US20200308023A1 - Salt recovery solution and processes of use thereof - Google Patents
Salt recovery solution and processes of use thereof Download PDFInfo
- Publication number
- US20200308023A1 US20200308023A1 US16/753,263 US201816753263A US2020308023A1 US 20200308023 A1 US20200308023 A1 US 20200308023A1 US 201816753263 A US201816753263 A US 201816753263A US 2020308023 A1 US2020308023 A1 US 2020308023A1
- Authority
- US
- United States
- Prior art keywords
- salt
- solution
- salt recovery
- recovery solution
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 150000003839 salts Chemical class 0.000 title claims abstract description 350
- 238000011084 recovery Methods 0.000 title claims abstract description 273
- 238000000034 method Methods 0.000 title claims abstract description 85
- 230000008569 process Effects 0.000 title claims abstract description 81
- 239000000243 solution Substances 0.000 claims abstract description 306
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 claims abstract description 136
- 239000012267 brine Substances 0.000 claims abstract description 133
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 112
- 150000001875 compounds Chemical class 0.000 claims abstract description 36
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims abstract description 29
- 150000003512 tertiary amines Chemical class 0.000 claims abstract description 28
- 239000007864 aqueous solution Substances 0.000 claims abstract description 27
- 239000012141 concentrate Substances 0.000 claims abstract description 7
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 146
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid group Chemical group C(CC(O)(C(=O)O)CC(=O)O)(=O)O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 50
- 125000006701 (C1-C7) alkyl group Chemical group 0.000 claims description 27
- 125000001475 halogen functional group Chemical group 0.000 claims description 25
- -1 monocyclic ketone Chemical class 0.000 claims description 19
- 230000003204 osmotic effect Effects 0.000 claims description 19
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims description 17
- 125000002950 monocyclic group Chemical group 0.000 claims description 17
- 239000000654 additive Substances 0.000 claims description 13
- 230000000996 additive effect Effects 0.000 claims description 13
- 239000012528 membrane Substances 0.000 claims description 12
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 claims description 10
- 239000007788 liquid Substances 0.000 claims description 9
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 8
- 125000005842 heteroatom Chemical group 0.000 claims description 7
- 125000000217 alkyl group Chemical group 0.000 claims description 6
- 229910052717 sulfur Inorganic materials 0.000 claims description 6
