferons (Pegylated IFN-a2a, Peglyated IFN-a2b, IFN-a, IFN-λ);
RIG-I agonists (SB-9200);
STING agonists (cGAMP, cGAMP bisphosphorothioate, ADU SI 00, and other small molecule compounds);
TLR9 agonists (CYT-009, CpG dinucleotides);
TLR7 agonists (GS-9620);
TLR8 agonists (GS-9688);
TLR3 agonists (Ampligen/poly LC12U);
IL-7 (CYT107); and
IL-2 (aldesleukin).
The term "Hepatitis B virus" (abbreviated as HBV) refers to a virus species of the genus Orthohepadnavirus, which is a part of the Hepadnaviridae family of viruses, and that is capable of causing liver inflammation in humans.
The term "Hepatitis D virus" (abbreviated as HDV) refers to a virus species of the genus
Deltaviridae, which is capable of causing liver inflammation in humans.
As used herein, "treatment" (and grammatical variations thereof such as "treat" or "treating") refers to clinical intervention in an attempt to alter the typical disease course of the individual being treated. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, antibodies of the invention are used to delay development of a disease or to slow the progression of a disease.
The term "small -interfering RNA" or "siRNA" as used herein refers to double stranded
RNA (i.e., duplex RNA) that is capable of reducing or inhibiting the expression of a target gene or sequence (e.g., by mediating the degradation or inhibiting the translation of mRNAs which are complementary to the siRNA sequence) when the siRNA is in the same cell as the target gene or sequence. The siRNA may have substantial or complete identity to the target gene or sequence, or may comprise a region of mismatch (i.e., a mismatch motif). In certain
embodiments, the siRNAs may be about 19-25 (duplex) nucleotides in length, and is preferably about 20-24, 21-22, or 21-23 (duplex) nucleotides in length. siRNA duplexes may comprise 3' overhangs of about 1 to about 4 nucleotides or about 2 to about 3 nucleotides and 5' phosphate termini. Examples of siRNA include, without limitation, a double-stranded polynucleotide
molecule assembled from two separate stranded molecules, wherein one strand is the sense strand and the other is the complementary antisense strand.
Preferably, siRNA are chemically synthesized. siRNA can also be generated by cleavage of longer dsRNA (e.g., dsRNA greater than about 25 nucleotides in length) with the E. coli RNase III or Dicer. These enzymes process the dsRNA into biologically active siRNA (see, e.g., Yang et al, Proc. Natl. Acad. Sci. USA, 99:9942-9947 (2002); Calegari et al, Proc. Natl. Acad. Sci. USA, 99: 14236 (2002); Byrom et al., Ambion TechNotes, 10(l):4-6 (2003); Kawasaki et al, Nucleic Acids Res., 31 :981-987 (2003); Knight et al, Science, 293 :2269-2271 (2001); and Robertson et al, J. Biol. Chem., 243 :82 (1968)). Preferably, dsRNA are at least 50 nucleotides to about 100, 200, 300, 400, or 500 nucleotides in length. A dsRNA may be as long as 1000, 1500, 2000, 5000 nucleotides in length, or longer. The dsRNA can encode for an entire gene transcript or a partial gene transcript. In certain instances, siRNA may be encoded by a plasmid (e.g., transcribed as sequences that automatically fold into duplexes with hairpin loops).
The phrase "inhibiting expression of a target gene" refers to the ability of a siRNA to silence, reduce, or inhibit expression of a target gene (e.g., a gene within the HBV genome). To examine the extent of gene silencing, a test sample (e.g., a biological sample from an organism of interest expressing the target gene or a sample of cells in culture expressing the target gene) is contacted with a siRNA that silences, reduces, or inhibits expression of the target gene.
Expression of the target gene in the test sample is compared to expression of the target gene in a control sample (e.g., a biological sample from an organism of interest expressing the target gene or a sample of cells in culture expressing the target gene) that is not contacted with the siRNA. Control samples (e.g., samples expressing the target gene) may be assigned a value of 100%. In particular embodiments, silencing, inhibition, or reduction of expression of a target gene is achieved when the value of the test sample relative to the control sample (e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.) is about 100%, 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91%, 90%, 89%, 88%, 87%, 86%, 85%, 84%, 83%, 82%, 81%, 80%, 79%, 78%, 77%, 76%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%, 25%, 20%, 15%, 10%, 5%, or 0%. Suitable assays include, without limitation, examination of protein or mRNA levels using techniques known to those of skill in the art, such as, e.g., dot blots, Northern blots, in situ hybridization, ELISA, immunoprecipitation, enzyme function, as well as phenotypic assays known to those of skill in the art. An "effective amount" or "therapeutically effective amount" of a therapeutic nucleic acid such as an siRNA is an amount sufficient to produce the desired effect, e.g., an inhibition of expression of a target sequence in comparison to the normal expression level detected in the absence of a siRNA. In particular embodiments, inhibition of expression of a target gene or target sequence is achieved
when the value obtained with a siRNA relative to the control (e.g., buffer only, an siRNA sequence that targets a different gene, a scrambled siRNA sequence, etc.) is about 100%, 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91%, 90%, 89%, 88%, 87%, 86%, 85%, 84%, 83%, 82%, 81%, 80%, 79%, 78%, 77%, 76%, 75%, 70%, 65%, 60%, 55%, 50%, 45%, 40%, 35%, 30%), 25%), 20%), 15%), 10%, 5%, or 0%. Suitable assays for measuring the expression of a target gene or target sequence include, but are not limited to, examination of protein or mRNA levels using techniques known to those of skill in the art, such as, e.g., dot blots, Northern blots, in situ hybridization, ELISA, immunoprecipitation, enzyme function, as well as phenotypic assays known to those of skill in the art.
The term "nucleic acid" as used herein refers to a polymer containing at least two nucleotides (i.e., deoxyribonucleotides or ribonucleotides) in either single- or double-stranded form and includes DNA and RNA. "Nucleotides" contain a sugar deoxyribose (DNA) or ribose (RNA), a base, and a phosphate group. Nucleotides are linked together through the phosphate groups. "Bases" include purines and pyrimidines, which further include natural compounds adenine, thymine, guanine, cytosine, uracil, inosine, and natural analogs, and synthetic derivatives of purines and pyrimidines, which include, but are not limited to, modifications which place new reactive groups such as, but not limited to, amines, alcohols, thiols,
carboxylates, and alkylhalides. Nucleic acids include nucleic acids containing known nucleotide analogs or modified backbone residues or linkages, which are synthetic, naturally occurring, and non-naturally occurring, and which have similar binding properties as the reference nucleic acid. Examples of such analogs and/or modified residues include, without limitation,
phosphorothioates, phosphorami dates, methyl phosphonates, chiral -methyl phosphonates, 2'-0- methyl ribonucleotides, and peptide-nucleic acids (PNAs). Additionally, nucleic acids can include one or more UNA moieties.
The term "nucleic acid" includes any oligonucleotide or polynucleotide, with fragments containing up to 60 nucleotides generally termed oligonucleotides, and longer fragments termed polynucleotides. A deoxyribooligonucleotide consists of a 5-carbon sugar called deoxyribose joined covalently to phosphate at the 5' and 3' carbons of this sugar to form an alternating, unbranched polymer. DNA may be in the form of, e.g., antisense molecules, plasmid DNA, pre- condensed DNA, a PCR product, vectors, expression cassettes, chimeric sequences,
chromosomal DNA, or derivatives and combinations of these groups. A ribooligonucleotide consists of a similar repeating structure where the 5-carbon sugar is ribose. RNA may be in the form, for example, of small interfering RNA (siRNA), Dicer- substrate dsRNA, small hairpin RNA (shRNA), asymmetrical interfering RNA (aiRNA), microRNA (miRNA), mRNA, tRNA, rRNA, tRNA, viral RNA (vRNA), and combinations thereof. Accordingly, the terms
"polynucleotide" and "oligonucleotide" refer to a polymer or oligomer of nucleotide or nucleoside monomers consisting of naturally-occurring bases, sugars and intersugar (backbone) linkages. The terms "polynucleotide" and "oligonucleotide" also include polymers or oligomers comprising non-naturally occurring monomers, or portions thereof, which function similarly. Such modified or substituted oligonucleotides are often preferred over native forms because of properties such as, for example, enhanced cellular uptake, reduced immunogenicity, and increased stability in the presence of nucleases.
Unless otherwise indicated, a particular nucleic acid sequence also implicitly
encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, S Ps, and complementary sequences as well as the sequence explicitly indicated. Specifically, degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res., 19:5081 (1991); Ohtsuka et al., J. Biol. Chem., 260:2605-2608 (1985); Rossolini et al., Mol. Cell. Probes, 8:91-98 (1994)).
An "isolated" or "purified" DNA molecule or RNA molecule is a DNA molecule or
RNA molecule that exists apart from its native environment. An isolated DNA molecule or RNA molecule may exist in a purified form or may exist in a non-native environment such as, for example, a transgenic host cell. For example, an "isolated" or "purified" nucleic acid molecule or biologically active portion thereof, is substantially free of other cellular material, or culture medium when produced by recombinant techniques, or substantially free of chemical precursors or other chemicals when chemically synthesized. In one embodiment, an "isolated" nucleic acid is free of sequences that naturally flank the nucleic acid (i.e., sequences located at the 5' and 3' ends of the nucleic acid) in the genomic DNA of the organism from which the nucleic acid is derived. For example, in various embodiments, the isolated nucleic acid molecule can contain less than about 5 kb, 4 kb, 3 kb, 2 kb, 1 kb, 0.5 kb, or 0.1 kb of nucleotide sequences that naturally flank the nucleic acid molecule in genomic DNA of the cell from which the nucleic acid is derived.
The term "gene" refers to a nucleic acid (e.g., DNA or RNA) sequence that comprises partial length or entire length coding sequences necessary for the production of a polypeptide or precursor polypeptide.
"Gene product," as used herein, refers to a product of a gene such as an RNA transcript or a polypeptide.
The term "unlocked nucleobase analogue" (abbreviated as "UNA") refers to an acyclic nucleobase in which the C2' and C3' atoms of the ribose ring are not covalently linked. The
term "unlocked nucleobase analogue" includes nucleobase analogues having the following structure identified as Structure A:
Structure A
wherein R is hydroxyl, and Base is any natural or unnatural base such as, for example, adenine (A), cytosine (C), guanine (G) and thymine (T). UNA include the molecules identified as acyclic 2' -3 '-seco-nucleotide monomers in U.S. patent serial number 8,314,227.
The term "lipid" refers to a group of organic compounds that include, but are not limited to, esters of fatty acids and are characterized by being insoluble in water, but soluble in many organic solvents. They are usually divided into at least three classes: (1) "simple lipids," which include fats and oils as well as waxes; (2) "compound lipids," which include phospholipids and glycolipids; and (3) "derived lipids" such as steroids.
The term "lipid particle" includes a lipid formulation that can be used to deliver a therapeutic nucleic acid (e.g., siRNA) to a target site of interest (e.g., cell, tissue, organ, and the like). In preferred embodiments, the lipid particle is typically formed from a cationic lipid, a non-cationic lipid, and optionally a conjugated lipid that prevents aggregation of the particle. A lipid particle that includes a nucleic acid molecule (e.g., siRNA molecule) is referred to as a nucleic acid-lipid particle. Typically, the nucleic acid is fully encapsulated within the lipid particle, thereby protecting the nucleic acid from enzymatic degradation.
In certain instances, nucleic acid-lipid particles are extremely useful for systemic applications, as they can exhibit extended circulation lifetimes following intravenous (i.v.) injection, they can accumulate at distal sites (e.g., sites physically separated from the
administration site), and they can mediate silencing of target gene expression at these distal sites. The nucleic acid may be complexed with a condensing agent and encapsulated within a lipid particle as set forth in PCT Publication No. WO 00/03683, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
The lipid particles typically have a mean diameter of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 1 10 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, or about 30 nm, 35
nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125 nm, 130 nm, 135 nm, 140 nm, 145 nm, or 150 nm, and are substantially non-toxic. In addition, nucleic acids, when present in the lipid particles, are resistant in aqueous solution to degradation with a nuclease. Nucleic acid-lipid particles and their method of preparation are disclosed in, e.g., U.S. Patent Publication Nos. 20040142025 and 20070042031, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
As used herein, "lipid encapsulated" can refer to a lipid particle that provides a therapeutic nucleic acid such as a siRNA, with full encapsulation, partial encapsulation, or both. In a preferred embodiment, the nucleic acid (e.g., siRNA) is fully encapsulated in the lipid particle (e.g., to form a nucleic acid-lipid particle).
The term "lipid conjugate" refers to a conjugated lipid that inhibits aggregation of lipid particles. Such lipid conjugates include, but are not limited to, PEG-lipid conjugates such as, e.g., PEG coupled to dialkyloxypropyls (e.g., PEG-DAA conjugates), PEG coupled to diacylglycerols (e.g., PEG-DAG conjugates), PEG coupled to cholesterol, PEG coupled to phosphatidylethanolamines, and PEG conjugated to ceramides (see, e.g., U.S. Patent No.
5,885,613), cationic PEG lipids, polyoxazoline (POZ)-lipid conjugates (e.g., POZ-DAA conjugates), polyamide oligomers (e.g., ATTA-lipid conjugates), and mixtures thereof.
Additional examples of POZ-lipid conjugates are described in PCT Publication No. WO
2010/006282. PEG or POZ can be conjugated directly to the lipid or may be linked to the lipid via a linker moiety. Any linker moiety suitable for coupling the PEG or the POZ to a lipid can be used including, e.g., non-ester containing linker moieties and ester-containing linker moieties. In certain preferred embodiments, non-ester containing linker moieties, such as amides or carbamates, are used.
The term "amphipathic lipid" refers, in part, to any suitable material wherein the hydrophobic portion of the lipid material orients into a hydrophobic phase, while the hydrophilic portion orients toward the aqueous phase. Hydrophilic characteristics derive from the presence of polar or charged groups such as carbohydrates, phosphate, carboxylic, sulfato, amino, sulfhydryl, nitro, hydroxyl, and other like groups. Hydrophobicity can be conferred by the inclusion of apolar groups that include, but are not limited to, long-chain saturated and unsaturated aliphatic hydrocarbon groups and such groups substituted by one or more aromatic, cycloaliphatic, or heterocyclic group(s). Examples of amphipathic compounds include, but are not limited to, phospholipids, aminolipids, and sphingolipids.
Representative examples of phospholipids include, but are not limited to,
phosphatidylcholine, phosphatidyl ethanolamine, phosphatidylserine, phosphatidylinositol,
phosphatidic acid, palmitoyloleoyl phosphatidylcholine, lysophosphatidylcholine, lysophosphatidylethanolamine, dipalmitoylphosphatidylcholine, dioleoylphosphatidylcholine, distearoylphosphatidylcholine, and dilinoleoylphosphatidylcholine. Other compounds lacking in phosphorus, such as sphingolipid, glycosphingolipid families, diacylglycerols, and β- acyloxyacids, are also within the group designated as amphipathic lipids. Additionally, the amphipathic lipids described above can be mixed with other lipids including triglycerides and sterols.
The term "neutral lipid" refers to any of a number of lipid species that exist either in an uncharged or neutral zwitterionic form at a selected pH. At physiological pH, such lipids include, for example, diacylphosphatidyl choline, diacylphosphatidylethanolamine, ceramide, sphingomyelin, cephalin, cholesterol, cerebrosides, and diacylglycerols.
The term "non-cationic lipid" refers to any amphipathic lipid as well as any other neutral lipid or anionic lipid.
The term "anionic lipid" refers to any lipid that is negatively charged at physiological pH. These lipids include, but are not limited to, phosphatidylglycerols, cardiolipins,
diacylphosphatidylserines, diacylphosphatidic acids, N-dodecanoyl phosphatidylethanolamines, N-succinyl phosphatidylethanolamines, N-glutarylphosphatidylethanolamines,
lysylphosphatidylglycerols, palmitoyloleyolphosphatidylglycerol (POPG), and other anionic modifying groups joined to neutral lipids.
The term "hydrophobic lipid" refers to compounds having apolar groups that include, but are not limited to, long-chain saturated and unsaturated aliphatic hydrocarbon groups and such groups optionally substituted by one or more aromatic, cycloaliphatic, or heterocyclic group(s). Suitable examples include, but are not limited to, diacylglycerol, dialkylglycerol, N- N-dialkylamino, l,2-diacyloxy-3-aminopropane, and l,2-dialkyl-3-aminopropane.
The terms "cationic lipid" and "amino lipid" are used interchangeably herein to include those lipids and salts thereof having one, two, three, or more fatty acid or fatty alkyl chains and a pH-titratable amino head group (e.g., an alkylamino or dialkylamino head group). The cationic lipid is typically protonated (i.e., positively charged) at a pH below the pKa of the cationic lipid and is substantially neutral at a pH above the pKa. The cationic lipids may also be termed titratable cationic lipids. In some embodiments, the cationic lipids comprise: a protonatable tertiary amine (e.g., pH-titratable) head group; C18 alkyl chains, wherein each alkyl chain independently has 0 to 3 (e.g., 0, 1, 2, or 3) double bonds; and ether, ester, or ketal linkages between the head group and alkyl chains. Such cationic lipids include, but are not limited to, DSDMA, DODMA, DLinDMA, DLenDMA, γ-DLenDMA, DLin-K-DMA, DLin-K-C2-DMA (also known as DLin-C2K-DMA, XTC2, and C2K), DLin-K-C3 -DMA, DLin-K-C4-DMA,
DLen-C2K-DMA, y-DLen-C2K-DMA, DLin-M-C2-DMA (also known as MC2), and DLin-M- C3-DMA (also known as MC3).
