Red phosphorus
 A sample of red phosphorus
| |
| Names | |
|---|---|
| Other names
Amorphous phosphorus
| |
| Identifiers | |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| P | |
| Molar mass | 30.974 g·mol−1 |
| Density | 2.34 g/cm3 |
| Melting point | 590 °C (1,094 °F; 863 K) |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Red phosphorus is an allotrope of phosphorus. It is an amorphous polymeric red solid that is stable in air. It can be easily converted from white phosphorus under light or heating. It finds applications as matches and fire retardants. It was discovered in 1847 by Anton von Schrötter.[1]
Structure
[edit]Red phosphorus is an amorphous form of phosphorus. Crystalline forms of red phosphorus include Hittorf's phosphorus and fibrous red phosphorus. The structure of red phosphorus contains the fragments illustrated below:[2][3]


Preparation
[edit]One method of preparing red phosphorus involves heating white phosphorus in an inert atmosphere like nitrogen or carbon dioxide, with iodine as catalyst.[4]
Another theoretically possible method of red phosphorus production is via light irradiation of white phosphorus.[5] However, it has not been used industrially, likely due to the suspicious quality and unidentified structure the product.[6][better source needed]
Properties
[edit]Under standard conditions, red phosphorus is more stable than white phosphorus, but less stable than the thermodynamically stable black phosphorus. The standard enthalpy of formation of red phosphorus is −17.6 kJ/mol.[3] Red phosphorus is kinetically most stable.
Being polymeric, red phosphorus is insoluble in solvents. It shows semiconductor properties.[7]
Due to such a kinetic stability, red phosphorus doesn't spontaneously ignite in air. It doesn't disproportionate in the presence of alkali, and is less reactive towards halogens, sulfur, and metals compared with white phosphorus.[7]
Applications
[edit]Red phosphorus can be used as a flame retardant in resins. Its mechanism of action involves the formation of polyphosphoric acid (the hydrogen atoms are from the resin) and char, which prevents flame propagation.[8]
However, for electronic/electrical systems, red phosphorus flame retardant has been effectively banned by major OEMs due to its tendency to induce premature failures.[9] One persistent problem is that red phosphorus in epoxy molding compounds induces elevated leakage current in semiconductor devices.[10] Another problem was acceleration of hydrolysis reactions in PBT insulating material.[11]
Red phosphorus is used, along with abrasives, on the strike pads of modern safety matches. The match head, containing potassium chlorate, will ignite upon friction with the strike pad.[12] It should be noted, however, that the red color of the matchhead is due to addition of red dyes, and has nothing to do with red phosphorus content.[13]
Red phosphorus reacts with bromine and iodine to form phosphorus tribromide[14][15] and phosphorus triiodide. Both are useful as halogenation agents, like replacing the hydroxyl group of alcohols. Phosphorus triiodide can also be used to produce hydroiodic acid after hydrolysis. This reaction is notable in the illicit production of methamphetamine and Krokodil, where hydrogen iodide acts as a reducing agent.[16]
Red phosphorus is often used to prepare chemicals where the P-P bond is retained. Upon room temperature action with sodium chlorite, Na2H2P2O6 is formed.[17]
Red phosphorus can be used as an elemental photocatalyst for hydrogen formation from the water.[18][7] It has also been researched as a sodium ion battery anode.[19][2]
Violet or Hittorf's phosphorus
[edit]Hittorf's phosphorus, or violet phosphorus, is one of the crystalline forms of red phosphorus.[20][7] It adopts the following structure:
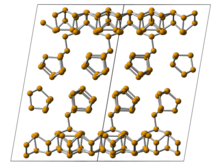
Violet phosphorus can be prepared by sublimation of red phosphorus in a vacuum, in the presence of an iodine catalyst.[7]
It is chemically similar to red phosphorus. There are, however, subtle differences. Violet phosphorus ignites upon impact in air, while red phosphorus is impact stable. Violet phosphorus doesn't ignite in the presence of air upon room temperature contact with bromine, unlike red phosphorus.[21][better source needed] It should be noted, however, that the reaction of red phosphorus and bromine alone does not generate a flame.
Fibrous red phosphorus
[edit]Fibrous red phosphorus is another crystalline form of red phosphorus.[7] It is obtained along with violet phosphorus when red phosphorus is sublimed in vacuum in the presence of iodine.[22]
It is structurally similar to violet phosphorus. However, in fibrous red phosphorus, phosphorus chains lie parallel instead of orthogonal, unlike violet phosphorus. Such a structure is depicted below:[23][7]

Fibrous red phosphorus, similar to red phosphorus, displays activity as a photocatalyst.[22][24]
See also
[edit]References
[edit]- ^ Kohn, Moritz (November 1944). "The discovery of red phosphorus (1847) by Anton von Schrötter (1802-1875)". Journal of Chemical Education. 21 (11): 522. Bibcode:1944JChEd..21..522K. doi:10.1021/ed021p522. ISSN 0021-9584.
- ^ a b Zhou, Yuxing; Elliott, Stephen R.; Deringer, Volker L. (2023-06-12). "Structure and Bonding in Amorphous Red Phosphorus**". Angewandte Chemie International Edition. 62 (24): e202216658. doi:10.1002/anie.202216658. ISSN 1433-7851. PMC 10952455. PMID 36916828.