- HVCNXQOWACZAFN-UHFFFAOYSA-N 4-ethylmorpholine Chemical compound CCN1CCOCC1 HVCNXQOWACZAFN-UHFFFAOYSA-N 0.000 claims description 5
- 238000000926 separation method Methods 0.000 claims description 5
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 claims description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 3
- 125000003118 aryl group Chemical group 0.000 claims description 3
- 229910052799 carbon Inorganic materials 0.000 claims description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 3
- 125000001424 substituent group Chemical group 0.000 claims description 3
- ONQBOTKLCMXPOF-UHFFFAOYSA-N 1-ethylpyrrolidine Chemical compound CCN1CCCC1 ONQBOTKLCMXPOF-UHFFFAOYSA-N 0.000 claims description 2
- 125000001931 aliphatic group Chemical group 0.000 claims description 2
- 238000010924 continuous production Methods 0.000 claims description 2
- 239000013535 sea water Substances 0.000 claims description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 101
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 60
- 239000011780 sodium chloride Substances 0.000 description 50
- 238000010521 absorption reaction Methods 0.000 description 29
- 150000002576 ketones Chemical group 0.000 description 18
- 239000012492 regenerant Substances 0.000 description 17
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 16
- 150000001412 amines Chemical class 0.000 description 14
- 229960004106 citric acid Drugs 0.000 description 13
- 239000012266 salt solution Substances 0.000 description 10
- 239000007789 gas Substances 0.000 description 9
- 0 [1*]C([2*])=O Chemical compound [1*]C([2*])=O 0.000 description 8
- AKGGYBADQZYZPD-UHFFFAOYSA-N benzylacetone Chemical compound CC(=O)CCC1=CC=CC=C1 AKGGYBADQZYZPD-UHFFFAOYSA-N 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 7
- 238000002474 experimental method Methods 0.000 description 7
- 239000000463 material Substances 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 230000001172 regenerating effect Effects 0.000 description 7
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 6
- 229920006395 saturated elastomer Polymers 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 239000002351 wastewater Substances 0.000 description 6
- QNXSIUBBGPHDDE-UHFFFAOYSA-N indan-1-one Chemical group C1=CC=C2C(=O)CCC2=C1 QNXSIUBBGPHDDE-UHFFFAOYSA-N 0.000 description 5
- 239000012071 phase Substances 0.000 description 5
- UGSGKYKRLRQHBS-UHFFFAOYSA-N 3-butyl-4-hydroxy-1-methylquinolin-2-one Chemical compound C1=CC=C2N(C)C(=O)C(CCCC)=C(O)C2=C1 UGSGKYKRLRQHBS-UHFFFAOYSA-N 0.000 description 4
- RUORWXQKVXTQJJ-UHFFFAOYSA-N 4-methyl-2,3-dihydroinden-1-one Chemical compound CC1=CC=CC2=C1CCC2=O RUORWXQKVXTQJJ-UHFFFAOYSA-N 0.000 description 4
- VGVHNLRUAMRIEW-UHFFFAOYSA-N 4-methylcyclohexan-1-one Chemical compound CC1CCC(=O)CC1 VGVHNLRUAMRIEW-UHFFFAOYSA-N 0.000 description 4