Administration of a compound as a pharmaceutically acceptable acid or base salt may be appropriate. Examples of pharmaceutically acceptable salts are organic acid addition salts formed with acids which form a physiologically acceptable anion, for example, tosylate, methanesulfonate, acetate, citrate, malonate, tartrate, succinate, benzoate, ascorbate, a- ketoglutarate, and a-glycerophosphate. Suitable inorganic salts may also be formed, including hydrochloride, sulfate, nitrate, bicarbonate, and carbonate salts.
Pharmaceutically acceptable salts may be obtained using standard procedures well known in the art, for example by reacting a sufficiently basic compound such as an amine with a suitable acid affording a physiologically acceptable anion. Alkali metal (for example, sodium, potassium or lithium) or alkaline earth metal (for example calcium) salts of carboxylic acids can also be made.
The term "salts" includes any anionic and cationic complex, such as the complex formed between a cationic lipid and one or more anions. Non-limiting examples of anions include inorganic and organic anions, e.g., hydride, fluoride, chloride, bromide, iodide, oxalate (e.g., hemioxalate), phosphate, phosphonate, hydrogen phosphate, dihydrogen phosphate, oxide, carbonate, bicarbonate, nitrate, nitrite, nitride, bisulfite, sulfide, sulfite, bisulfate, sulfate, thiosulfate, hydrogen sulfate, borate, formate, acetate, benzoate, citrate, tartrate, lactate, acrylate, polyacrylate, fumarate, maleate, itaconate, glycolate, gluconate, malate, mandelate, tiglate, ascorbate, salicylate, polymethacrylate, perchlorate, chlorate, chlorite, hypochlorite, bromate, hypobromite, iodate, an alkyl sulfonate, an aryl sulfonate, arsenate, arsenite, chromate, dichromate, cyanide, cyanate, thiocyanate, hydroxide, peroxide, permanganate, and mixtures thereof. In particular embodiments, the salts of the cationic lipids disclosed herein are crystalline salts.
The term "alkyl" includes a straight chain or branched, noncyclic or cyclic, saturated aliphatic hydrocarbon containing from 1 to 24 carbon atoms. Representative saturated straight chain alkyls include, but are not limited to, methyl, ethyl, ^-propyl, «-butyl, «-pentyl, «-hexyl, and the like, while saturated branched alkyls include, without limitation, isopropyl, sec-butyl, isobutyl, tert-butyl, isopentyl, and the like. Representative saturated cyclic alkyls include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and the like, while unsaturated cyclic alkyls include, without limitation, cyclopentenyl, cyclohexenyl, and the like.
The term "alkenyl" includes an alkyl, as defined above, containing at least one double bond between adjacent carbon atoms. Alkenyls include both cis and trans isomers.
Representative straight chain and branched alkenyls include, but are not limited to, ethyl enyl,
propylenyl, 1-butenyl, 2-butenyl, isobutylenyl, 1-pentenyl, 2-pentenyl, 3 -methyl- 1-butenyl, 2- methyl-2-butenyl, 2,3 -dimethyl -2-butenyl, and the like.
The term "alkynyl" includes any alkyl or alkenyl, as defined above, which additionally contains at least one triple bond between adjacent carbons. Representative straight chain and branched alkynyls include, without limitation, acetyl enyl, propynyl, 1-butynyl, 2-butynyl, 1- pentynyl, 2-pentynyl, 3 -methyl- 1 butynyl, and the like.
The term "acyl" includes any alkyl, alkenyl, or alkynyl wherein the carbon at the point of attachment is substituted with an oxo group, as defined below. The following are non- limiting examples of acyl groups: -C(=0)alkyl, -C(=0)alkenyl, and -C(=0)alkynyl.
The term "heterocycle" includes a 5- to 7-membered monocyclic, or 7- to 10- membered bicyclic, heterocyclic ring which is either saturated, unsaturated, or aromatic, and which contains from 1 or 2 heteroatoms independently selected from nitrogen, oxygen and sulfur, and wherein the nitrogen and sulfur heteroatoms may be optionally oxidized, and the nitrogen heteroatom may be optionally quaternized, including bicyclic rings in which any of the above heterocycles are fused to a benzene ring. The heterocycle may be attached via any heteroatom or carbon atom. Heterocycles include, but are not limited to, heteroaryls as defined below, as well as morpholinyl, pyrrolidinonyl, pyrrolidinyl, piperidinyl, piperizynyl,
hydantoinyl, valerolactamyl, oxiranyl, oxetanyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydropyridinyl, tetrahydroprimidinyl, tetrahydrothiophenyl, tetrahydrothiopyranyl, tetrahydropyrimidinyl, tetrahydrothiophenyl, tetrahydrothiopyranyl, and the like.
The terms "optionally substituted alkyl", "optionally substituted alkenyl", "optionally substituted alkynyl", "optionally substituted acyl", and "optionally substituted heterocycle" mean that, when substituted, at least one hydrogen atom is replaced with a substituent. In the case of an oxo substituent (=0), two hydrogen atoms are replaced. In this regard, substituents include, but are not limited to, oxo, halogen, heterocycle, -CN, -ORx, - RxRy, - RxC(=0)Ry - RxS02Ry, -C(=0)Rx, -C(=0)ORx, -C(=0) RxRy, -SOnRx, and -SOnNRW, wherein n is 0, 1, or 2, Rx and Ry are the same or different and are independently hydrogen, alkyl, or heterocycle, and each of the alkyl and heterocycle substituents may be further substituted with one or more of oxo, halogen, -OH, -CN, alkyl, -ORx, heterocycle, -NR¾y, -NRxC(=0)Ry -NRxS02Ry, - C(=0)Rx, -C(=0)ORx, -C(=0)NR¾y, -SOnRx, and -SOnNRxRy. The term "optionally substituted," when used before a list of substituents, means that each of the substituents in the list may be optionally substituted as described herein.
The term "halogen" includes fluoro, chloro, bromo, and iodo.
The term "fusogenic" refers to the ability of a lipid particle to fuse with the membranes of a cell. The membranes can be either the plasma membrane or membranes surrounding organelles, e.g., endosome, nucleus, etc.
As used herein, the term "aqueous solution" refers to a composition comprising in whole, or in part, water.
As used herein, the term "organic lipid solution" refers to a composition comprising in whole, or in part, an organic solvent having a lipid.
The term "electron dense core", when used to describe a lipid particle, refers to the dark appearance of the interior portion of a lipid particle when visualized using cryo
transmission electron microscopy ("cyroTEM"). Some lipid particles have an electron dense core and lack a lipid bilayer structure. Some lipid particles have an elctron dense core, lack a lipid bilayer structure, and have an inverse Hexagonal or Cubic phase structure. While not wishing to be bound by theory, it is thought that the non-bilayer lipid packing provides a 3- dimensional network of lipid cylinders with water and nucleic acid on the inside, i.e., essentially a lipid droplet interpenetrated with aqueous channels containing the nucleic acid.
"Distal site," as used herein, refers to a physically separated site, which is not limited to an adjacent capillary bed, but includes sites broadly distributed throughout an organism.
"Serum-stable" in relation to nucleic acid-lipid particles means that the particle is not significantly degraded after exposure to a serum or nuclease assay that would significantly degrade free DNA or RNA. Suitable assays include, for example, a standard serum assay, a DNAse assay, or an RNAse assay.
"Systemic delivery," as used herein, refers to delivery of lipid particles that leads to a broad biodistribution of an active agent such as a siRNA within an organism. Some techniques of administration can lead to the systemic delivery of certain agents, but not others. Systemic delivery means that a useful, preferably therapeutic, amount of an agent is exposed to most parts of the body. To obtain broad biodistribution generally requires a blood lifetime such that the agent is not rapidly degraded or cleared (such as by first pass organs (liver, lung, etc.) or by rapid, nonspecific cell binding) before reaching a disease site distal to the site of administration. Systemic delivery of lipid particles can be by any means known in the art including, for example, intravenous, subcutaneous, and intraperitoneal. In a preferred embodiment, systemic delivery of lipid particles is by intravenous delivery.
"Local delivery," as used herein, refers to delivery of an active agent such as a siRNA directly to a target site within an organism. For example, an agent can be locally delivered by direct injection into a disease site, other target site, or a target organ such as the liver, heart, pancreas, kidney, and the like.
The term "virus particle load", as used herein, refers to a measure of the number of virus particles (e.g., HBV and/or HDV) present in a bodily fluid, such as blood. For example, particle load may be expressed as the number of virus particles per milliliter of, e.g., blood. Particle load testing may be performed using nucleic acid amplification based tests, as well as non-nucleic acid-based tests (see, e.g., Puren et al., The Journal of Infectious Diseases, 201 :S27- 36 (2010)).
The oligonucleotides (such as the sense and antisense RNA strands set forth in the Example) specifically hybridize to or are complementary to a target polynucleotide sequence. The terms "specifically hybridizable" and "complementary" as used herein indicate a sufficient degree of complementarity such that stable and specific binding occurs between the DNA or RNA target and the oligonucleotide. It is understood that an oligonucleotide need not be 100% complementary to its target nucleic acid sequence to be specifically hybridizable. In preferred embodiments, an oligonucleotide is specifically hybridizable when binding of the
oligonucleotide to the target sequence interferes with the normal function of the target sequence to cause a loss of utility or expression therefrom, and there is a sufficient degree of
complementarity to avoid non-specific binding of the oligonucleotide to non-target sequences under conditions in which specific binding is desired, i.e., under physiological conditions in the case of in vivo assays or therapeutic treatment, or, in the case of in vitro assays, under conditions in which the assays are conducted. Thus, the oligonucleotide may include 1, 2, 3, or more base substitutions as compared to the region of a gene or mRNA sequence that it is targeting or to which it specifically hybridizes.
Generating siRNA Molecules
siRNA can be provided in several forms including, e.g., as one or more isolated small- interfering RNA (siRNA) duplexes, as longer double-stranded RNA (dsRNA), or as siRNA or dsRNA transcribed from a transcriptional cassette in a DNA plasmid. In some embodiments, siRNA may be produced enzymatically or by partial/total organic synthesis, and modified ribonucleotides can be introduced by in vitro enzymatic or organic synthesis. In certain instances, each strand is prepared chemically. Methods of synthesizing RNA molecules are known in the art, e.g., the chemical synthesis methods as described in Verma and Eckstein (1998) or as described herein.
Methods for isolating RNA, synthesizing RNA, hybridizing nucleic acids, making and screening cDNA libraries, and performing PCR are well known in the art (see, e.g., Gubler and Hoffman, Gene, 25:263-269 (1983); Sambrook et al, supra; Ausubel et al, supra), as are PCR methods (see, U.S. Patent Nos. 4,683, 195 and 4,683,202; PCR Protocols: A Guide toMethods
and Applications (Innis et al., eds, 1990)). Expression libraries are also well known to those of skill in the art. Additional basic texts disclosing the general methods include Sambrook et al., Molecular Cloning, A Laboratory Manual (2nd ed. 1989); Kriegler, Gene Transfer and
Expression: A Laboratory Manual (1990); and Current Protocols in Molecular Biology
(Ausubel et al, eds., 1994). The disclosures of these references are herein incorporated by reference in their entirety for all purposes.
Typically, siRNA are chemically synthesized. The oligonucleotides that comprise the siRNA molecules can be synthesized using any of a variety of techniques known in the art, such as those described in Usman et al, J. Am. Chem. Soc, 109:7845 (1987); Scaringe et al, Nucl. Acids Res., 18:5433 (1990); Wincott et al, Nucl. Acids Res., 23 :2677-2684 (1995); and Wincott et al., Methods Mol. Bio., 74:59 (1997). The synthesis of oligonucleotides makes use of common nucleic acid protecting and coupling groups, such as dimethoxytrityl at the 5 '-end and phosphoramidites at the 3 '-end. As a non-limiting example, small scale syntheses can be conducted on an Applied Biosystems synthesizer using a 0.2 μιηοΐ scale protocol. Alternatively, syntheses at the 0.2 μιηοΐ scale can be performed on a 96-well plate synthesizer from Protogene (Palo Alto, CA). However, a larger or smaller scale of synthesis is also within the scope.
Suitable reagents for oligonucleotide synthesis, methods for RNA deprotection, and methods for RNA purification are known to those of skill in the art.
siRNA molecules can be assembled from two distinct oligonucleotides, wherein one oligonucleotide comprises the sense strand and the other comprises the antisense strand of the siRNA. For example, each strand can be synthesized separately and joined together by hybridization or ligation following synthesis and/or deprotection.
Carrier Systems Containing Therapeutic Nucleic Acids
Lipid Particles
Lipid particles may be used to deliver siRNA molecules described herein (e.g., a combination comprising the three siRNA molecules described in Example 1). Lipid particles may comprise one or more siRNA {e.g., the three siRNA molecules described in Example 1), a cationic lipid, a non-cationic lipid, and a conjugated lipid that inhibits aggregation of particles. In some embodiments, the siRNA molecule(s) is fully encapsulated within the lipid portion of the lipid particle such that the siRNA molecule(s) in the lipid particle is resistant in aqueous solution to nuclease degradation. In other embodiments, the lipid particles described herein are substantially non-toxic to humans. The lipid particles typically have a mean diameter of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 110 nm, or from about
70 to about 90 nm. In certain embodiments, the lipid particles have a median diameter of from about 30 nm to about 150 nm. The lipid particles also typically have a lipid:nucleic acid ratio (e.g., a lipid: siRNA ratio) (mass/mass ratio) of from about 1 : 1 to about 100: 1, from about 1 : 1 to about 50: 1, from about 2: 1 to about 25 : 1, from about 3 : 1 to about 20: 1, from about 5 : 1 to about 15 : 1, or from about 5 : 1 to about 10: 1. In certain embodiments, the nucleic acid-lipid particle has a lipid: siRNA mass ratio of from about 5 : 1 to about 15 : 1.
The lipid particles include serum-stable nucleic acid-lipid particles which comprise one or more siRNA molecules (e.g., a combination comprising the three siRNA molecules described in Example 1), a cationic lipid (e.g., one or more cationic lipids of Formula I-III or salts thereof as set forth herein), a non-cationic lipid (e.g., mixtures of one or more phospholipids and cholesterol), and a conjugated lipid that inhibits aggregation of the particles (e.g., one or more PEG-lipid conjugates). The lipid particle may comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more siRNA molecules (e.g., a combination comprising the three siRNA molecules described in Example 1) that target one or more of the genes described herein. Nucleic acid-lipid particles and their method of preparation are described in, e.g., U.S. Patent Nos. 5,753,613; 5,785,992; 5,705,385; 5,976,567; 5,981,501 ; 6, 1 10,745; and 6,320,017; and PCT Publication No. WO 96/40964, the disclosures of which are each herein incorporated by reference in their entirety for all purposes.
In the nucleic acid-lipid particles, the one or more siRNA molecules (e.g., a
combination comprising the three siRNA molecules described in Example 1) may be fully encapsulated within the lipid portion of the particle, thereby protecting the siRNA from nuclease degradation. In certain instances, the siRNA in the nucleic acid-lipid particle is not substantially degraded after exposure of the particle to a nuclease at 37°C for at least about 20, 30, 45, or 60 minutes. In certain other instances, the siRNA in the nucleic acid-lipid particle is not substantially degraded after incubation of the particle in serum at 37°C for at least about 30, 45, or 60 minutes or at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32,
34, or 36 hours. In other embodiments, the siRNA is complexed with the lipid portion of the particle. One of the benefits of the formulations is that the nucleic acid-lipid particle
compositions are substantially non-toxic to humans.
The term "fully encapsulated" indicates that the siRNA (e.g., a combination
comprising the three siRNA molecules described in Example 1) in the nucleic acid-lipid particle is not significantly degraded after exposure to serum or a nuclease assay that would significantly degrade free DNA or RNA. In a fully encapsulated system, preferably less than about 25% of the siRNA in the particle is degraded in a treatment that would normally degrade 100% of free siRNA, more preferably less than about 10%, and most preferably less than about 5% of the
siRNA in the particle is degraded. "Fully encapsulated" also indicates that the nucleic acid-lipid particles are serum-stable, that is, that they do not rapidly decompose into their component parts upon in vivo administration.
In the context of nucleic acids, full encapsulation may be determined by performing a membrane-impermeable fluorescent dye exclusion assay, which uses a dye that has enhanced fluorescence when associated with nucleic acid. Specific dyes such as OliGreen® and
RiboGreen® (Invitrogen Corp.; Carlsbad, CA) are available for the quantitative determination of plasmid DNA, single-stranded deoxyribonucleotides, and/or single- or double-stranded ribonucleotides. Encapsulation is determined by adding the dye to a liposomal formulation, measuring the resulting fluorescence, and comparing it to the fluorescence observed upon addition of a small amount of nonionic detergent. Detergent-mediated disruption of the liposomal bilayer releases the encapsulated nucleic acid, allowing it to interact with the membrane-impermeable dye. Nucleic acid encapsulation may be calculated as E = (I0 - I)/L, where / and I0 refer to the fluorescence intensities before and after the addition of detergent (see, Wheeler et al, Gene Ther., 6:271-281 (1999)).
In some instances, the nucleic acid-lipid particle composition comprises a siRNA molecule that is fully encapsulated within the lipid portion of the particles, such that from about 30% to about 100%, from about 40% to about 100%, from about 50% to about 100%, from about 60% to about 100%, from about 70% to about 100%, from about 80% to about 100%, from about 90% to about 100%, from about 30% to about 95%, from about 40% to about 95%, from about 50% to about 95%, from about 60%> to about 95%, from about 70% to about 95%, from about 80% to about 95%, from about 85% to about 95%, from about 90% to about 95%, from about 30% to about 90%, from about 40% to about 90%, from about 50% to about 90%, from about 60% to about 90%, from about 70% to about 90%, from about 80% to about 90%, or at least about 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% (or any fraction thereof or range therein) of the particles have the siRNA encapsulated therein.