- ^ a b Housecroft, C. E.; Sharpe, A. G. (2004). Inorganic Chemistry (2nd ed.). Prentice Hall. p. 392. ISBN 978-0-13-039913-7.
- ^ "Red Phosphorus - Structure, Properties, Preparation and Applications". VEDANTU. Retrieved 2024-08-12.
- ^ "White phosphorus". American Chemical Society. Retrieved 2024-08-12.
- ^ Extractions&Ire (2018-07-25). Turning White Phosphorus into Red using Sunlight. Retrieved 2024-08-12 – via YouTube.
- ^ a b c d e f g Housecroft, Catherine E. (2018). Inorganic chemistry (Fifth ed.). Harlow, England ; New York: Pearson. p. 511. ISBN 978-1-292-13414-7.
- ^ "Features of red phosphorus fire retardants | Product descriptions|RIN KAGAKU KOGYO". www.rinka.co.jp. Retrieved 2024-08-15.
- ^ "Red Phosphorus Reliability Alert" (PDF). Archived from the original (PDF) on 2018-01-02. Retrieved 2018-01-01.
- ^ Craig Hillman, Red Phosphorus Induced Failures in Encapsulated Circuits, https://www.dfrsolutions.com/hubfs/Resources/services/Red-Phosphorus-Induced-Failures-in-Encapsulated-Circuits.pdf?t=1513022462214
- ^ Dock Brown, The Return of the Red Retardant, SMTAI 2015, https://www.dfrsolutions.com/hubfs/Resources/services/The-Return-of-the-Red-Retardant.pdf?t=1513022462214
- ^ "Fire". Archived from the original on 4 November 2011. Retrieved 19 November 2011.
- ^ "How Do Safety Matches Work?". 2020-05-27. Retrieved 2024-08-12.
- ^ J. F. Gay, R. N. Maxson "Phosphorus(III) Bromide" Inorganic Syntheses, 1947, vol. 2, 147ff. doi:10.1002/9780470132333.ch43
- ^ Burton, T. M.; Degerping, E. F. (1940). "The Preparation of Acetyl Bromide". Journal of the American Chemical Society. 62 (1): 227. doi:10.1021/ja01858a502.
- ^ Skinner, Harry F. (1990). "Methamphetamine synthesis via hydriodic acid/Red phosphorus reduction of ephedrine". Forensic Science International. 48 (2): 123–134. doi:10.1016/0379-0738(90)90104-7.
- ^ Phosphorus: Chemistry, Biochemistry and Technology, Sixth Edition, 2013, D.E.C. Corbridge, CRC Pres, Taylor Francis Group, ISBN 978-1-4398-4088-7
- ^ Applied Catalysis B: Environmental, 2012, 111–112, 409–414.
- ^ Liu, Yihang; Liu, Qingzhou; Jian, Cheng; Cui, Dingzhou; Chen, Mingrui; Li, Zhen; Li, Teng; Nilges, Tom; He, Kai; Jia, Zheng; Zhou, Chongwu (2020-05-20). "Red-phosphorus-impregnated carbon nanofibers for sodium-ion batteries and liquefaction of red phosphorus". Nature Communications. 11 (1): 2520. Bibcode:2020NatCo..11.2520L. doi:10.1038/s41467-020-16077-z. ISSN 2041-1723. PMC 7239945. PMID 32433557.
- ^ Liu, Cheng; Wang, Yinghao; Sun, Jie; Chen, Aibing (April 2020). "A Review on Applications of Layered Phosphorus in Energy Storage". Transactions of Tianjin University. 26 (2): 104–126. Bibcode:2020TrTU...26..104L. doi:10.1007/s12209-019-00230-x. ISSN 1006-4982.
- ^ ChemicalForce (2021-12-07). Violet violent Phosphorus EXPLODES on impact!. Retrieved 2024-08-12 – via YouTube.
- ^ a b Athira, T. K.; Roshith, M.; Satheesh Babu, T. G.; Ravi Kumar, Darbha V. (2021-01-15). "Fibrous red phosphorus as a non-metallic photocatalyst for the effective reduction of Cr(VI) under direct sunlight". Materials Letters. 283: 128750. Bibcode:2021MatL..28328750A. doi:10.1016/j.matlet.2020.128750. ISSN 0167-577X.
- ^ Ruck, Michael; Hoppe, Diana; Wahl, Bernhard; Simon, Paul; Wang, Yuekui; Seifert, Gotthard (2005-11-25). "Fibrous Red Phosphorus". Angewandte Chemie International Edition. 44 (46): 7616–7619. doi:10.1002/anie.200503017. ISSN 1433-7851. PMID 16245382.
- ^ Hu, Zhuofeng; Guo, Weiqing (May 2021). "Fibrous Phase Red Phosphorene as a New Photocatalyst for Carbon Dioxide Reduction and Hydrogen Evolution". Small. 17 (19): e2008004. doi:10.1002/smll.202008004. ISSN 1613-6810. PMID 33792191.