- 238000011088 calibration curve Methods 0.000 description 4
- 125000004432 carbon atom Chemical group C* 0.000 description 4
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 description 4
- 239000008346 aqueous phase Substances 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 238000004364 calculation method Methods 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 238000001704 evaporation Methods 0.000 description 3
- YLQWCDOCJODRMT-UHFFFAOYSA-N fluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3C2=C1 YLQWCDOCJODRMT-UHFFFAOYSA-N 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 125000004430 oxygen atom Chemical group O* 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 230000008929 regeneration Effects 0.000 description 3
- 238000011069 regeneration method Methods 0.000 description 3
- 238000001223 reverse osmosis Methods 0.000 description 3
- XZPVPNZTYPUODG-UHFFFAOYSA-M sodium;chloride;dihydrate Chemical compound O.O.[Na+].[Cl-] XZPVPNZTYPUODG-UHFFFAOYSA-M 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 125000004434 sulfur atom Chemical group 0.000 description 3
- APAZZDBYJISGLX-UHFFFAOYSA-N 1-bromo-4-phenylbutan-2-one Chemical compound BrCC(=O)CCC1=CC=CC=C1 APAZZDBYJISGLX-UHFFFAOYSA-N 0.000 description 2
- YOXUOHDHFCBGHY-UHFFFAOYSA-N 1-bromofluoren-9-one Chemical compound C12=CC=CC=C2C(=O)C2=C1C=CC=C2Br YOXUOHDHFCBGHY-UHFFFAOYSA-N 0.000 description 2
- POMQQLMTQXTWLP-UHFFFAOYSA-N 1-methylfluoren-9-one Chemical compound C12=CC=CC=C2C(=O)C2=C1C=CC=C2C POMQQLMTQXTWLP-UHFFFAOYSA-N 0.000 description 2
- CHSCXMSQYNEWEL-UHFFFAOYSA-N 2,2-bis(methylsulfanyl)-3H-inden-1-one Chemical compound C1=CC=C2C(=O)C(SC)(SC)CC2=C1 CHSCXMSQYNEWEL-UHFFFAOYSA-N 0.000 description 2
- GBKIMWUIKCBINN-UHFFFAOYSA-N 2,3,7-trinitrofluoren-9-one Chemical compound [O-][N+](=O)C1=C([N+]([O-])=O)C=C2C3=CC=C([N+](=O)[O-])C=C3C(=O)C2=C1 GBKIMWUIKCBINN-UHFFFAOYSA-N 0.000 description 2
- LSWRRPBOJDRHSV-UHFFFAOYSA-N 2,6-dimethyl-1h-quinolin-4-one Chemical compound N1C(C)=CC(=O)C2=CC(C)=CC=C21 LSWRRPBOJDRHSV-UHFFFAOYSA-N 0.000 description 2
- XHBAHYMCKJTKJV-UHFFFAOYSA-N 2,6-dinitrofluoren-9-one Chemical compound C1=C([N+]([O-])=O)C=C2C3=CC=C([N+](=O)[O-])C=C3C(=O)C2=C1 XHBAHYMCKJTKJV-UHFFFAOYSA-N 0.000 description 2
- CWGRCRZFJOXQFV-UHFFFAOYSA-N 2,7-dibromofluoren-9-one Chemical compound C1=C(Br)C=C2C(=O)C3=CC(Br)=CC=C3C2=C1 CWGRCRZFJOXQFV-UHFFFAOYSA-N 0.000 description 2
- HEYWYQQFXVEUSH-UHFFFAOYSA-N 2,7-dichlorofluoren-9-one Chemical compound C1=C(Cl)C=C2C(=O)C3=CC(Cl)=CC=C3C2=C1 HEYWYQQFXVEUSH-UHFFFAOYSA-N 0.000 description 2
- HDVGAFBXTXDYIB-UHFFFAOYSA-N 2,7-dinitrofluoren-9-one Chemical compound C1=C([N+]([O-])=O)C=C2C(=O)C3=CC([N+](=O)[O-])=CC=C3C2=C1 HDVGAFBXTXDYIB-UHFFFAOYSA-N 0.000 description 2