In other instances, the nucleic acid-lipid particle composition comprises siRNA that is fully encapsulated within the lipid portion of the particles, such that from about 30% to about 100%, from about 40% to about 100%, from about 50% to about 100%, from about 60% to about 100%, from about 70% to about 100%, from about 80% to about 100%, from about 90% to about 100%, from about 30% to about 95%, from about 40% to about 95%, from about 50% to about 95%), from about 60% to about 95%, from about 70% to about 95%, from about 80% to about 95%, from about 85% to about 95%, from about 90% to about 95%, from about 30% to about 90%, from about 40% to about 90%, from about 50% to about 90%, from about 60% to
about 90%, from about 70% to about 90%, from about 80% to about 90%, or at least about 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%., 96%., 97%., 98%., or 99% (or any fraction thereof or range therein) of the input siRNA is encapsulated in the particles.
Depending on the intended use of the lipid particles, the proportions of the components can be varied and the delivery efficiency of a particular formulation can be measured using, e.g., an endosomal release parameter (ERP) assay.
Cationic Lipids
Any of a variety of cationic lipids or salts thereof may be used in the lipid particles either alone or in combination with one or more other cationic lipid species or non-cationic lipid species. The cationic lipids include the (R) and/or (S) enantiomers thereof.
In one aspect, the cationic lipid is a dialkyl lipid. For example, dialkyl lipids may include lipids that comprise two saturated or unsaturated alkyl chains, wherein each of the alkyl chains may be substituted or unsubstituted. In certain embodiments, each of the two alkyl chains comprise at least, e.g., 8 carbon atoms, 10 carbon atoms, 12 carbon atoms, 14 carbon atoms, 16 carbon atoms, 18 carbon atoms, 20 carbon atoms, 22 carbon atoms or 24 carbon atoms.
In one aspect, the cationic lipid is a trialkyl lipid. For example, trialkyl lipids may include lipids that comprise three saturated or unsaturated alkyl chains, wherein each of the alkyl chains may be substituted or unsubstituted. In certain embodiments, each of the three alkyl chains comprise at least, e.g., 8 carbon atoms, 10 carbon atoms, 12 carbon atoms, 14 carbon atoms, 16 carbon atoms, 18 carbon atoms, 20 carbon atoms, 22 carbon atoms or 24 carbon atoms.
In one aspect, cationic li ids of Formula I having the following structure are useful:
or salts thereof, wherein:
R1 and R2 are either the same or different and are independently hydrogen (H) or an optionally substituted Ci-C6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R1 and R2 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms selected from the group consisting of nitrogen (N), oxygen (O), and mixtures thereof;
R3 is either absent or is hydrogen (H) or a Ci-C6 alkyl to provide a quaternary amine;
R4 and R5 are either the same or different and are independently an optionally substituted C10-C24 alkyl, C10-C24 alkenyl, C10-C24 alkynyl, or C10-C24 acyl, wherein at least one of R4 and R5 comprises at least two sites of unsaturation; and
n is 0, 1, 2, 3, or 4.
In some embodiments, R1 and R2 are independently an optionally substituted C1-C4 alkyl, C2-C4 alkenyl, or C2-C4 alkynyl. In one preferred embodiment, R1 and R2 are both methyl groups. In other preferred embodiments, n is 1 or 2. In other embodiments, R3 is absent when the pH is above the pKa of the cationic lipid and R3 is hydrogen when the pH is below the pKa of the cationic lipid such that the amino head group is protonated. In an alternative embodiment, R3 is an optionally substituted C1-C4 alkyl to provide a quaternary amine. In further
embodiments, R4 and R5 are independently an optionally substituted C12-C20 or C14-C22 alkyl, C12-C20 or C14-C22 alkenyl, C12-C20 or C14-C22 alkynyl, or C12-C20 or C14-C22 acyl, wherein at least one of R4 and R5 comprises at least two sites of unsaturation.
In certain embodiments, R4 and R5 are independently selected from the group consisting of a dodecadienyl moiety, a tetradecadienyl moiety, a hexadecadienyl moiety, an octadecadienyl moiety, an icosadienyl moiety, a dodecatrienyl moiety, a tetradectrienyl moiety, a hexadecatrienyl moiety, an octadecatrienyl moiety, an icosatrienyl moiety, an arachidonyl moiety, and a docosahexaenoyl moiety, as well as acyl derivatives thereof (e.g., linoleoyl, linolenoyl, γ-linolenoyl, etc.). In some instances, one of R4 and R5 comprises a branched alkyl group (e.g., a phytanyl moiety) or an acyl derivative thereof (e.g., a phytanoyl moiety). In certain instances, the octadecadienyl moiety is a linoleyl moiety. In certain other instances, the octadecatrienyl moiety is a linolenyl moiety or a γ-linolenyl moiety. In certain embodiments, R4 and R5 are both linoleyl moieties, linolenyl moieties, or γ-linolenyl moieties. In particular embodiments, the cationic lipid of Formula I is l,2-dilinoleyloxy-N,N-dimethylaminopropane (DLinDMA), l,2-dilinolenyloxy-N,N-dimethylaminopropane (DLenDMA), 1,2-dilinoleyloxy- (N,N-dimethyl)-butyl-4-amine (C2-DLinDMA), 1 ,2-dilinoleoyloxy-(N,N-dimethyl)-butyl-4- amine (C2-DLinDAP), or mixtures thereof.
In some embodiments, the cationic lipid of Formula I forms a salt (preferably a crystalline salt) with one or more anions. In one particular embodiment, the cationic lipid of Formula I is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
The synthesis of cationic lipids such as DLinDMA and DLenDMA, as well as additional cationic lipids, is described in U.S. Patent Publication No. 20060083780, the disclosure of which is herein incorporated by reference in its entirety for all purposes. The synthesis of cationic lipids such as C2-DLinDMA and C2-DLinDAP, as well as additional
cationic lipids, is described in international patent application number WO201 1/000106 the disclosure of which is herein incorporated by reference in its entirety for all purposes.
In another aspect, cationic lipids of Formula II having the following structure (or salts thereof) are useful:
wherein R1 and R2 are either the same or different and are independently an optionally substituted C12-C24 alkyl, C12-C24 alkenyl, C12-C24 alkynyl, or C12-C24 acyl; R3 and R4 are either the same or different and are independently an optionally substituted Ci-C6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R3 and R4 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms chosen from nitrogen and oxygen; R5 is either absent or is hydrogen (H) or a Ci-C6 alkyl to provide a quaternary amine; m, n, and p are either the same or different and are independently either 0, 1, or 2, with the proviso that m, n, and p are not simultaneously 0; q is 0, 1, 2, 3, or 4; and Y and Z are either the same or different and are independently O, S, or NH. In a preferred embodiment, q is 2.
In some embodiments, the cationic lipid of Formula II is 2,2-dilinoleyl-4-(2- dimethylaminoethyl)-[l,3]-dioxolane (DLin-K-C2-DMA; "XTC2" or "C2K"), 2,2-dilinoleyl-4- (3-dimethylaminopropyl)-[l,3]-dioxolane (DLin-K-C3 -DMA; "C3K"), 2,2-dilinoleyl-4-(4- dimethylaminobutyl)-[l,3]-dioxolane (DLin-K-C4-DMA; "C4K"), 2,2-dilinoleyl-5- dimethylaminomethyl-[l,3]-dioxane (DLin-K6-DMA), 2,2-dilinoleyl-4-N-methylpepiazino- [l,3]-dioxolane (DLin-K-MPZ), 2,2-dilinoleyl-4-dimethylaminomethyl-[l,3]-dioxolane (DLin- K-DMA), 2,2-dioleoyl-4-dimethylaminomethyl-[l,3]-dioxolane (DO-K-DMA), 2,2-distearoyl- 4-dimethylaminomethyl-[l,3]-dioxolane (DS-K-DMA), 2,2-dilinoleyl-4-N-morpholino-[l,3]- dioxolane (DLin-K-MA), 2,2-Dilinoleyl-4-trimethylamino-[l,3]-dioxolane chloride (DLin-K- TMA.C1), 2,2-dilinoleyl-4,5-bis(dimethylaminomethyl)-[l,3]-dioxolane (DLin-K2-DMA), 2,2- dilinoleyl-4-methylpiperzine-[l,3]-dioxolane (D-Lin-K-N-methylpiperzine), or mixtures thereof. In one embodiment the cationic lipid of Formula II is DLin-K-C2-DMA.
In some embodiments, the cationic lipid of Formula II forms a salt (preferably a crystalline salt) with one or more anions. In one particular embodiment, the cationic lipid of Formula II is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
The synthesis of cationic lipids such as DLin-K-DMA, as well as additional cationic lipids, is described in PCT Publication No. WO 09/086558, the disclosure of which is herein
incorporated by reference in its entirety for all purposes. The synthesis of cationic lipids such as DLin-K-C2-DMA, DLin-K-C3-DMA, DLin-K-C4-DMA, DLin-K6-DMA, DLin-K-MPZ, DO- K-DMA, DS-K-DMA, DLin-K-MA, DLin-K-TMA.Cl, DLin-K2-DMA, and D-Lin-K-N- methylpiperzine, as well as additional cationic lipids, is described in PCT Application No.
PCT/US2009/060251, entitled "Improved Amino Lipids and Methods for the Delivery of Nucleic Acids," filed October 9, 2009, the disclosure of which is incorporated herein by reference in its entirety for all purposes.
In a further aspect, cationic lipids of Formula III having the following structure are useful:
(III)
or salts thereof, wherein: R1 and R2 are either the same or different and are independently an optionally substituted Ci-C6 alkyl, C2-C6 alkenyl, or C2-C6 alkynyl, or R1 and R2 may join to form an optionally substituted heterocyclic ring of 4 to 6 carbon atoms and 1 or 2 heteroatoms selected from the group consisting of nitrogen (N), oxygen (O), and mixtures thereof; R3 is either absent or is hydrogen (H) or a Ci-C6 alkyl to provide a quaternary amine; R4 and R5 are either absent or present and when present are either the same or different and are independently an optionally substituted C1-C10 alkyl or C2-C10 alkenyl; and n is 0, 1, 2, 3, or 4.
In some embodiments, R1 and R2 are independently an optionally substituted C1-C4 alkyl, C2-C4 alkenyl, or C2-C4 alkynyl. In a preferred embodiment, R1 and R2 are both methyl groups. In another preferred embodiment, R4 and R5 are both butyl groups. In yet another preferred embodiment, n is 1. In other embodiments, R3 is absent when the pH is above the pKa of the cationic lipid and R3 is hydrogen when the pH is below the pKa of the cationic lipid such that the amino head group is protonated. In an alternative embodiment, R3 is an optionally substituted C1-C4 alkyl to provide a quaternary amine. In further embodiments, R4 and R5 are independently an optionally substituted C2-C6 or C2-C4 alkyl or C2-C6 or C2-C4 alkenyl.
In an alternative embodiment, the cationic lipid of Formula III comprises ester linkages between the amino head group and one or both of the alkyl chains. In some embodiments, the cationic lipid of Formula III forms a salt (preferably a crystalline salt) with one or more anions. In one particular embodiment, the cationic lipid of Formula III is the oxalate (e.g., hemioxalate) salt thereof, which is preferably a crystalline salt.
Although each of the alkyl chains in Formula III contains cis double bonds at positions 6, 9, and 12 (i.e., cis,cis,cis-A6, 9, u), in an alternative embodiment, one, two, or three of these double bonds in one or both alkyl chains may be in the trans configuration.
γ-DLenDMA (15)
The synthesis of cationic lipids such as γ-DLenDMA (15), as well as additional cationic lipids, is described in U.S. Provisional Application No. 61/222,462, entitled "Improved Cationic Lipids and Methods for the Delivery of Nucleic Acids," filed July 1, 2009, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
The synthesis of cationic lipids such as DLin-M-C3 -DMA ("MC3"), as well as additional cationic lipids (e.g., certain analogs of MC3), is described in U.S. Provisional Application No. 61/185,800, entitled "Novel Lipids and Compositions for the Delivery of
Therapeutics," filed June 10, 2009, and U.S. Provisional Application No. 61/287,995, entitled "Methods and Compositions for Delivery of Nucleic Acids," filed December 18, 2009, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
Examples of other cationic lipids or salts thereof which may be included in the lipid particles include, but are not limited to, cationic lipids such as those described in
WO2011/000106, the disclosure of which is herein incorporated by reference in its entirety for all purposes, as well as cationic lipids such as N,N-dioleyl-N,N-dimethylammonium chloride (DODAC), l,2-dioleyloxy-N,N-dimethylaminopropane (DODMA), l,2-distearyloxy-N,N- dimethylaminopropane (DSDMA), N-(l -(2,3-dioleyloxy)propyl)-N,N,N-trimethylammonium chloride (DOTMA), N,N-distearyl-N,N-dimethylammonium bromide (DDAB), N-(l-(2,3- dioleoyloxy)propyl)-N,N,N-trimethylammonium chloride (DOTAP), 3 -(Ν-(Ν',Ν'- dimethylaminoethane)-carbamoyl)cholesterol (DC-Choi), N-(l,2-dimyristyloxyprop-3-yl)-N,N- dimethyl-N-hydroxy ethyl ammonium bromide (DMRIE), 2,3-dioleyloxy-N-[2(spermine- carboxamido)ethyl]-N,N-dimethyl-l-propanaminiumtrifluoroacetate (DOSPA),
dioctadecylamidoglycyl spermine (DOGS), 3-dimethylamino-2-(cholest-5-en-3-beta-oxybutan- 4-oxy)-l-(cis,cis-9, 12-octadecadienoxy)propane (CLinDMA), 2-[5'-(cholest-5-en-3-beta-oxy)- 3 ' -oxapentoxy)-3 -dimethy- 1 -(cis,cis-9 ' , 1 -2' -octadecadienoxy)propane (CpLinDMA), N,N- dimethyl-3,4-dioleyloxybenzylamine (DMOBA), l,2-N,N'-dioleylcarbamyl-3- dimethylaminopropane (DOcarbDAP), l,2-N,N'-dilinoleylcarbamyl-3-dimethylaminopropane (DLincarbDAP), 1, 2-dilinoleylcarbamoyloxy-3 -dimethylaminopropane (DLin-C-DAP), 1,2-
dilinoleyoxy-3-(dimethylamino)acetoxypropane (DLin-DAC), l,2-dilinoleyoxy-3- morpholinopropane (DLin-MA), l,2-dilinoleoyl-3-dimethylaminopropane (DLinDAP), 1,2- dilinoleylthio-3-dimethylaminopropane (DLin-S-DMA), l-linoleoyl-2-linoleyloxy-3- dimethylaminopropane (DLin-2-DMAP), l,2-dilinoleyloxy-3-trimethylaminopropane chloride salt (DLin-TMA.Cl), l,2-dilinoleoyl-3-trimethylaminopropane chloride salt (DLin-TAP.Cl), l,2-dilinoleyloxy-3-(N-methylpiperazino)propane (DLin-MPZ), 3-(N,N-dilinoleylamino)-l,2- propanediol (DLinAP), 3-(N,N-dioleylamino)-l,2-propanedio (DOAP), l,2-dilinoleyloxo-3-(2- N,N-dimethylamino)ethoxypropane (DLin-EG-DMA), l,2-dioeylcarbamoyloxy-3- dimethylaminopropane (DO-C-DAP), l,2-dimyristoleoyl-3-dimethylaminopropane (DMDAP), l,2-dioleoyl-3-trimethylaminopropane chloride (DOTAP.Cl), dilinoleylmethyl-3- dimethylaminopropionate (DLin-M-C2-DMA; also known as DLin-M-K-DMA or DLin-M- DMA), and mixtures thereof. Additional cationic lipids or salts thereof which may be included in the lipid particles are described in U.S. Patent Publication No. 20090023673, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
The synthesis of cationic lipids such as CLinDMA, as well as additional cationic lipids, is described in U.S. Patent Publication No. 20060240554, the disclosure of which is herein incorporated by reference in its entirety for all purposes. The synthesis of cationic lipids such as DLin-C-DAP, DLinDAC, DLinMA, DLinDAP, DLin-S-DMA, DLin-2-DMAP, DLinTMA.Cl, DLinTAP.Cl, DLinMPZ, DLinAP, DOAP, and DLin-EG-DMA, as well as additional cationic lipids, is described in PCT Publication No. WO 09/086558, the disclosure of which is herein incorporated by reference in its entirety for all purposes. The synthesis of cationic lipids such as DO-C-DAP, DMDAP, DOTAP.Cl, DLin-M-C2-DMA, as well as additional cationic lipids, is described in PCT Application No. PCT/US2009/060251, entitled "Improved Amino Lipids and Methods for the Delivery of Nucleic Acids," filed October 9, 2009, the disclosure of which is incorporated herein by reference in its entirety for all purposes. The synthesis of a number of other cationic lipids and related analogs has been described in U.S. Patent Nos. 5,208,036;
5,264,618; 5,279,833; 5,283,185; 5,753,613; and 5,785,992; and PCT Publication No. WO 96/10390, the disclosures of which are each herein incorporated by reference in their entirety for all purposes. Additionally, a number of commercial preparations of cationic lipids can be used, such as, e.g., LIPOFECTIN® (including DOTMA and DOPE, available from Invitrogen);
LIPOFECTAMINE® (including DOSPA and DOPE, available from Invitrogen); and
TRANSFECTAM® (including DOGS, available from Promega Corp.).