- WYJOVVXUZNRJQY-UHFFFAOYSA-N 2-Acetylthiophene Chemical compound CC(=O)C1=CC=CS1 WYJOVVXUZNRJQY-UHFFFAOYSA-N 0.000 description 2
- GQWWGRUJOCIUKI-UHFFFAOYSA-N 2-[3-(2-methyl-1-oxopyrrolo[1,2-a]pyrazin-3-yl)propyl]guanidine Chemical compound O=C1N(C)C(CCCN=C(N)N)=CN2C=CC=C21 GQWWGRUJOCIUKI-UHFFFAOYSA-N 0.000 description 2
- IEMMBWWQXVXBEU-UHFFFAOYSA-N 2-acetylfuran Chemical compound CC(=O)C1=CC=CO1 IEMMBWWQXVXBEU-UHFFFAOYSA-N 0.000 description 2
- IGJQUJNPMOYEJY-UHFFFAOYSA-N 2-acetylpyrrole Chemical compound CC(=O)C1=CC=CN1 IGJQUJNPMOYEJY-UHFFFAOYSA-N 0.000 description 2
- UXVCSPSWUNMPMT-UHFFFAOYSA-N 2-bromo-2,3-dihydroinden-1-one Chemical compound C1=CC=C2C(=O)C(Br)CC2=C1 UXVCSPSWUNMPMT-UHFFFAOYSA-N 0.000 description 2
- MTCARZDHUIEYMB-UHFFFAOYSA-N 2-bromofluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(Br)=CC=C3C2=C1 MTCARZDHUIEYMB-UHFFFAOYSA-N 0.000 description 2
- CNCFLCLXJBPMIR-UHFFFAOYSA-N 2-fluorofluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(F)=CC=C3C2=C1 CNCFLCLXJBPMIR-UHFFFAOYSA-N 0.000 description 2
- GXUBPHMYNSICJC-UHFFFAOYSA-N 2-hydroxyfluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(O)=CC=C3C2=C1 GXUBPHMYNSICJC-UHFFFAOYSA-N 0.000 description 2
- BEKNOGMQVKBMQN-UHFFFAOYSA-N 2-methyl-2,3-dihydroinden-1-one Chemical compound C1=CC=C2C(=O)C(C)CC2=C1 BEKNOGMQVKBMQN-UHFFFAOYSA-N 0.000 description 2
- AJEAHBZZHSLIQP-UHFFFAOYSA-N 2-nitrofluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC([N+](=O)[O-])=CC=C3C2=C1 AJEAHBZZHSLIQP-UHFFFAOYSA-N 0.000 description 2
- TZOYXRMEFDYWDQ-UHFFFAOYSA-N 3,4-dihydro-1h-quinolin-2-one Chemical compound C1=CC=C2NC(=O)CCC2=C1 TZOYXRMEFDYWDQ-UHFFFAOYSA-N 0.000 description 2
- GLVSVKSIYXDZHY-UHFFFAOYSA-N 3-nitrofluoren-9-one Chemical compound C1=CC=C2C3=CC([N+](=O)[O-])=CC=C3C(=O)C2=C1 GLVSVKSIYXDZHY-UHFFFAOYSA-N 0.000 description 2
- GNKZMNRKLCTJAY-UHFFFAOYSA-N 4'-Methylacetophenone Chemical compound CC(=O)C1=CC=C(C)C=C1 GNKZMNRKLCTJAY-UHFFFAOYSA-N 0.000 description 2
- MSGQVFMAKBIPNF-UHFFFAOYSA-N 4,7-dimethoxy-2,3-dihydroinden-1-one Chemical compound COC1=CC=C(OC)C2=C1CCC2=O MSGQVFMAKBIPNF-UHFFFAOYSA-N 0.000 description 2
- GCZQTQUHBZZQAC-UHFFFAOYSA-N 4,7-dimethyl-2,3-dihydroinden-1-one Chemical compound CC1=CC=C(C)C2=C1CCC2=O GCZQTQUHBZZQAC-UHFFFAOYSA-N 0.000 description 2
- PCBSXBYCASFXTM-UHFFFAOYSA-N 4-(4-Methoxyphenyl)-2-butanone Chemical compound COC1=CC=C(CCC(C)=O)C=C1 PCBSXBYCASFXTM-UHFFFAOYSA-N 0.000 description 2
- UVVYFYLSZIMKMC-UHFFFAOYSA-N 4-bromo-2,3-dihydroinden-1-one Chemical compound BrC1=CC=CC2=C1CCC2=O UVVYFYLSZIMKMC-UHFFFAOYSA-N 0.000 description 2
- CKSCMRNFDBWFND-UHFFFAOYSA-N 4-hydroxy-2,3-dihydroinden-1-one Chemical compound OC1=CC=CC2=C1CCC2=O CKSCMRNFDBWFND-UHFFFAOYSA-N 0.000 description 2
- OZLKIIAMQWXVKQ-UHFFFAOYSA-N 4-hydroxyfluoren-9-one Chemical compound O=C1C2=CC=CC=C2C2=C1C=CC=C2O OZLKIIAMQWXVKQ-UHFFFAOYSA-N 0.000 description 2
- BTYSYELHQDGJAB-UHFFFAOYSA-N 4-methoxy-2,3-dihydroinden-1-one Chemical compound COC1=CC=CC2=C1CCC2=O BTYSYELHQDGJAB-UHFFFAOYSA-N 0.000 description 2
- NTPLXRHDUXRPNE-UHFFFAOYSA-N 4-methoxyacetophenone Chemical compound COC1=CC=C(C(C)=O)C=C1 NTPLXRHDUXRPNE-UHFFFAOYSA-N 0.000 description 2
- CAVWKDDIVRHSEI-UHFFFAOYSA-N 4-nitrofluoren-9-one Chemical compound O=C1C2=CC=CC=C2C2=C1C=CC=C2[N+](=O)[O-] CAVWKDDIVRHSEI-UHFFFAOYSA-N 0.000 description 2
- IHMQOBPGHZFGLC-UHFFFAOYSA-N 5,6-dimethoxy-2,3-dihydroinden-1-one Chemical compound C1=C(OC)C(OC)=CC2=C1C(=O)CC2 IHMQOBPGHZFGLC-UHFFFAOYSA-N 0.000 description 2
- KSONICAHAPRCMV-UHFFFAOYSA-N 5-bromo-2,3-dihydroinden-1-one Chemical compound BrC1=CC=C2C(=O)CCC2=C1 KSONICAHAPRCMV-UHFFFAOYSA-N 0.000 description 2
- MEDSHTHCZIOVPU-UHFFFAOYSA-N 5-chloro-2,3-dihydroinden-1-one Chemical compound ClC1=CC=C2C(=O)CCC2=C1 MEDSHTHCZIOVPU-UHFFFAOYSA-N 0.000 description 2
- WVPPBVAMKNQXJA-UHFFFAOYSA-N 5-fluoro-2,3-dihydroinden-1-one Chemical compound FC1=CC=C2C(=O)CCC2=C1 WVPPBVAMKNQXJA-UHFFFAOYSA-N 0.000 description 2
- ZRKQOVXGDIZYDS-UHFFFAOYSA-N 5-hydroxy-2,3-dihydroinden-1-one Chemical compound OC1=CC=C2C(=O)CCC2=C1 ZRKQOVXGDIZYDS-UHFFFAOYSA-N 0.000 description 2
- IGDFHUWAXWFKMW-UHFFFAOYSA-N 6-(trifluoromethyl)-2,3-dihydroinden-1-one Chemical compound FC(F)(F)C1=CC=C2CCC(=O)C2=C1 IGDFHUWAXWFKMW-UHFFFAOYSA-N 0.000 description 2
- SEQHEDQNODAFIU-UHFFFAOYSA-N 6-bromo-2,3-dihydroinden-1-one Chemical compound BrC1=CC=C2CCC(=O)C2=C1 SEQHEDQNODAFIU-UHFFFAOYSA-N 0.000 description 2
- SMSGJDOJSQHQIW-UHFFFAOYSA-N 6-chloro-2,3-dihydroinden-1-one Chemical compound ClC1=CC=C2CCC(=O)C2=C1 SMSGJDOJSQHQIW-UHFFFAOYSA-N 0.000 description 2
- LVUUCFIQQHEFEJ-UHFFFAOYSA-N 6-fluoro-2,3-dihydroinden-1-one Chemical compound FC1=CC=C2CCC(=O)C2=C1 LVUUCFIQQHEFEJ-UHFFFAOYSA-N 0.000 description 2
- FKSFQJWXHXGGJM-UHFFFAOYSA-N 6-fluoro-4,4-dimethyl-1,3-dihydroquinolin-2-one Chemical compound C1=C(F)C=C2C(C)(C)CC(=O)NC2=C1 FKSFQJWXHXGGJM-UHFFFAOYSA-N 0.000 description 2
- HOSGXJWQVBHGLT-UHFFFAOYSA-N 6-hydroxy-3,4-dihydro-1h-quinolin-2-one Chemical compound N1C(=O)CCC2=CC(O)=CC=C21 HOSGXJWQVBHGLT-UHFFFAOYSA-N 0.000 description 2
- UJGDLLGKMWVCPT-UHFFFAOYSA-N 6-methoxy-2,3-dihydroinden-1-one Chemical compound COC1=CC=C2CCC(=O)C2=C1 UJGDLLGKMWVCPT-UHFFFAOYSA-N 0.000 description 2
- HFMZPBSZKCDKOR-UHFFFAOYSA-N 7-hydroxy-2,3-dihydroinden-1-one Chemical compound OC1=CC=CC2=C1C(=O)CC2 HFMZPBSZKCDKOR-UHFFFAOYSA-N 0.000 description 2
- LKLSFDWYIBUGNT-UHFFFAOYSA-N 7-hydroxy-3,4-dihydro-1h-quinolin-2-one Chemical compound C1CC(=O)NC2=CC(O)=CC=C21 LKLSFDWYIBUGNT-UHFFFAOYSA-N 0.000 description 2
- UHCQKTPURTYSFF-UHFFFAOYSA-N 8-bromo-2,3-dihydro-1h-quinolin-4-one Chemical compound O=C1CCNC2=C1C=CC=C2Br UHCQKTPURTYSFF-UHFFFAOYSA-N 0.000 description 2