In some embodiments, the cationic lipid comprises from about 50 mol % to about 90 mol %, from about 50 mol % to about 85 mol %, from about 50 mol % to about 80 mol %, from about 50 mol % to about 75 mol %, from about 50 mol % to about 70 mol %, from about
50 mol % to about 65 mol %, from about 50 mol % to about 60 mol %, from about 55 mol % to about 65 mol %, or from about 55 mol % to about 70 mol % (or any fraction thereof or range therein) of the total lipid present in the particle. In particular embodiments, the cationic lipid comprises about 50 mol %, 51 mol %, 52 mol %, 53 mol %, 54 mol %, 55 mol %, 56 mol %, 57 mol %, 58 mol %, 59 mol %, 60 mol %, 61 mol %, 62 mol %, 63 mol %, 64 mol %, or 65 mol % (or any fraction thereof) of the total lipid present in the particle.
In other embodiments, the cationic lipid comprises from about 2 mol % to about 60 mol %, from about 5 mol % to about 50 mol %, from about 10 mol % to about 50 mol %, from about 20 mol % to about 50 mol %, from about 20 mol % to about 40 mol %, from about 30 mol % to about 40 mol %, or about 40 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
Additional percentages and ranges of cationic lipids suitable for use in the lipid particles are described in PCT Publication No. WO 09/127060, U.S. Published Application No. US 2011/0071208, PCT Publication No. WO2011/000106, and U.S. Published Application No. US 2011/0076335, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
It should be understood that the percentage of cationic lipid present in the lipid particles is a target amount, and that the actual amount of cationic lipid present in the formulation may vary, for example, by ± 5 mol %. For example, in one exemplary lipid particle formulation, the target amount of cationic lipid is 57.1 mol %, but the actual amount of cationic lipid may be ± 5 mol %, ± 4 mol %, ± 3 mol %, ± 2 mol %, ± 1 mol %, ± 0.75 mol %, ± 0.5 mol %, ± 0.25 mol %, or ± 0.1 mol % of that target amount, with the balance of the formulation being made up of other lipid components (adding up to 100 mol % of total lipids present in the particle; however, one skilled in the art will understand that the total mol % may deviate slightly from 100% due to rounding, for example, 99.9 mol % or 100.1 mol %.).
N,N-dimethyl-2,3-bis((9Z,12Z)-octadeca-9,12-dienyloxy)propan-l-amine (5)
2-(2,2-di((9Z,12Z)-octadeca-9,12-dienyl)-l,3-dioxolan-4-yl)-N,N-dimethylethanamine (6)
3-((6Z,9Z,28Z,31Z)-heptatriaconta-6,9,28,31-tetraen-19-yloxy)-N,N-dimethylpropan-l-amine
(Z)-12-((Z)-dec-4-enyl)docos-16-en-l 1-yl 5-(dimethylamino)pentanoate (53)
(6Z,16Z)-12-((Z)-dec-4-enyl)docosa-6, 16-dien-l 1-yl 5-(dimethylamino)pentanoate (13)
12-decyldocosan-l l-yl 5-(dimethylamino)pentanoate (14).
Non-cationic Lipids
The non-cationic lipids used in the lipid particles can be any of a variety of neutral uncharged, zwitterionic, or anionic lipids capable of producing a stable complex.
Non-limiting examples of non-cationic lipids include phospholipids such as lecithin, phosphatidylethanolamine, lysolecithin, lysophosphatidylethanolamine, phosphatidylserine,
phosphatidylinositol, sphingomyelin, egg sphingomyelin (ESM), cephalin, cardiolipin, phosphatidic acid, cerebrosides, dicetylphosphate, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC),
dioleoylphosphatidylglycerol (DOPG), dipalmitoylphosphatidylglycerol (DPPG),
dioleoylphosphatidylethanolamine (DOPE), palmitoyloleoyl-phosphatidylcholine (POPC), palmitoyloleoyl-phosphatidylethanolamine (POPE), palmitoyloleyol-phosphatidylglycerol (POPG), dioleoylphosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane-l-carboxylate (DOPE-mal), dipalmitoyl-phosphatidylethanolamine (DPPE), dimyristoyl- phosphatidylethanolamine (DMPE), distearoyl-phosphatidylethanolamine (DSPE),
monomethyl-phosphatidylethanolamine, dimethyl -phosphatidyl ethanolamine, dielaidoyl- phosphatidylethanolamine (DEPE), stearoyloleoyl-phosphatidylethanolamine (SOPE), lysophosphatidylcholine, dilinoleoylphosphatidylcholine, and mixtures thereof. Other diacylphosphatidylcholine and diacylphosphatidylethanolamine phospholipids can also be used. The acyl groups in these lipids are preferably acyl groups derived from fatty acids having C10- C24 carbon chains, e.g., lauroyl, myristoyl, palmitoyl, stearoyl, or oleoyl.
Additional examples of non-cationic lipids include sterols such as cholesterol and derivatives thereof. Non-limiting examples of cholesterol derivatives include polar analogues such as 5a-cholestanol, 5P-coprostanol, cholesteryl-(2'-hydroxy)-ethyl ether, cholesteryl-(4'- hydroxy)-butyl ether, and 6-ketocholestanol; non-polar analogues such as 5a-cholestane, cholestenone, 5a-cholestanone, 5P-cholestanone, and cholesteryl decanoate; and mixtures thereof. In preferred embodiments, the cholesterol derivative is a polar analogue such as cholesteryl-(4'-hydroxy)-butyl ether. The synthesis of cholesteryl-(2'-hydroxy)-ethyl ether is described in PCT Publication No. WO 09/127060, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
In some embodiments, the non-cationic lipid present in the lipid particles comprises or consists of a mixture of one or more phospholipids and cholesterol or a derivative thereof. In other embodiments, the non-cationic lipid present in the lipid particles comprises or consists of one or more phospholipids, e.g., a cholesterol -free lipid particle formulation. In yet other embodiments, the non-cationic lipid present in the lipid particles comprises or consists of cholesterol or a derivative thereof, e.g., a phospholipid-free lipid particle formulation.
Other examples of non-cationic lipids suitable for use include nonphosphorous containing lipids such as, e.g., stearylamine, dodecylamine, hexadecylamine, acetyl palmitate, glycerolricinoleate, hexadecyl stereate, isopropyl myristate, amphoteric acrylic polymers, triethanolamine-lauryl sulfate, alkyl-aryl sulfate poly ethyl oxylated fatty acid amides, dioctadecyl dimethyl ammonium bromide, ceramide, sphingomyelin, and the like.
In some embodiments, the non-cationic lipid comprises from about 10 mol % to about 60 mol %, from about 20 mol % to about 55 mol %, from about 20 mol % to about 45 mol %, from about 20 mol % to about 40 mol %, from about 25 mol % to about 50 mol %, from about 25 mol % to about 45 mol %, from about 30 mol % to about 50 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 35 mol % to about 45 mol %, from about 37 mol % to about 45 mol %, or about 35 mol %, 36 mol %, 37 mol %, 38 mol %, 39 mol %, 40 mol %, 41 mol %, 42 mol %, 43 mol %, 44 mol %, or 45 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
In embodiments where the lipid particles contain a mixture of phospholipid and cholesterol or a cholesterol derivative, the mixture may comprise up to about 40 mol %, 45 mol %, 50 mol %, 55 mol %, or 60 mol % of the total lipid present in the particle.
In some embodiments, the phospholipid component in the mixture may comprise from about 2 mol % to about 20 mol %, from about 2 mol % to about 15 mol %, from about 2 mol % to about 12 mol %, from about 4 mol % to about 15 mol %, or from about 4 mol % to about 10 mol % (or any fraction thereof or range therein) of the total lipid present in the particle. In certain embodiments, the phospholipid component in the mixture comprises from about 5 mol % to about 17 mol %, from about 7 mol % to about 17 mol %, from about 7 mol % to about 15 mol %, from about 8 mol % to about 15 mol %, or about 8 mol %, 9 mol %, 10 mol %, 1 1 mol %, 12 mol %, 13 mol %, 14 mol %, or 15 mol % (or any fraction thereof or range therein) of the total lipid present in the particle. As a non-limiting example, a lipid particle formulation comprising a mixture of phospholipid and cholesterol may comprise a phospholipid such as DPPC or DSPC at about 7 mol % (or any fraction thereof), e.g., in a mixture with cholesterol or a cholesterol derivative at about 34 mol % (or any fraction thereof) of the total lipid present in the particle. As another non-limiting example, a lipid particle formulation comprising a mixture of phospholipid and cholesterol may comprise a phospholipid such as DPPC or DSPC at about 7 mol % (or any fraction thereof), e.g., in a mixture with cholesterol or a cholesterol derivative at about 32 mol % (or any fraction thereof) of the total lipid present in the particle.
By way of further example, a lipid formulation useful has a lipid to drug (e.g., siRNA) ratio of about 10: 1 (e.g., a lipid:drug ratio of from 9.5 : 1 to 1 1 : 1, or from 9.9: 1 to 1 1 : 1, or from 10: 1 to 10.9: 1). In certain other embodiments, a lipid formulation useful has a lipid to drug (e.g., siRNA) ratio of about 9: 1 (e.g., a lipid:drug ratio of from 8.5 : 1 to 10: 1, or from 8.9: 1 to 10: 1, or from 9: 1 to 9.9: 1, including 9.1 : 1, 9.2: 1, 9.3 : 1, 9.4: 1, 9.5 : 1, 9.6: 1, 9.7: 1, and 9.8: 1).
In other embodiments, the cholesterol component in the mixture may comprise from about 25 mol % to about 45 mol %, from about 25 mol % to about 40 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 27 mol % to about
37 mol %, from about 25 mol % to about 30 mol %, or from about 35 mol % to about 40 mol % (or any fraction thereof or range therein) of the total lipid present in the particle. In certain preferred embodiments, the cholesterol component in the mixture comprises from about 25 mol % to about 35 mol %, from about 27 mol % to about 35 mol %, from about 29 mol % to about 35 mol %, from about 30 mol % to about 35 mol %, from about 30 mol % to about 34 mol %, from about 31 mol % to about 33 mol %, or about 30 mol %, 31 mol %, 32 mol %, 33 mol %, 34 mol %, or 35 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
In embodiments where the lipid particles are phospholipid-free, the cholesterol or derivative thereof may comprise up to about 25 mol %, 30 mol %, 35 mol %, 40 mol %, 45 mol %, 50 mol %, 55 mol %, or 60 mol % of the total lipid present in the particle.
In some embodiments, the cholesterol or derivative thereof in the phospholipid-free lipid particle formulation may comprise from about 25 mol % to about 45 mol %, from about 25 mol % to about 40 mol %, from about 30 mol % to about 45 mol %, from about 30 mol % to about 40 mol %, from about 31 mol % to about 39 mol %, from about 32 mol % to about 38 mol %, from about 33 mol % to about 37 mol %, from about 35 mol % to about 45 mol %, from about 30 mol % to about 35 mol %, from about 35 mol % to about 40 mol %, or about 30 mol %, 31 mol %, 32 mol %, 33 mol %, 34 mol %, 35 mol %, 36 mol %, 37 mol %, 38 mol %, 39 mol %, or 40 mol % (or any fraction thereof or range therein) of the total lipid present in the particle. As a non-limiting example, a lipid particle formulation may comprise cholesterol at about 37 mol % (or any fraction thereof) of the total lipid present in the particle. As another non-limiting example, a lipid particle formulation may comprise cholesterol at about 35 mol % (or any fraction thereof) of the total lipid present in the particle.
In other embodiments, the non-cationic lipid comprises from about 5 mol % to about 90 mol %, from about 10 mol % to about 85 mol %, from about 20 mol % to about 80 mol %, about 10 mol % (e.g., phospholipid only), or about 60 mol % (e.g., phospholipid and cholesterol or derivative thereof) (or any fraction thereof or range therein) of the total lipid present in the particle.
Additional percentages and ranges of non-cationic lipids suitable for use in the lipid particles are described in PCT Publication No. WO 09/127060, U. S. Published Application No. US 201 1/0071208, PCT Publication No. WO201 1/000106, and U. S. Published Application No. US 201 1/0076335, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
It should be understood that the percentage of non-cationic lipid present in the lipid particles is a target amount, and that the actual amount of non-cationic lipid present in the
formulation may vary, for example, by ± 5 mol %, ± 4 mol %, ± 3 mol %, ± 2 mol %, ± 1 mol %, ± 0.75 mol %, ± 0.5 mol %, ± 0.25 mol %, or ± 0.1 mol %.
J/p/i/ Conjugates
In addition to cationic and non-cationic lipids, the lipid particles may further comprise a lipid conjugate. The conjugated lipid is useful in that it prevents the aggregation of particles. Suitable conjugated lipids include, but are not limited to, PEG-lipid conjugates, POZ-lipid conjugates, ATTA-lipid conjugates, cationic-polymer-lipid conjugates (CPLs), and mixtures thereof. In certain embodiments, the particles comprise either a PEG-lipid conjugate or an ATTA-lipid conjugate together with a CPL.
In a preferred embodiment, the lipid conjugate is a PEG-lipid. Examples of PEG-lipids include, but are not limited to, PEG coupled to dialkyloxypropyls (PEG-DAA) as described in, e.g., PCT Publication No. WO 05/026372, PEG coupled to diacylglycerol (PEG-DAG) as described in, e.g., U.S. Patent Publication Nos. 20030077829 and 2005008689, PEG coupled to phospholipids such as phosphatidylethanolamine (PEG-PE), PEG conjugated to ceramides as described in, e.g., U.S. Patent No. 5,885,613, PEG conjugated to cholesterol or a derivative thereof, and mixtures thereof. The disclosures of these patent documents are herein incorporated by reference in their entirety for all purposes.
Additional PEG-lipids suitable for use include, without limitation, mPEG2000-l,2-di- 0-alkyl-s«3-carbomoylglyceride (PEG-C-DOMG). The synthesis of PEG-C-DOMG is described in PCT Publication No. WO 09/086558, the disclosure of which is herein incorporated by reference in its entirety for all purposes. Yet additional suitable PEG-lipid conjugates include, without limitation, l-[8'-(l,2-dimyristoyl-3-propanoxy)-carboxamido-3',6'- dioxaoctanyl]carbamoyl-ro-methyl-poly(ethylene glycol) (2KPEG-DMG). The synthesis of 2KPEG-DMG is described in U.S. Patent No. 7,404,969, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
PEG is a linear, water-soluble polymer of ethylene PEG repeating units with two terminal hydroxyl groups. PEGs are classified by their molecular weights; for example, PEG 2000 has an average molecular weight of about 2,000 daltons, and PEG 5000 has an average molecular weight of about 5,000 daltons. PEGs are commercially available from Sigma
Chemical Co. and other companies and include, but are not limited to, the following:
monomethoxypolyethylene glycol (MePEG-OH), monomethoxypolyethylene glycol-succinate
(MePEG-S), monomethoxypolyethylene glycol-succinimidyl succinate (MePEG-S-NHS), monomethoxypolyethylene glycol-amine (MePEG-NEh), monomethoxypolyethylene glycol- tresylate (MePEG-TRES), monomethoxypolyethylene glycol-imidazolyl-carbonyl (MePEG- EVI), as well as such compounds containing a terminal hydroxyl group instead of a terminal
methoxy group (e.g., HO-PEG-S, HO-PEG-S-NHS, HO-PEG- H2, etc.). Other PEGs such as those described in U. S. Patent Nos. 6,774, 180 and 7,053, 150 (e.g., mPEG (20 KDa) amine) are also useful for preparing the PEG-lipid conjugates. The disclosures of these patents are herein incorporated by reference in their entirety for all purposes. In addition,
monomethoxypolyethyleneglycol-acetic acid (MePEG-CH2COOH) is particularly useful for preparing PEG-lipid conjugates including, e.g. , PEG-DAA conjugates.
The PEG moiety of the PEG-lipid conjugates described herein may comprise an average molecular weight ranging from about 550 daltons to about 10,000 daltons. In certain instances, the PEG moiety has an average molecular weight of from about 750 daltons to about 5,000 daltons (e.g., from about 1,000 daltons to about 5,000 daltons, from about 1 ,500 daltons to about 3,000 daltons, from about 750 daltons to about 3,000 daltons, from about 750 daltons to about 2,000 daltons, etc.). In preferred embodiments, the PEG moiety has an average molecular weight of about 2,000 daltons or about 750 daltons.
In certain instances, the PEG can be optionally substituted by an alkyl, alkoxy, acyl, or aryl group. The PEG can be conjugated directly to the lipid or may be linked to the lipid via a linker moiety. Any linker moiety suitable for coupling the PEG to a lipid can be used including, e.g., non-ester containing linker moieties and ester-containing linker moieties. In a preferred embodiment, the linker moiety is a non-ester containing linker moiety. As used herein, the term "non-ester containing linker moiety" refers to a linker moiety that does not contain a carboxylic ester bond (-OC(O)-). Suitable non-ester containing linker moieties include, but are not limited to, amido (-C(O) H-), amino (- R-), carbonyl (-C(O)-), carbamate (- HC(O)O-), urea (- HC(O) H-), disulphide (-S-S-), ether (-0-), succinyl (-(0)CCH2CH2C(0)-), succinamidyl (- HC(0)CH2CH2C(0) H-), ether, disulphide, as well as combinations thereof (such as a linker containing both a carbamate linker moiety and an amido linker moiety). In a preferred embodiment, a carbamate linker is used to couple the PEG to the lipid.
In other embodiments, an ester containing linker moiety is used to couple the PEG to the lipid. Suitable ester containing linker moieties include, e.g., carbonate (-OC(O)O-), succinoyl, phosphate esters (-O-(O)POH-O-), sulfonate esters, and combinations thereof.
Phosphatidylethanolamines having a variety of acyl chain groups of varying chain lengths and degrees of saturation can be conjugated to PEG to form the lipid conjugate. Such phosphatidylethanolamines are commercially available, or can be isolated or synthesized using conventional techniques known to those of skill in the art. Phosphatidyl-ethanolamines containing saturated or unsaturated fatty acids with carbon chain lengths in the range of C10 to
C2o are preferred. Phosphatidylethanolamines with mono- or diunsaturated fatty acids and mixtures of saturated and unsaturated fatty acids can also be used. Suitable
phosphatidylethanolamines include, but are not limited to, dimyristoyl- phosphatidylethanolamine (DMPE), dipalmitoyl-phosphatidylethanolamine (DPPE), dioleoylphosphatidylethanolamine (DOPE), and distearoyl-phosphatidylethanolamine (DSPE).
The term " ATTA" or "polyamide" includes, without limitation, compounds described in U.S. Patent Nos. 6,320,017 and 6,586,559, the disclosures of which are herein incorporated by reference in their entirety for all purposes. These compounds include a compound having the formula:
wherein R is a member selected from the group consisting of hydrogen, alkyl and acyl; R1 is a member selected from the group consisting of hydrogen and alkyl; or optionally, R and R1 and the nitrogen to which they are bound form an azido moiety; R2 is a member of the group selected from hydrogen, optionally substituted alkyl, optionally substituted aryl and a side chain of an amino acid; R3 is a member selected from the group consisting of hydrogen, halogen, hydroxy, alkoxy, mercapto, hydrazino, amino and R4R5, wherein R4 and R5 are independently hydrogen or alkyl; n is 4 to 80; m is 2 to 6; p is 1 to 4; and q is 0 or 1. It will be apparent to those of skill in the art that other polyamides can be.
The term "diacylglycerol" or "DAG" includes a compound having 2 fatty acyl chains, R1 and R2, both of which have independently between 2 and 30 carbons bonded to the 1- and 2- position of glycerol by ester linkages. The acyl groups can be saturated or have varying degrees of unsaturation. Suitable acyl groups include, but are not limited to, lauroyl (C12), myristoyl (C14), palmitoyl (C16), stearoyl (C18), and icosoyl (C20). In preferred embodiments, R1 and R2 are the same, i.e., R1 and R2 are both myristoyl {i.e., dimyristoyl), R1 and R2 are both stearoyl {i.e., distearoyl), etc. Diacylglycerols have the following general formula:
The term "dialkyloxypropyl" or "DAA" includes a compound having 2 alkyl chains, R1 and R2, both of which have independently between 2 and 30 carbons. The alkyl groups can
be saturated or have varying degrees of unsaturation Dialkyloxypropyls have the following general formula:
In a preferred embodiment, the PEG-lipid is a PEG-DAA conjugate having the following formula:
wherein R1 and R2 are independently selected and are long-chain alkyl groups having from about 10 to about 22 carbon atoms; PEG is a polyethyleneglycol; and L is a non-ester containing linker moiety or an ester containing linker moiety as described above. The long-chain alkyl groups can be saturated or unsaturated. Suitable alkyl groups include, but are not limited to, decyl (Cio), lauryl (C12), myristyl (C14), palmityl (C16), stearyl (C18), and icosyl (C20). In preferred embodiments, R1 and R2 are the same, i.e., R1 and R2 are both myristyl {i.e., dimyristyl), R1 and R2 are both stearyl {i.e., distearyl), etc.
In Formula VII above, the PEG has an average molecular weight ranging from about 550 daltons to about 10,000 daltons. In certain instances, the PEG has an average molecular weight of from about 750 daltons to about 5,000 daltons {e.g., from about 1 ,000 daltons to about 5,000 daltons, from about 1,500 daltons to about 3,000 daltons, from about 750 daltons to about 3,000 daltons, from about 750 daltons to about 2,000 daltons, etc.). In preferred embodiments, the PEG has an average molecular weight of about 2,000 daltons or about 750 daltons. The PEG can be optionally substituted with alkyl, alkoxy, acyl, or aryl groups. In certain embodiments, the terminal hydroxyl group is substituted with a methoxy or methyl group.
In a preferred embodiment, "L" is a non-ester containing linker moiety. Suitable non- ester containing linkers include, but are not limited to, an amido linker moiety, an amino linker moiety, a carbonyl linker moiety, a carbamate linker moiety, a urea linker moiety, an ether linker moiety, a disulphide linker moiety, a succinamidyl linker moiety, and combinations thereof. In a preferred embodiment, the non-ester containing linker moiety is a carbamate linker moiety {i.e. , a PEG-C-DAA conjugate). In another preferred embodiment, the non-ester containing linker moiety is an amido linker moiety {i.e. , a PEG- 4-DAA conjugate). In yet another
preferred embodiment, the non-ester containing linker moiety is a succinamidyl linker moiety (i.e., a PEG-^-DAA conjugate).
In particular embodiments, the PEG-lipid conjugate is selected from:
Ό'
H
..O
« (66) (PEG-C-DMA); and
The PEG-DAA conjugates are synthesized using standard techniques and reagents known to those of skill in the art. It will be recognized that the PEG-DAA conjugates will contain various amide, amine, ether, thio, carbamate, and urea linkages. Those of skill in the art will recognize that methods and reagents for forming these bonds are well known and readily available. See, e.g., March, ADVANCED ORGANIC CHEMISTRY (Wiley 1992); Larock,
COMPREHENSIVE ORGANIC TRANSFORMATIONS (VCH 1989); and Furniss, VOGEL' S TEXTBOOK OF PRACTICAL ORGANIC CHEMISTRY, 5th ed. (Longman 1989). It will also be appreciated that any functional groups present may require protection and deprotection at different points in the synthesis of the PEG-DAA conjugates. Those of skill in the art will recognize that such techniques are well known. See, e.g., Green and Wuts, PROTECTIVE GROUPS IN ORGANIC SYNTHESIS (Wiley 1991).
Preferably, the PEG-DAA conjugate is a PEG-didecyloxypropyl (Cio) conjugate, a PEG-dilauryloxypropyl (C12) conjugate, a PEG-dimyristyloxypropyl (C14) conjugate, a PEG- dipalmityloxypropyl (Ci6) conjugate, or a PEG-distearyloxypropyl (Cis) conjugate. In these embodiments, the PEG preferably has an average molecular weight of about 750 or about 2,000 daltons. In one particularly preferred embodiment, the PEG-lipid conjugate comprises
PEG2000-C-DMA, wherein the "2000" denotes the average molecular weight of the PEG, the "C" denotes a carbamate linker moiety, and the "DMA" denotes dimyristyloxypropyl. In another particularly preferred embodiment, the PEG-lipid conjugate comprises PEG750-C- DMA, wherein the "750" denotes the average molecular weight of the PEG, the "C" denotes a carbamate linker moiety, and the "DMA" denotes dimyristyloxypropyl. In particular embodiments, the terminal hydroxyl group of the PEG is substituted with a methyl group.
Those of skill in the art will readily appreciate that other dialkyloxypropyls can be used in the PEG-DAA conjugates.
In addition to the foregoing, it will be readily apparent to those of skill in the art that other hydrophilic polymers can be used in place of PEG. Examples of suitable polymers that can be used in place of PEG include, but are not limited to, polyvinylpyrrolidone,
polymethyloxazoline, polyethyloxazoline, polyhydroxypropyl methacrylamide,
polymethacrylamide and polydimethylacrylamide, polylactic acid, polyglycolic acid, and derivatized celluloses such as hydroxymethyl cellulose or hydroxyethylcellulose.
In addition to the foregoing components, the lipid particles can further comprise cationic poly(ethylene glycol) (PEG) lipids or CPLs {see, e.g., Chen et al., Bioconj. Chem., 11 :433-437 (2000); U.S. Patent No. 6,852,334; PCT Publication No. WO 00/62813, the disclosures of which are herein incorporated by reference in their entirety for all purposes).
Suitable CPLs include compounds of Formula VIII:
A-W-Y (VIII),
wherein A, W, and Y are as described below.
With reference to Formula VIII, "A" is a lipid moiety such as an amphipathic lipid, a neutral lipid, or a hydrophobic lipid that acts as a lipid anchor. Suitable lipid examples include, but are not limited to, diacylglycerolyls, dialkylglycerolyls, N-N-dialkylaminos, 1,2-diacyloxy- 3-aminopropanes, and l,2-dialkyl-3-aminopropanes.
"W" is a polymer or an oligomer such as a hydrophilic polymer or oligomer.
Preferably, the hydrophilic polymer is a biocompatable polymer that is nonimmunogenic or possesses low inherent immunogenicity. Alternatively, the hydrophilic polymer can be weakly antigenic if used with appropriate adjuvants. Suitable nonimmunogenic polymers include, but are not limited to, PEG, polyamides, polylactic acid, polyglycolic acid, polylactic
acid/polyglycolic acid copolymers, and combinations thereof. In a preferred embodiment, the polymer has a molecular weight of from about 250 to about 7,000 daltons.
"Y" is a polycationic moiety. The term polycationic moiety refers to a compound, derivative, or functional group having a positive charge, preferably at least 2 positive charges at a selected pH, preferably physiological pH. Suitable polycationic moieties include basic amino acids and their derivatives such as arginine, asparagine, glutamine, lysine, and histidine;
spermine; spermidine; cationic dendrimers; polyamines; polyamine sugars; and amino polysaccharides. The polycationic moieties can be linear, such as linear tetralysine, branched or dendrimeric in structure. Polycationic moieties have between about 2 to about 15 positive charges, preferably between about 2 to about 12 positive charges, and more preferably between about 2 to about 8 positive charges at selected pH values. The selection of which polycationic moiety to employ may be determined by the type of particle application which is desired.
The charges on the polycationic moieties can be either distributed around the entire particle moiety, or alternatively, they can be a discrete concentration of charge density in one particular area of the particle moiety e.g., a charge spike. If the charge density is distributed on the particle, the charge density can be equally distributed or unequally distributed. All variations of charge distribution of the polycationic moiety are encompassed.
The lipid "A" and the nonimmunogenic polymer "W" can be attached by various methods and preferably by covalent attachment. Methods known to those of skill in the art can be used for the covalent attachment of "A" and "W." Suitable linkages include, but are not limited to, amide, amine, carboxyl, carbonate, carbamate, ester, and hydrazone linkages. It will be apparent to those skilled in the art that "A" and "W" must have complementary functional groups to effectuate the linkage. The reaction of these two groups, one on the lipid and the other on the polymer, will provide the desired linkage. For example, when the lipid is a diacylglycerol and the terminal hydroxyl is activated, for instance with NHS and DCC, to form an active ester, and is then reacted with a polymer which contains an amino group, such as with a polyamide (see, e.g., U.S. Patent Nos. 6,320,017 and 6,586,559, the disclosures of which are herein incorporated by reference in their entirety for all purposes), an amide bond will form between the two groups.
In certain instances, the polycationic moiety can have a ligand attached, such as a targeting ligand or a chelating moiety for complexing calcium. Preferably, after the ligand is attached, the cationic moiety maintains a positive charge. In certain instances, the ligand that is attached has a positive charge. Suitable ligands include, but are not limited to, a compound or device with a reactive functional group and include lipids, amphipathic lipids, carrier
compounds, bioaffinity compounds, biomaterials, biopolymers, biomedical devices, analytically detectable compounds, therapeutically active compounds, enzymes, peptides, proteins, antibodies, immune stimulators, radiolabels, fluorogens, biotin, drugs, haptens, DNA, RNA, polysaccharides, liposomes, virosomes, micelles, immunoglobulins, functional groups, other targeting moieties, or toxins.
In some embodiments, the lipid conjugate (e.g., PEG-lipid) comprises from about 0.1 mol % to about 3 mol %, from about 0.5 mol % to about 3 mol %, or about 0.6 mol %, 0.7 mol %, 0.8 mol %, 0.9 mol %, 1.0 mol %, 1.1 mol %, 1.2 mol %, 1.3 mol %, 1.4 mol %, 1.5 mol %, 1.6 mol %, 1.7 mol %, 1.8 mol %, 1.9 mol %, 2.0 mol %, 2.1 mol%, 2.2 mol%, 2.3 mol %, 2.4 mol %, 2.5 mol %, 2.6 mol %, 2.7 mol %, 2.8 mol %, 2.9 mol % or 3 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
In other embodiments, the lipid conjugate (e.g., PEG-lipid) comprises from about 0 mol % to about 20 mol %, from about 0.5 mol % to about 20 mol %, from about 2 mol % to
about 20 mol %, from about 1.5 mol % to about 18 mol %, from about 2 mol % to about 15 mol %, from about 4 mol % to about 15 mol %, from about 2 mol % to about 12 mol %, from about 5 mol % to about 12 mol %, or about 2 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
In further embodiments, the lipid conjugate (e.g., PEG-lipid) comprises from about 4 mol % to about 10 mol %, from about 5 mol % to about 10 mol %, from about 5 mol % to about 9 mol %, from about 5 mol % to about 8 mol %, from about 6 mol % to about 9 mol %, from about 6 mol % to about 8 mol %, or about 5 mol %, 6 mol %, 7 mol %, 8 mol %, 9 mol %, or 10 mol % (or any fraction thereof or range therein) of the total lipid present in the particle.
It should be understood that the percentage of lipid conjugate present in the lipid particles is a target amount, and that the actual amount of lipid conjugate present in the formulation may vary, for example, by ± 5 mol %, ± 4 mol %, ± 3 mol %, ± 2 mol %, ± 1 mol %, ± 0.75 mol %, ± 0.5 mol %, ± 0.25 mol %, or ± 0.1 mol %.
Additional percentages and ranges of lipid conjugates suitable for use in the lipid particles are described in PCT Publication No. WO 09/127060, U.S. Published Application No. US 2011/0071208, PCT Publication No. WO2011/000106, and U.S. Published Application No. US 2011/0076335, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
One of ordinary skill in the art will appreciate that the concentration of the lipid conjugate can be varied depending on the lipid conjugate employed and the rate at which the lipid particle is to become fusogenic.
By controlling the composition and concentration of the lipid conjugate, one can control the rate at which the lipid conjugate exchanges out of the lipid particle and, in turn, the rate at which the lipid particle becomes fusogenic. For instance, when a PEG-DAA conjugate is used as the lipid conjugate, the rate at which the lipid particle becomes fusogenic can be varied, for example, by varying the concentration of the lipid conjugate, by varying the molecular weight of the PEG, or by varying the chain length and degree of saturation of the alkyl groups on the PEG-DAA conjugate. In addition, other variables including, for example, pH, temperature, ionic strength, etc. can be used to vary and/or control the rate at which the lipid particle becomes fusogenic. Other methods which can be used to control the rate at which the lipid particle becomes fusogenic will become apparent to those of skill in the art upon reading this disclosure. Also, by controlling the composition and concentration of the lipid conjugate, one can control the lipid particle size.
Additional Carrier Systems
Non-limiting examples of additional lipid-based carrier systems suitable for use include lipoplexes {see, e.g., U.S. Patent Publication No. 20030203865; and Zhang et al, J. Control Release, 100: 165-180 (2004)), pH-sensitive lipoplexes (see, e.g., U.S. Patent
Publication No. 20020192275), reversibly masked lipoplexes (see, e.g., U.S. Patent Publication Nos. 20030180950), cationic lipid-based compositions (see, e.g., U.S. Patent No. 6,756,054; and U.S. Patent Publication No. 20050234232), cationic liposomes (see, e.g., U.S. Patent Publication Nos. 20030229040, 20020160038, and 20020012998; U.S. Patent No. 5,908,635; and PCT Publication No. WO 01/72283), anionic liposomes (see, e.g., U.S. Patent Publication No.
20030026831), pH-sensitive liposomes (see, e.g., U.S. Patent Publication No. 20020192274; and AU 2003210303), antibody-coated liposomes (see, e.g., U.S. Patent Publication No.
20030108597; and PCT Publication No. WO 00/50008), cell-type specific liposomes (see, e.g., U.S. Patent Publication No. 20030198664), liposomes containing nucleic acid and peptides (see, e.g., U.S. Patent No. 6,207,456), liposomes containing lipids derivatized with releasable hydrophilic polymers (see, e.g., U.S. Patent Publication No. 20030031704), lipid-entrapped nucleic acid (see, e.g., PCT Publication Nos. WO 03/057190 and WO 03/059322), lipid- encapsulated nucleic acid (see, e.g., U.S. Patent Publication No. 20030129221; and U.S. Patent No. 5,756,122), other liposomal compositions (see, e.g., U.S. Patent Publication Nos.
20030035829 and 20030072794; and U.S. Patent No. 6,200,599), stabilized mixtures of liposomes and emulsions (see, e.g., EP1304160), emulsion compositions (see, e.g., U.S. Patent No. 6,747,014), and nucleic acid micro-emulsions (see, e.g., U.S. Patent Publication No.
20050037086).
Examples of polymer-based carrier systems suitable for use include, but are not limited to, cationic polymer-nucleic acid complexes (i.e., polyplexes). To form a polyplex, a nucleic acid (e.g., a siRNA molecule described herein or a combination thereof) is typically complexed with a cationic polymer having a linear, branched, star, or dendritic polymeric structure that condenses the nucleic acid into positively charged particles capable of interacting with anionic proteoglycans at the cell surface and entering cells by endocytosis. In some embodiments, the polyplex comprises nucleic acid (e.g., a combination comprising the three siRNA molecules described in Example 1) complexed with a cationic polymer such as polyethylenimine (PEI)
(see, e.g., U.S. Patent No. 6,013,240; commercially available from Qbiogene, Inc. (Carlsbad,
CA) as In v/vojetPEI™, a linear form of PEI), polypropylenimine (PPI), polyvinylpyrrolidone
(PVP), poly-L-lysine (PLL), diethylaminoethyl (DEAE)-dextran, poly(P-amino ester) (PAE) polymers (see, e.g., Lynn et al, J. Am. Chem. Soc, 123 :8155-8156 (2001)), chitosan, polyamidoamine (PAMAM) dendrimers (see, e.g., Kukowska-Latallo et al, Proc. Natl. Acad.
Sci. USA, 93 :4897-4902 (1996)), porphyrin (see, e.g., U.S. Patent No. 6,620,805), polyvinylether (see, e.g., U.S. Patent Publication No. 20040156909), polycyclic amidinium (see, e.g., U.S. Patent Publication No. 20030220289), other polymers comprising primary amine, imine, guanidine, and/or imidazole groups (see, e.g., U.S. Patent No. 6,013,240; PCT
Publication No. WO/9602655; PCT Publication No. W095/21931; Zhang et al, J. Control
Release, 100: 165-180 (2004); and Tiera et al, Curr. Gene Ther., 6:59-71 (2006)), and a mixture thereof. In other embodiments, the polyplex comprises cationic polymer-nucleic acid complexes as described in U.S. Patent Publication Nos. 20060211643, 20050222064, 20030125281, and 20030185890, and PCT Publication No. WO 03/066069; biodegradable poly( -amino ester) polymer-nucleic acid complexes as described in U.S. Patent Publication No. 20040071654; microparticles containing polymeric matrices as described in U.S. Patent Publication No.
20040142475; other microparticle compositions as described in U.S. Patent Publication No. 20030157030; condensed nucleic acid complexes as described in U.S. Patent Publication No. 20050123600; and nanocapsule and microcapsule compositions as described in AU 2002358514 and PCT Publication No. WO 02/096551.
In certain instances, the siRNA may be complexed with cyclodextrin or a polymer thereof. Non-limiting examples of cyclodextrin-based carrier systems include the cyclodextrin- modified polymer-nucleic acid complexes described in U.S. Patent Publication No.
20040087024; the linear cyclodextrin copolymer-nucleic acid complexes described in U.S. Patent Nos. 6,509,323, 6,884,789, and 7,091, 192; and the cyclodextrin polymer-complexing agent-nucleic acid complexes described in U.S. Patent No. 7,018,609. In certain other instances, the siRNA may be complexed with a peptide or polypeptide. An example of a protein-based carrier system includes, but is not limited to, the cationic oligopeptide-nucleic acid complex described in PCT Publication No. W095/21931.
Preparation of Lipid Particles
The nucleic acid-lipid particles, in which a nucleic acid (e.g., a combination
comprising the three siRNA molecules described in Example 1) is entrapped within the lipid portion of the particle and is protected from degradation, can be formed by any method known in the art including, but not limited to, a continuous mixing method, a direct dilution process, and an in-line dilution process.
In particular embodiments, the cationic lipids may comprise lipids of Formula I-III or salts thereof, alone or in combination with other cationic lipids. In other embodiments, the non- cationic lipids are egg sphingomyelin (ESM), distearoylphosphatidylcholine (DSPC), dioleoylphosphatidyl choline (DOPC), l-palmitoyl-2-oleoyl-phosphatidyl choline (POPC), dipalmitoyl-phosphatidyl choline (DPPC), monomethyl-phosphatidylethanolamine, dimethyl -
phosphatidylethanolamine, 14:0 PE (1,2-dimyristoyl-phosphatidylethanolamine (DMPE)), 16:0 PE (1,2-dipalmitoyl-phosphatidylethanolamine (DPPE)), 18:0 PE (1,2-distearoyl- phosphatidylethanolamine (DSPE)), 18: 1 PE (1,2-dioleoyl-phosphatidylethanolamine (DOPE)), 18: 1 trans PE (1,2-dielaidoyl -phosphatidyl ethanolamine (DEPE)), 18:0-18: 1 PE (l-stearoyl-2- oleoyl-phosphatidylethanolamine (SOPE)), 16:0-18: 1 PE (l-palmitoyl-2-oleoyl- phosphatidylethanolamine (POPE)), polyethylene glycol-based polymers (e.g., PEG 2000, PEG 5000, PEG-modified diacylglycerols, or PEG-modified dialkyloxypropyls), cholesterol, derivatives thereof, or combinations thereof.
In certain embodiments, the nucleic acid-lipid particles produced via a continuous mixing method, e.g., a process that includes providing an aqueous solution comprising a siRNA in a first reservoir, providing an organic lipid solution in a second reservoir (wherein the lipids present in the organic lipid solution are solubilized in an organic solvent, e.g., a lower alkanol such as ethanol), and mixing the aqueous solution with the organic lipid solution such that the organic lipid solution mixes with the aqueous solution so as to substantially instantaneously produce a lipid vesicle (e.g., liposome) encapsulating the siRNA within the lipid vesicle. This process and the apparatus for carrying out this process are described in detail in U.S. Patent Publication No. 20040142025, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
The action of continuously introducing lipid and buffer solutions into a mixing environment, such as in a mixing chamber, causes a continuous dilution of the lipid solution with the buffer solution, thereby producing a lipid vesicle substantially instantaneously upon mixing. As used herein, the phrase "continuously diluting a lipid solution with a buffer solution" (and variations) generally means that the lipid solution is diluted sufficiently rapidly in a hydration process with sufficient force to effectuate vesicle generation. By mixing the aqueous solution comprising a nucleic acid with the organic lipid solution, the organic lipid solution undergoes a continuous stepwise dilution in the presence of the buffer solution (i.e., aqueous solution) to produce a nucleic acid-lipid particle.
The nucleic acid-lipid particles formed using the continuous mixing method typically have a size of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 110 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, less than about 120 nm, 110 nm, 100 nm, 90 nm, or 80 nm, or about 30 nm, 35 nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125 nm, 130 nm, 135 nm, 140 nm,
145 nm, or 150 nm (or any fraction thereof or range therein). The particles thus formed do not aggregate and are optionally sized to achieve a uniform particle size.
In another embodiment, the nucleic acid-lipid particles produced via a direct dilution process that includes forming a lipid vesicle (e.g., liposome) solution and immediately and directly introducing the lipid vesicle solution into a collection vessel containing a controlled amount of dilution buffer. In preferred aspects, the collection vessel includes one or more elements configured to stir the contents of the collection vessel to facilitate dilution. In one aspect, the amount of dilution buffer present in the collection vessel is substantially equal to the volume of lipid vesicle solution introduced thereto. As a non-limiting example, a lipid vesicle solution in 45% ethanol when introduced into the collection vessel containing an equal volume of dilution buffer will advantageously yield smaller particles.
In yet another embodiment, the nucleic acid-lipid particles produced via an in-line dilution process in which a third reservoir containing dilution buffer is fluidly coupled to a second mixing region. In this embodiment, the lipid vesicle (e.g., liposome) solution formed in a first mixing region is immediately and directly mixed with dilution buffer in the second mixing region. In preferred aspects, the second mixing region includes a T-connector arranged so that the lipid vesicle solution and the dilution buffer flows meet as opposing 180° flows; however, connectors providing shallower angles can be used, e.g., from about 27° to about 180° (e.g., about 90°). A pump mechanism delivers a controllable flow of buffer to the second mixing region. In one aspect, the flow rate of dilution buffer provided to the second mixing region is controlled to be substantially equal to the flow rate of lipid vesicle solution introduced thereto from the first mixing region. This embodiment advantageously allows for more control of the flow of dilution buffer mixing with the lipid vesicle solution in the second mixing region, and therefore also the concentration of lipid vesicle solution in buffer throughout the second mixing process. Such control of the dilution buffer flow rate advantageously allows for small particle size formation at reduced concentrations.
These processes and the apparatuses for carrying out these direct dilution and in-line dilution processes are described in detail in U.S. Patent Publication No. 20070042031, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
The nucleic acid-lipid particles formed using the direct dilution and in-line dilution processes typically have a size of from about 30 nm to about 150 nm, from about 40 nm to about 150 nm, from about 50 nm to about 150 nm, from about 60 nm to about 130 nm, from about 70 nm to about 1 10 nm, from about 70 nm to about 100 nm, from about 80 nm to about 100 nm, from about 90 nm to about 100 nm, from about 70 to about 90 nm, from about 80 nm to about 90 nm, from about 70 nm to about 80 nm, less than about 120 nm, 1 10 nm, 100 nm, 90 nm, or
80 nm, or about 30 nm, 35 nm, 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 1 10 nm, 1 15 nm, 120 nm, 125 nm, 130 nm, 135 nm, 140 nm, 145 nm, or 150 nm (or any fraction thereof or range therein). The particles thus formed do not aggregate and are optionally sized to achieve a uniform particle size.
The lipid particles can be sized by any of the methods available for sizing liposomes.
The sizing may be conducted in order to achieve a desired size range and relatively narrow distribution of particle sizes.
Several techniques are available for sizing the particles to a desired size. One sizing method, used for liposomes and equally applicable to the present particles, is described in U. S. Patent No. 4,737,323, the disclosure of which is herein incorporated by reference in its entirety for all purposes. Sonicating a particle suspension either by bath or probe sonication produces a progressive size reduction down to particles of less than about 50 nm in size. Homogenization is another method which relies on shearing energy to fragment larger particles into smaller ones. In a typical homogenization procedure, particles are recirculated through a standard emulsion homogenizer until selected particle sizes, typically between about 60 and about 80 nm, are observed. In both methods, the particle size distribution can be monitored by conventional laser-beam particle size discrimination, or QELS.
Extrusion of the particles through a small-pore polycarbonate membrane or an asymmetric ceramic membrane is also an effective method for reducing particle sizes to a relatively well-defined size distribution. Typically, the suspension is cycled through the membrane one or more times until the desired particle size distribution is achieved. The particles may be extruded through successively smaller-pore membranes, to achieve a gradual reduction in size.
In some embodiments, the nucleic acids present in the particles (e.g., the siRNA molecules) are precondensed as described in, e.g., U. S. Patent Application No. 09/744, 103, the disclosure of which is herein incorporated by reference in its entirety for all purposes.
In other embodiments, the methods may further comprise adding non-lipid polycations which are useful to effect the lipofection of cells using the present compositions. Examples of suitable non-lipid polycations include, hexadimethrine bromide (sold under the brand name POLYBRENE®, from Aldrich Chemical Co., Milwaukee, Wisconsin, USA) or other salts of hexadimethrine. Other suitable polycations include, for example, salts of poly-L-ornithine, poly-L-arginine, poly-L-lysine, poly-D-lysine, polyallylamine, and polyethyleneimine. Addition of these salts is preferably after the particles have been formed.
In some embodiments, the nucleic acid (e.g., siRNA) to lipid ratios (mass/mass ratios) in a formed nucleic acid-lipid particle will range from about 0.01 to about 0.2, from about 0.05
to about 0.2, from about 0.02 to about 0.1, from about 0.03 to about 0.1, or from about 0.01 to about 0.08. The ratio of the starting materials (input) also falls within this range. In other embodiments, the particle preparation uses about 400 μg nucleic acid per 10 mg total lipid or a nucleic acid to lipid mass ratio of about 0.01 to about 0.08 and, more preferably, about 0.04, which corresponds to 1.25 mg of total lipid per 50 μg of nucleic acid. In other preferred embodiments, the particle has a nucleic acid:lipid mass ratio of about 0.08.
In other embodiments, the lipid to nucleic acid (e.g., siRNA) ratios (mass/mass ratios) in a formed nucleic acid-lipid particle will range from about 1 (1 : 1) to about 100 (100: 1), from about 5 (5: 1) to about 100 (100: 1), from about 1 (1 : 1) to about 50 (50: 1), from about 2 (2: 1) to about 50 (50: 1), from about 3 (3 : 1) to about 50 (50: 1), from about 4 (4: 1) to about 50 (50: 1), from about 5 (5: 1) to about 50 (50: 1), from about 1 (1 : 1) to about 25 (25: 1), from about 2 (2: 1) to about 25 (25 : 1), from about 3 (3 : 1) to about 25 (25 : 1), from about 4 (4: 1) to about 25 (25 : 1), from about 5 (5: 1) to about 25 (25: 1), from about 5 (5: 1) to about 20 (20: 1), from about 5 (5: 1) to about 15 (15: 1), from about 5 (5: 1) to about 10 (10: 1), or about 5 (5: 1), 6 (6: 1), 7 (7: 1), 8 (8: 1), 9 (9: 1), 10 (10: 1), 11 (11 : 1), 12 (12: 1), 13 (13 : 1), 14 (14: 1), 15 (15: 1), 16 (16: 1), 17
(17: 1), 18 (18: 1), 19 (19: 1), 20 (20: 1), 21 (21 : 1), 22 (22: 1), 23 (23 : 1), 24 (24: 1), or 25 (25: 1), or any fraction thereof or range therein. The ratio of the starting materials (input) also falls within this range.
As previously discussed, the conjugated lipid may further include a CPL. A variety of general methods for making lipid particle-CPLs (CPL-containing lipid particles) are discussed herein. Two general techniques include the "post-insertion" technique, that is, insertion of a CPL into, for example, a pre-formed lipid particle, and the "standard" technique, wherein the CPL is included in the lipid mixture during, for example, the lipid particle formation steps. The post-insertion technique results in lipid particles having CPLs mainly in the external face of the lipid particle bilayer membrane, whereas standard techniques provide lipid particles having CPLs on both internal and external faces. The method is especially useful for vesicles made from phospholipids (which can contain cholesterol) and also for vesicles containing PEG-lipids (such as PEG-DAAs and PEG-DAGs). Methods of making lipid particle-CPLs are taught, for example, in U.S. Patent Nos. 5,705,385; 6,586,410; 5,981,501; 6,534,484; and 6,852,334; U.S. Patent Publication No. 20020072121; and PCT Publication No. WO 00/62813, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
Administration of Lipid Particles
The lipid particles (e.g., a nucleic-acid lipid particle) can be adsorbed to almost any cell type with which they are mixed or contacted. Once adsorbed, the particles can either be endocytosed by a portion of the cells, exchange lipids with cell membranes, or fuse with the
cells. Transfer or incorporation of the siRNA portion of the particle can take place via any one of these pathways. In particular, when fusion takes place, the particle membrane is integrated into the cell membrane and the contents of the particle combine with the intracellular fluid.
The lipid particles (e.g., nucleic acid-lipid particles) can be administered either alone or in a mixture with a pharmaceutically acceptable carrier (e.g., physiological saline or phosphate buffer) selected in accordance with the route of administration and standard pharmaceutical practice. Generally, normal buffered saline (e.g., 135-150 mM NaCl) will be employed as the pharmaceutically acceptable carrier. Other suitable carriers include, e.g., water, buffered water, 0.4% saline, 0.3% glycine, and the like, including glycoproteins for enhanced stability, such as albumin, lipoprotein, globulin, etc. Additional suitable carriers are described in, e.g.,
REMINGTON'S PHARMACEUTICAL SCIENCES, Mack Publishing Company, Philadelphia, PA, 17th ed. (1985). As used herein, "carrier" includes any and all solvents, dispersion media, vehicles, coatings, diluents, antibacterial and antifungal agents, isotonic and absorption delaying agents, buffers, carrier solutions, suspensions, colloids, and the like. The phrase
"pharmaceutically acceptable" refers to molecular entities and compositions that do not produce an allergic or similar untoward reaction when administered to a human.
The pharmaceutically acceptable carrier is generally added following lipid particle formation. Thus, after the lipid particle is formed, the particle can be diluted into
pharmaceutically acceptable carriers such as normal buffered saline.
The concentration of particles in the pharmaceutical formulations can vary widely, i.e., from less than about 0.05%, usually at or at least about 2 to 5%, to as much as about 10 to 90% by weight, and will be selected primarily by fluid volumes, viscosities, etc., in accordance with the particular mode of administration selected. For example, the concentration may be increased to lower the fluid load associated with treatment. This may be particularly desirable in patients having atherosclerosis-associated congestive heart failure or severe hypertension. Alternatively, particles composed of irritating lipids may be diluted to low concentrations to lessen
inflammation at the site of administration.
The pharmaceutical compositions may be sterilized by conventional, well-known sterilization techniques. Aqueous solutions can be packaged for use or filtered under aseptic conditions and lyophilized, the lyophilized preparation being combined with a sterile aqueous solution prior to administration. The compositions can contain pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents and the like, for example, sodium acetate, sodium lactate, sodium chloride, potassium chloride, and calcium chloride. Additionally, the particle suspension may include lipid-protective agents which protect lipids against free-radical and
lipid-peroxidative damages on storage. Lipophilic free-radical quenchers, such as alphatocopherol, and water-soluble iron-specific chelators, such as ferrioxamine, are suitable.
In vivo Administration
Systemic delivery for in vivo therapy, e.g., delivery of a siRNA molecule described herein, such as a combination comprising the three siRNA molecules described in Example 1, to a distal target cell via body systems such as the circulation, may be achieved using nucleic acid- lipid particles, such as those described in PCT Publication Nos. WO 05/007196, WO 05/121348, WO 05/120152, and WO 04/002453, the disclosures of which are herein incorporated by reference in their entirety for all purposes.
For in vivo administration, administration can be in any manner known in the art, e.g., by injection, oral administration, inhalation {e.g., intransal or intratracheal), transdermal application, or rectal administration. Administration can be accomplished via single or divided doses. The pharmaceutical compositions can be administered parenterally, i.e., intraarticularly, intravenously, intraperitoneally, subcutaneously, or intramuscularly. In some embodiments, the pharmaceutical compositions are administered intravenously or intraperitoneally by a bolus injection {see, e.g., U.S. Patent No. 5,286,634). Intracellular nucleic acid delivery has also been discussed in Straubringer et al. , Methods Enzymol. , 101 :512 (1983); Mannino et al,
Biotechniques, 6:682 (1988); Nicolau et al, Crit. Rev. Ther. Drug Carrier Syst., 6:239 (1989); and Behr, Acc. Chem. Res., 26:274 (1993). Still other methods of administering lipid-based therapeutics are described in, for example, U.S. Patent Nos. 3,993,754; 4, 145,410; 4,235,871; 4,224,179; 4,522,803; and 4,588,578. The lipid particles can be administered by direct injection at the site of disease or by injection at a site distal from the site of disease {see, e.g., Culver, HUMAN GENE THERAPY, Mary Ann Liebert, Inc., Publishers, New York. pp.70-71(1994)). The disclosures of the above-described references are herein incorporated by reference in their entirety for all purposes.
In embodiments where the lipid particles are administered intravenously, at least about 5%, 10%, 15%), 20%), or 25% of the total injected dose of the particles is present in plasma about 8, 12, 24, 36, or 48 hours after injection. In other embodiments, more than about 20%, 30%, 40%) and as much as about 60%, 70% or 80% of the total injected dose of the lipid particles is present in plasma about 8, 12, 24, 36, or 48 hours after injection. In certain instances, more than about 10%) of a plurality of the particles is present in the plasma of a human about 1 hour after administration. In certain other instances, the presence of the lipid particles is detectable at least about 1 hour after administration of the particle. In some embodiments, the presence of a siRNA molecule is detectable in cells at about 8, 12, 24, 36, 48, 60, 72 or 96 hours after administration. In other embodiments, downregulation of expression of a target sequence, such
as a viral or host sequence, by a siRNA molecule is detectable at about 8, 12, 24, 36, 48, 60, 72 or 96 hours after administration. In yet other embodiments, downregulation of expression of a target sequence, such as a viral or host sequence, by a siRNA molecule occurs preferentially in infected cells and/or cells capable of being infected. In further embodiments, the presence or effect of a siRNA molecule in cells at a site proximal or distal to the site of administration is detectable at about 12, 24, 48, 72, or 96 hours, or at about 6, 8, 10, 12, 14, 16, 18, 19, 20, 22, 24, 26, or 28 days after administration. In additional embodiments, the lipid particles are
administered parenterally or intraperitoneally.
The compositions, either alone or in combination with other suitable components, can be made into aerosol formulations (i.e., they can be "nebulized") to be administered via inhalation (e.g., intranasally or intratracheally) (see, Brigham et al, Am. J. Sci., 298:278 (1989)). Aerosol formulations can be placed into pressurized acceptable propellants, such as
dichlorodifluoromethane, propane, nitrogen, and the like.
In certain embodiments, the pharmaceutical compositions may be delivered by intranasal sprays, inhalation, and/or other aerosol delivery vehicles. Methods for delivering nucleic acid compositions directly to the lungs via nasal aerosol sprays have been described, e.g., in U.S. Patent Nos. 5,756,353 and 5,804,212. Likewise, the delivery of drugs using intranasal microparticle resins and lysophosphatidyl -glycerol compounds (U.S. Patent
5,725,871) are also well-known in the pharmaceutical arts. Similarly, transmucosal drug delivery in the form of a polytetrafluoroetheylene support matrix is described in U.S. Patent No. 5,780,045. The disclosures of the above-described patents are herein incorporated by reference in their entirety for all purposes.
Formulations suitable for parenteral administration, such as, for example, by intraarticular (in the joints), intravenous, intramuscular, intradermal, intraperitoneal, and subcutaneous routes, include aqueous and non-aqueous, isotonic sterile injection solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic with the blood of the intended recipient, and aqueous and non-aqueous sterile suspensions that can include suspending agents, solubilizers, thickening agents, stabilizers, and preservatives.
Generally, when administered intravenously, the lipid particle formulations are formulated with a suitable pharmaceutical carrier. Suitable formulations are found, for example, in REMINGTON' S PHARMACEUTICAL SCIENCES, Mack Publishing Company,
Philadelphia, PA, 17th ed. (1985). A variety of aqueous carriers may be used, for example, water, buffered water, 0.4% saline, 0.3% glycine, and the like, and may include glycoproteins for enhanced stability, such as albumin, lipoprotein, globulin, etc. Generally, normal buffered
saline (135-150 mM NaCl) will be employed as the pharmaceutically acceptable carrier, but other suitable carriers will suffice. These compositions can be sterilized by conventional liposomal sterilization techniques, such as filtration. The compositions may contain
pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents, wetting agents and the like, for example, sodium acetate, sodium lactate, sodium chloride, potassium chloride, calcium chloride, sorbitan monolaurate, triethanolamine oleate, etc. These compositions can be sterilized using the techniques referred to above or, alternatively, they can be produced under sterile conditions. The resulting aqueous solutions may be packaged for use or filtered under aseptic conditions and lyophilized, the lyophilized preparation being combined with a sterile aqueous solution prior to administration.
In certain applications, the lipid particles disclosed herein may be delivered via oral administration to the individual. The particles may be incorporated with excipients and used in the form of ingestible tablets, buccal tablets, troches, capsules, pills, lozenges, elixirs, mouthwash, suspensions, oral sprays, syrups, wafers, and the like {see, e.g., U.S. Patent Nos. 5,641,515, 5,580,579, and 5,792,451, the disclosures of which are herein incorporated by reference in their entirety for all purposes). These oral dosage forms may also contain the following: binders, gelatin; excipients, lubricants, and/or flavoring agents. When the unit dosage form is a capsule, it may contain, in addition to the materials described above, a liquid carrier. Various other materials may be present as coatings or to otherwise modify the physical form of the dosage unit. Of course, any material used in preparing any unit dosage form should be pharmaceutically pure and substantially non-toxic in the amounts employed.
Typically, these oral formulations may contain at least about 0.1% of the lipid particles or more, although the percentage of the particles may, of course, be varied and may
conveniently be between about 1% or 2% and about 60% or 70% or more of the weight or volume of the total formulation. Naturally, the amount of particles in each therapeutically useful composition may be prepared is such a way that a suitable dosage will be obtained in any given unit dose of the compound. Factors such as solubility, bioavailability, biological half-life, route of administration, product shelf life, as well as other pharmacological considerations will be contemplated by one skilled in the art of preparing such pharmaceutical formulations, and as such, a variety of dosages and treatment regimens may be desirable.
Formulations suitable for oral administration can consist of: (a) liquid solutions, such as an effective amount of a packaged siRNA molecule(s) {e.g., a combination comprising the three siRNA molecules described in Example 1) suspended in diluents such as water, saline, or PEG 400; (b) capsules, sachets, or tablets, each containing a predetermined amount of a siRNA
molecule(s), as liquids, solids, granules, or gelatin; (c) suspensions in an appropriate liquid; and (d) suitable emulsions. Tablet forms can include one or more of lactose, sucrose, mannitol, sorbitol, calcium phosphates, corn starch, potato starch, microcrystalline cellulose, gelatin, colloidal silicon dioxide, talc, magnesium stearate, stearic acid, and other excipients, colorants, fillers, binders, diluents, buffering agents, moistening agents, preservatives, flavoring agents, dyes, disintegrating agents, and pharmaceutically compatible carriers. Lozenge forms can comprise a siRNA molecule in a flavor, e.g., sucrose, as well as pastilles comprising the therapeutic nucleic acid in an inert base, such as gelatin and glycerin or sucrose and acacia emulsions, gels, and the like containing, in addition to the siRNA molecule(s), carriers known in the art.
In another example of their use, lipid particles can be incorporated into a broad range of topical dosage forms. For instance, a suspension containing nucleic acid-lipid particles can be formulated and administered as gels, oils, emulsions, topical creams, pastes, ointments, lotions, foams, mousses, and the like.
The amount of particles administered will depend upon the ratio of siRNA molecules to lipid, the particular siRNA used, the strain of HBV being treated, the age, weight, and condition of the patient, and the judgment of the clinician, but will generally be between about
0.01 and about 50 mg per kilogram of body weight, preferably between about 0.1 and about 5 mg/kg of body weight, or about 108-1010 particles per administration (e.g., injection).
As used herein, the term "combination", means that the combined siRNA molecules are present together in the same composition of matter (e.g., dissolved together within the same solution; or present together within the same lipid particle; or present together in the same pharmaceutical formulation of lipid particles, although each lipid particle within the
pharmaceutical formulation may or may not include each different siRNA of the siRNA combination). The combined siRNA molecules usually are not covalently linked together.
In certain embodiments, a combination of siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2
(SEQ ID NO: 3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6) are used in methods of the invention.
In certain embodiments, the siRNA is administered via nucleic acid lipid particle.
In certain embodiments, with respect to methods that include the use of a cocktail of siRNAs encapsulated within lipid particles, the different siRNA molecules are co-encapsulated in the same lipid particle.
In certain embodiments, the with respect to methods that include the use of a cocktail of siRNAs encapsulated within lipid particles, each type of siRNA species present in the cocktail is encapsulated in its own particle.
In certain embodiments, the with respect to methods that include the use of a cocktail of
siRNAs encapsulated within lipid particles, some siRNA species are co-encapsulated in the same particle while other siRNA species are encapsulated in different particles.
Formulation and Administration of Two or More Agents
As described herein, methods of the invention include the administration of a lipid nanoparticle formulation, an interferon, and optionally, at least one additional therapeutic agent. It will be understood that these agents can be formulated together in a single preparation or that they can be formulated separately and, thus, administered separately, either simultaneously or sequentially. In one embodiment, when the agents are administered sequentially (e.g. at different times), the agents may be administered so that their biological effects overlap (i.e. each agent is producing a biological effect at a single given time).
In certain embodiments, the lipid nanoparticle formulation is administered first and the IFN is administered second.
In certain embodiments, the IFN is administered first and the lipid nanoparticle formulation is administered second.
In certain embodiments, the administration of the lipid nanoparticle formulation begins, e.g., about 30 weeks to about 1 week, about 24 weeks to about 6 weeks, or about 12 weeks to about 6 weeks before the administration of the IFN (i.e., the lipid nanoparticle is administered in a monotherapy phase). In certain embodiments, the lipid nanoparticle is administered two or more times prior to the administration of the IFN.
In certain embodiments, such a monotherapy phase is followed by a co-therapy phase.
In certain embodiments, the lipid nanoparticle formulation and the IFN are administered to the human concurrently (e.g., within the same week, day or hour) during the co-therapy phase. For example, in certain embodiments, the IFN and the lipid nanoparticle formulation are
administered within about 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 days of each other. In certain
embodiments, the administration of the lipid nanoparticle formulation and the IFN begin on the same day. In certain embodiments, the IFN and the lipid nanoparticle are administered within about 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 hour of each other. In certain embodiments, the administration of the IFN begins about 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 days before the
administration of the lipid nanoparticle formulation. In certain embodiments, the administration of the IFN begins about 3 days before the administration of the lipid nanoparticle formulation.
In certain embodiments, the administration of the lipid nanoparticle formulation and the IFN begin on the same day. In certain embodiments, the administration of the IFN begins about 12,
11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 hour prior to the administration of the lipid nanoparticle formulation. In certain embodiments, the lipid nanoparticle formulation and/or the IFN are administered two or more times during the co-therapy phase.
The agents can be formulated for and administered using any acceptable route of administration depending on the agent selected. For example, suitable routes include, but are not limited to, oral, sublingual, buccal, topical, transdermal, parenteral, subcutaneous, intraperitoneal, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration. In one embodiment, the small molecule agents identified herein can be administered orally. In another embodiment, the oligomeric nucleotides can be administered by injection (e.g., into a blood vessel, such as a vein), or subcutaneously. In some embodiments, a subject in need thereof is administered one or more agents orally (e.g., in pill form), and also one or more oligomeric nucleotides by injection or subcutaneously. In some embodiments, a subject in need thereof is administered the interferon and the lipid nanoparticle formulation by injection or subcutaneously.
Typically, the oligomeric nucleotides targeted to the Hepatitis B genome are
administered intravenously, for example in a lipid nanoparticle formulation; however, the present invention is not limited to intravenous formulations comprising the oligomeric nucleotides or to treatment methods wherein an oligomeric nucleotide is administered intravenously.
The agents can be individually formulated by mixing at ambient temperature at the appropriate pH, and at the desired degree of purity, with physiologically acceptable carriers, i.e., carriers that are non-toxic to recipients at the dosages and concentrations employed. The pH of the formulation depends mainly on the particular use and the concentration of compound, but may typically range anywhere from about 3 to about 8. The agents ordinarily will be stored as a solid composition, although lyophilized formulations or aqueous solutions are acceptable.
Compositions comprising the agents can be formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular human being treated, the clinical condition of the individual patient, the cause of the disorder, the site of administration, the method of administration, the scheduling of administration, and other factors known to medical
practitioners.
The agents may be administered in any convenient administrative form, e.g., tablets, powders, capsules, solutions, dispersions, suspensions, syrups, sprays, suppositories, gels, emulsions, patches, etc. Such compositions may contain components conventional in
pharmaceutical preparations, e.g., diluents, carriers, pH modifiers, sweeteners, bulking agents, and further active agents. If parenteral administration is desired, the compositions will be sterile and in a solution or suspension form suitable for injection or infusion.
Suitable carriers and excipients are well known to those skilled in the art and are
described in detail in, e.g., Ansel, Howard C, et al., Ansel's Pharmaceutical Dosage Forms and Drug Delivery Systems. Philadelphia: Lippincott, Williams & Wilkins, 2004; Gennaro, Alfonso R., et al. Remington: The Science and Practice of Pharmacy. Philadelphia: Lippincott, Williams & Wilkins, 2000; and Rowe, Raymond C. Handbook of Pharmaceutical Excipients. Chicago, Pharmaceutical Press, 2005. The formulations may also include one or more buffers, stabilizing agents, surfactants, wetting agents, lubricating agents, emulsifiers, suspending agents, preservatives, antioxidants, opaquing agents, glidants, processing aids, colorants, sweeteners, perfuming agents, flavoring agents, diluents and other known additives to provide an elegant presentation of the drug or aid in the manufacturing of the pharmaceutical product (i.e., medicament).
The agents are typically dosed at least at a level to reach the desired biological effect. Thus, an effective dosing regimen will dose at least a minimum amount that reaches the desired biological effect, or biologically effective dose, however, the dose should not be so high as to outweigh the benefit of the biological effect with unacceptable side effects. Therefore, an effective dosing regimen will dose no more than the maximum tolerated dose ("MTD"). The maximum tolerated dose is defined as the highest dose that produces an acceptable incidence of dose-limiting toxicities ("DLT"). Doses that cause an unacceptable rate of DLT are considered non-tolerated. Typically, the MTD for a particular schedule is established in phase 1 clinical trials. These are usually conducted in patients by starting at a safe starting dose of 1/10 the severe toxic dose ("STD10") in rodents (on a mg/m^ basis) and accruing patients in cohorts of three, escalating the dose according to a modified Fibonacci sequence in which ever higher escalation steps have ever decreasing relative increments (e.g., dose increases of 100%, 65%, 50%, 40%, and 30% to 35% thereafter). The dose escalation is continued in cohorts of three patients until a non-tolerated dose is reached. The next lower dose level that produces an acceptable rate of DLT is considered to be the MTD.
The amount of the agents administered will depend upon the particular agent used, the strain of HBV being treated, the age, weight, and condition of the patient, and the judgment of the clinician, but will generally be between about 0.2 to 2.0 grams per day.
Kits
One embodiment provides a kit. The kit may comprise a container comprising the combination. Suitable containers include, for example, bottles, vials, syringes, blister pack, etc. The container may be formed from a variety of materials such as glass or plastic. The container may hold the combination which is effective for treating the condition and may have a sterile
access port (for example, the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
The kit may further comprise a label or package-insert on or associated with the container. The term "package-insert" is used to refer to instructions customarily included in commercial packages of therapeutic agents that contain information about the indications, usage, dosage, administration, contraindications and/or warnings concerning the use of such therapeutic agents. In one embodiment, the label or package inserts indicates that the therapeutic agents can be used to treat a viral infection, such as Hepatitis B.
In certain embodiments, the kits are suitable for the delivery of solid oral forms of the therapeutic agents, such as tablets or capsules. Such a kit preferably includes a number of unit dosages. Such kits can include a card having the dosages oriented in the order of their intended use. An example of such a kit is a "blister pack". Blister packs are well known in the packaging industry and are widely used for packaging pharmaceutical unit dosage forms. If desired, a memory aid can be provided, for example in the form of numbers, letters, or other markings or with a calendar insert, designating the days in the treatment schedule in which the dosages can be administered.
According to another embodiment, a kit may comprise (a) a first container with one agent contained therein; and (b) a second container with a second agent contained therein.
Alternatively, or additionally, the kit may further comprise a third container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
The kit may further comprise directions for the administration of the therapeutic agents. For example, the kit may further comprise directions for the simultaneous, sequential or separate administration of the therapeutic agents to a patient in need thereof.
In certain other embodiments, the kit may comprise a container for containing separate compositions such as a divided bottle or a divided foil packet, however, the separate
compositions may also be contained within a single, undivided container. In certain
embodiments, the kit comprises directions for the administration of the separate therapeutic agents. The kit form is particularly advantageous when the separate therapeutic agents are preferably administered in different dosage forms (e.g., oral and parenteral), are administered at different dosage intervals, or when titration of the individual therapeutic agents of the combination is desired by the prescribing physician.
The ability of a combination of therapeutic agents to treat Hepatitis B may be determined using pharmacological models which are well known to the art.
The invention will now be illustrated by the following non-limiting Example.
EXAMPLE 1
In vitro combination of IFNa2a and siRNA combination Study Goal:
To determine whether a two-drug combination of siRNA molecules (a lipid nanoparticle formulation of a mixture of three siRNAs targeting the HBV genome and that inhibits production of HBV DNA, HBsAg and HBeAg), and pegylated interferon alpha 2a (IFNa2a, an antiviral cytokine that activates innate immunity pathways in hepatocytes, and is used clinically for treatment of chronic hepatitis B), is additive, synergistic or antagonistic in vitro using HBV- infected human primary hepatocytes in a cell culture model system.
Lipid Nanoparticle Formulation:
The lipid nanoparticle formulation comprises a mixture of three siRNAs targeting the HBV genome. Specifically, the following lipid nanoparticle (LNP) formulation was used to deliver the HBV siRNAs in the experiments reported herein. This formulation is referred to as siRNA-NP2 throughout this Example. The values shown in the table are mole percentages. The abbreviation DSPC means distearoylphosphatidylcholine.
The cationic lipid had the following structure:
A mixture of three siRNAs targeting the HBV genome were used in this experiment. The sequences of the three siRNAs are shown below.
Underline = unlocked nucleobase analogue (UNA) moiety
Results and Conclusion:
IFNa2a (concentration range of 25.0 IU/mL to 0.31 IU/mL in a 3-fold dilution series and 5 point titration) was tested in combination with the siRNA-NP2 discussed above (concentration range of 5.0 ng/mL to 0.06 ng/mL in a 3-fold dilution series and 5 point titration). The average % inhibition in HBV DNA, HBsAg and HBeAg, and standard deviations of 3 replicates
observed either with IFNa2a or siRNA-NP2 treatments alone or in combination are shown in Tables, 1, 2, and 3 as indicated below. The EC50 values of IFNa2a and siRNA-NP2 were determined in an earlier experiment and are shown in Table 4; some variance was observed from different lots of PHH cells.
When the observed values of a two-inhibitor combination were compared to what is expected from additive interaction for the above concentration range, the combinations were found to be synergistic or additive depending on the measured end point (HBV DNA, HBsAg or HBeAg), (Table 4) as per MacSynergy II analysis and using the interpretive criteria described above by Prichard and Shipman (1992). No significant inhibition of cell viability was observed by microscopy or CellTiter-Glo assay.
Table 1: Effect on HBV DNA in In Vitro Combination of IFNa2a and siRNA-NP2
HBsAg Minor
IFNa2a siRNA-NP2 6.19 <1.37 28.92 6.58 0 0
Synergy
HBeAg IFNa2a siRNA-NP2 10.74 2.61 3.46 0.79 -3.62 -0.82 Additivity at 99.9% confidence interval
#determined in an earlier separate experiment
All publications, patents, and patent documents are incorporated by reference herein, as though individually incorporated by reference. The invention has been described with reference to various specific and preferred embodiments and techniques. However, it should be
understood that many variations and modifications may be made while remaining within the spirit and scope of the invention.
Claims
1. A method for treating hepatitis B in a human, comprising administering to the human:
1) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
2) an interferon (IFN).
2. The method of claim 1, wherein the IFN is IFN alpha (IFN-a), IFN beta (IFN-β) or IFN lambda (IFN- λ).
3. The method of claim 2, wherein the IFN is IFN-a.
4. The method of claim 3, wherein the IFN-a is pegylated IFN-a2a or peglyated IFN-a2b.
5. The method of any one of claims 1-4, wherein the lipid nanoparticle formulation further comprises a cationic lipid and a non-cationic lipid.
6. The method of claim 5, wherein the cationic lipid is selected from the group consisting of l,2-dilinoleyloxy-N,N-dimethylaminopropane (DLinDMA), l,2-dilinolenyloxy-N,N- dimethylaminopropane (DLenDMA), l,2-di-Y-linolenyloxy-N,N-dimethylaminopropane (γ- DLenDMA; Compound (15)) , 3-((6Z,9Z,28Z,31Z)-heptatriaconta-6,9,28,31-tetraen-19-yloxy)- N,N-dimethylpropan-l -amine (DLin-MP-DMA; Compound (8)), (6Z,9Z,28Z,31Z)- heptatriaconta-6,9,28,31-tetraen-19-yl 4-(dimethylamino)butanoate) (Compound (7)), (6Z, 16Z)- 12-((Z)-dec-4-enyl)docosa-6,16-dien-l 1-yl 5-(dimethylamino)pentanoate (Compound (13)), a salt thereof, and a mixture thereof.
8. The method of any one of claims 5-7, wherein the non-cationic lipid is cholesterol or a derivative thereof.
9. The method of any one of claims 5-7, wherein the non-cationic lipid is a phospholipid.
10. The method of any one of claims 5-7, wherein the non-cationic lipid is a mixture of a phospholipid and cholesterol or a derivative thereof.
11. The method of claim 9 or 10, wherein the phospholipid is selected from the group consisting of dipalmitoylphosphatidylcholine (DPPC), distearoylphosphatidylcholine (DSPC), and a mixture thereof.
12. The method of claim 11, wherein the phospholipid is DSPC.
13. The method of any one of claims 5-12, wherein the lipid formulation further comprises a conjugated lipid that inhibits aggregation of particles.
14. The method of claim 13, wherein the conjugated lipid that inhibits aggregation of particles is a polyethyleneglycol (PEG)-lipid conjugate.
15. The method of claim 14, wherein the PEG-lipid conjugate is selected from the group consisting of a PEG-diacylglycerol (PEG-DAG) conjugate, a PEG-dialkyloxypropyl (PEG- DAA) conjugate, a PEG-phospholipid conjugate, a PEG-ceramide (PEG-Cer) conjugate, a PEG- dimyristyloxypropyl (PEG-DMA) conjugate and a mixture thereof.
16. The method of claim 14, wherein the PEG-lipid conjugate is a PEG-C-DMA conjugate.
17. The method of any one of claims 5-16, wherein the cationic lipid comprises from about 48 mol % to about 62 mol % of the total lipid present in each particle within the formulation.
18. The method of any one of claims 10-17, comprising a phospholipid and cholesterol or cholesterol derivative, wherein the phospholipid comprises from about 7 mol % to about 17 mol % of the total lipid present in each particle within the formulation and the cholesterol or derivative thereof comprises from about 25 mol % to about 40 mol % of the total lipid present in each particle within the formulation.
19. The method of any one of claims 13-18, wherein the conjugated lipid that inhibits aggregation of particles comprises from about 0.5 mol % to about 3 mol % of the total lipid present in each particle within the formulation.
20. The method of any one of claims 1-19, wherein the lipid nanoparticle formulation is administered via injection.
21. The method of any one of claims 1-20, wherein the IFN is administered via injection.
22. The method of any one of claims 1-21, wherein the lipid nanoparticle formulation and the IFN are administered separately.
23. The method of any one of claims 1-22, wherein the lipid nanoparticle formulation and the IFN are administered sequentially.
24. The method of claim 23, wherein the lipid nanoparticle formulation is administered first and the IFN is administered second.
25. The method of claim 23, wherein the IFN is administered first and the lipid nanoparticle formulation is administered second.
26. The method of any one of claims 1-25, wherein the lipid nanoparticle formulation and the IFN are administered concurrently.
27. The method of any one of claims 1-21, wherein a pharmaceutical composition comprising the lipid nanoparticle formulation, the IFN and a pharmaceutically acceptable carrier is administered to the human.
28. The method of any one of claims 1-27, further comprising administering at least one additional therapeutic agent.
29. The method of claim 28, wherein the at least one additional therapeutic agent is selected from the group consisting of:
(A) an agent that controls viral replication;
(B) an agent that reduces viral Ags;
(C) an immune enhancer; and
(D) an immune stimulant.
30. The method of claim 29, wherein the agent that controls viral replication is a reverse transcriptase inhibitor, a capsid inhibitor, a cccDNA inhibitor or an entry inhibitor.
31. The method of claim 30, wherein the agent that controls viral replication is a reverse transcriptase inhibitor.
32. The method of claim 30, wherein the agent that controls viral replication is a capsid inhibitor.
33. The method of claim 30, wherein the agent that controls viral replication is a cccDNA inhibitor.
34. The method of claim 30, wherein the agent that controls viral replication is an entry inhibitor.
35. The method of claim 29, wherein the agent that controls viral replication is entecavir, clevudine, telbivudine, lamivudine, adefovir, and tenofovir, tenofovir disoproxil, tenofovir alafenamide, tenofovir disoproxil fumarate, adefovir dipivoxil, (lR,2R,3R,5R)-3-(6-amino-9H- 9-purinyl)-2-fluoro-5-(hydroxymethyl)-4-methylenecyclopentan- 1 -ol, emtricitabine, abacavir, elvucitabine, ganciclovir, lobucavir, famciclovir, penciclovir, amdoxovir or CMX157 (tenofovir exalidex).
36. The method of any one of claims 29-35, wherein the agent that reduces viral Ags is an siRNA that targets a portion of the HBV genome.
37. The method of any one of claims 29-35, wherein the agent that reduces viral Ags is a sAg secretion inhibitor.
38. The method of any one of claims 29-35, wherein the agent that reduces viral Ags is an anti-HBsAg agent.
The method of any one of claims 29-38, wherein the immune enhancer is a checkpoint
inhibitor.
40. The method of any one of claims 29-38, wherein the immune enhancer is an inhibitor of PD-L1.
41. The method of any one of claims 29-38, wherein the immune enhancer is an anti-PD-1 mAb, an anti-PD-Ll mAb, an anti-PD-L2 mAb, an anti-CTLA4 mAb, an anti-VISTA mAb, an anti-LAG3 mAb, an anti-TEVB mAb, a peptidomimetic or a small molecule compound.
42. The method of any one of claims 29-41, wherein the immune stimulant is an anti-HBV vaccine, an interferon, a RIG-I agonist, a STING agonist, a TLR9 agonist, a TLR7 agonist, a TLR8 agonist, a TLR3 agonist, IL-7, IL-2, an OX-40 agonist, or an anti-GITR agonist.
43. A lipid nanoparticle formulation in combination with an interferon for the prophylactic or therapeutic treatment of hepatitis B, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
44. The use a lipid nanoparticle formulation in combination with an interferon (IFN) for the preparation of a medicament for treating hepatitis B in a human, wherein the lipid nanoparticle formulation comprises siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6).
45. A composition comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: 1 and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID NO:5 and 6); and
(b) an interferon.
46. The composition of claim 45, which is a pharmaceutical composition further comprising a pharmaceutically acceptable carrier.
47. A kit comprising:
(a) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO: 3 and 4) and siRNA 3 (SEQ ID NO: 5 and 6);
(b) an interferon; and
(c) instructions for the simultaneous, sequential or separate administration of the lipid nanoparticle formulation and the interferon.
A method for treating hepatitis D in a human, comprising administering to the human:
1) a lipid nanoparticle formulation comprising siRNA 1 (SEQ ID NO: l and 2), siRNA 2 (SEQ ID NO:3 and 4) and siRNA 3 (SEQ ID N0 5 and 6); and
2) an interferon (IFN).
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762520981P | 2017-06-16 | 2017-06-16 | |
| US62/520,981 | 2017-06-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018232330A1 true WO2018232330A1 (en) | 2018-12-20 |
Family
ID=64660917
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2018/037890 WO2018232330A1 (en) | 2017-06-16 | 2018-06-15 | Therapeutic compositions and methods for treating hepatitis b |
Country Status (3)
| Country | Link |
|---|---|
| AR (1) | AR112166A1 (en) |
| TW (1) | TW201919653A (en) |
| WO (1) | WO2018232330A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021236389A3 (en) * | 2020-05-16 | 2022-01-20 | Viratides, Llc | Treatment of known and unknown viral infection with lipid agents |
| CN114173784A (en) * | 2019-07-18 | 2022-03-11 | 埃尼奥制药公司 | Method for reducing side effects of interferon |
| US11504421B2 (en) | 2017-05-08 | 2022-11-22 | Gritstone Bio, Inc. | Alphavirus neoantigen vectors |
| US11591619B2 (en) | 2019-05-30 | 2023-02-28 | Gritstone Bio, Inc. | Modified adenoviruses |
| US11771747B2 (en) | 2020-08-06 | 2023-10-03 | Gritstone Bio, Inc. | Multiepitope vaccine cassettes |
| WO2024214041A1 (en) * | 2023-04-12 | 2024-10-17 | Glaxosmithkline Intellectual Property Development Limited | Methods of treating chronic hepatitis b |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7067249B2 (en) * | 2003-05-19 | 2006-06-27 | The University Of Hong Kong | Inhibition of hepatitis B virus (HBV) replication by RNA interference |
| US20080317714A1 (en) * | 2005-08-15 | 2008-12-25 | Janssen Henricus Leonardus Ant | Method of Treating Hepatitis B Viral Infection |
| US20100063132A1 (en) * | 2005-03-09 | 2010-03-11 | Mogam Biotechnology Research Institute | Small interfering rna and pharmaceutical composition for treatment of hepatitis b comprising the same |
| WO2016054421A1 (en) * | 2014-10-02 | 2016-04-07 | Protiva Biotherapeutics, Inc | Compositions and methods for silencing hepatitis b virus gene expression |
| US9518272B2 (en) * | 2010-06-30 | 2016-12-13 | Protiva Biotherapeutics, Inc. | Non-liposomal systems for nucleic acid delivery |
-
2018
- 2018-06-15 WO PCT/US2018/037890 patent/WO2018232330A1/en active Application Filing
- 2018-06-15 TW TW107120864A patent/TW201919653A/en unknown
- 2018-06-15 AR ARP180101692A patent/AR112166A1/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7067249B2 (en) * | 2003-05-19 | 2006-06-27 | The University Of Hong Kong | Inhibition of hepatitis B virus (HBV) replication by RNA interference |
| US20100063132A1 (en) * | 2005-03-09 | 2010-03-11 | Mogam Biotechnology Research Institute | Small interfering rna and pharmaceutical composition for treatment of hepatitis b comprising the same |
| US20080317714A1 (en) * | 2005-08-15 | 2008-12-25 | Janssen Henricus Leonardus Ant | Method of Treating Hepatitis B Viral Infection |
| US9518272B2 (en) * | 2010-06-30 | 2016-12-13 | Protiva Biotherapeutics, Inc. | Non-liposomal systems for nucleic acid delivery |
| WO2016054421A1 (en) * | 2014-10-02 | 2016-04-07 | Protiva Biotherapeutics, Inc | Compositions and methods for silencing hepatitis b virus gene expression |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11504421B2 (en) | 2017-05-08 | 2022-11-22 | Gritstone Bio, Inc. | Alphavirus neoantigen vectors |
| US11510973B2 (en) | 2017-05-08 | 2022-11-29 | Gritstone Bio, Inc. | Alphavirus antigen vectors |
| US12109257B2 (en) | 2017-05-08 | 2024-10-08 | Gritstone Bio, Inc. | Alphavirus neoantigen vectors |
| US11591619B2 (en) | 2019-05-30 | 2023-02-28 | Gritstone Bio, Inc. | Modified adenoviruses |
| US12098383B2 (en) | 2019-05-30 | 2024-09-24 | Gritstone Bio, Inc. | Modified adenoviruses |
| CN114173784A (en) * | 2019-07-18 | 2022-03-11 | 埃尼奥制药公司 | Method for reducing side effects of interferon |
| CN114173784B (en) * | 2019-07-18 | 2023-12-01 | 埃尼奥制药公司 | Method for reducing side effects of interferon |
| WO2021236389A3 (en) * | 2020-05-16 | 2022-01-20 | Viratides, Llc | Treatment of known and unknown viral infection with lipid agents |
| US11771747B2 (en) | 2020-08-06 | 2023-10-03 | Gritstone Bio, Inc. | Multiepitope vaccine cassettes |
| WO2024214041A1 (en) * | 2023-04-12 | 2024-10-17 | Glaxosmithkline Intellectual Property Development Limited | Methods of treating chronic hepatitis b |
Also Published As
| Publication number | Publication date |
|---|---|
| AR112166A1 (en) | 2019-09-25 |
| TW201919653A (en) | 2019-06-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10626393B2 (en) | Delivering CRISPR therapeutics with lipid nanoparticles | |
| US10415037B2 (en) | Compositions and methods for silencing hepatitis B virus gene expression | |
| US20180245074A1 (en) | Treating hepatitis b virus infection using crispr | |
| US11904052B2 (en) | Methods for ameliorating infusion reactions | |
| WO2017019891A2 (en) | Compositions and methods for silencing hepatitis b virus gene expression | |
| US20180245077A1 (en) | Compositions and methods for treating hypertriglyceridemia | |
| WO2016071857A1 (en) | Compositions and methods for silencing ebola virus expression | |
| WO2018232330A1 (en) | Therapeutic compositions and methods for treating hepatitis b | |
| EP3634431A1 (en) | Therapeutic compositions and methods for treating hepatitis b | |
| WO2016183366A2 (en) | Compositions and methods for silencing expression of hepatitis d virus rna | |
| US20240050463A1 (en) | Therapeutic compositions and methods for treating hepatitis b | |
| TW202435851A (en) | Therapeutic compositions and methods for treating hepatitis b |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18816789 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205N DATED 06/02/2020) |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 18816789 Country of ref document: EP Kind code of ref document: A1 |