- QXQVWZSZFKWXFW-UHFFFAOYSA-N 8-fluoro-4,4-dimethyl-1,3-dihydroquinolin-2-one Chemical compound C1=CC=C2C(C)(C)CC(=O)NC2=C1F QXQVWZSZFKWXFW-UHFFFAOYSA-N 0.000 description 2
- AANYDYCSNSJWAU-UHFFFAOYSA-N C1CCC(N2CCCCC2)CC1.CCCCC(CC)CN(C)C.CCCCCN(C)CCCCC.CCCCN(C)C.CCCCN(C)CC.CCCCN(C)CCCC.CCCN(C)C.CCCN(C)CCC.CCN(C)C.CCN(C)CC.CCN(CC)C1CCCCC1.CCN1CCCCC1.CN(C)C.CN(C)C.CN(C)C.CN(C)C.CN(C)C1CCCCC1.CN(C)CC1=CC=CC=C1.CN(C)CCC1=CC=CC=C1.CN1CCCC1 Chemical compound C1CCC(N2CCCCC2)CC1.CCCCC(CC)CN(C)C.CCCCCN(C)CCCCC.CCCCN(C)C.CCCCN(C)CC.CCCCN(C)CCCC.CCCN(C)C.CCCN(C)CCC.CCN(C)C.CCN(C)CC.CCN(CC)C1CCCCC1.CCN1CCCCC1.CN(C)C.CN(C)C.CN(C)C.CN(C)C.CN(C)C1CCCCC1.CN(C)CC1=CC=CC=C1.CN(C)CCC1=CC=CC=C1.CN1CCCC1 AANYDYCSNSJWAU-UHFFFAOYSA-N 0.000 description 2
- VZVOCRMKAOWBMF-UHFFFAOYSA-N COC1CC(=O)c2ccc(OC)cc12 Chemical compound COC1CC(=O)c2ccc(OC)cc12 VZVOCRMKAOWBMF-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- SXVPOSFURRDKBO-UHFFFAOYSA-N Cyclododecanone Chemical compound O=C1CCCCCCCCCCC1 SXVPOSFURRDKBO-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 239000002879 Lewis base Substances 0.000 description 2
- HTLZVHNRZJPSMI-UHFFFAOYSA-N N-ethylpiperidine Chemical compound CCN1CCCCC1 HTLZVHNRZJPSMI-UHFFFAOYSA-N 0.000 description 2
- 150000008522 N-ethylpiperidines Chemical class 0.000 description 2
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 2
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 2
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 2
- 235000011130 ammonium sulphate Nutrition 0.000 description 2
- 229960004543 anhydrous citric acid Drugs 0.000 description 2
- XMINMPJQCBQFGN-UHFFFAOYSA-N azocan-3-one Chemical compound O=C1CCCCCNC1 XMINMPJQCBQFGN-UHFFFAOYSA-N 0.000 description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 238000009530 blood pressure measurement Methods 0.000 description 2
- 239000012159 carrier gas Substances 0.000 description 2
- 238000002425 crystallisation Methods 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- BAUZLFKYYIVGPM-UHFFFAOYSA-N cyclononanone Chemical compound O=C1CCCCCCCC1 BAUZLFKYYIVGPM-UHFFFAOYSA-N 0.000 description 2
- OSOIQJGOYGSIMF-UHFFFAOYSA-N cyclopentadecanone Chemical compound O=C1CCCCCCCCCCCCCC1 OSOIQJGOYGSIMF-UHFFFAOYSA-N 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- UJLFLOAXKVGYIA-UHFFFAOYSA-N indan-2-one oxime Chemical compound C1=CC=C2CC(=NO)CC2=C1 UJLFLOAXKVGYIA-UHFFFAOYSA-N 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 150000007527 lewis bases Chemical class 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- MQWCXKGKQLNYQG-UHFFFAOYSA-N methyl cyclohexan-4-ol Natural products CC1CCC(O)CC1 MQWCXKGKQLNYQG-UHFFFAOYSA-N 0.000 description 2
- CRNNFEKVPRFZKJ-UHFFFAOYSA-N n-fluoren-9-ylidenehydroxylamine Chemical compound C1=CC=C2C(=NO)C3=CC=CC=C3C2=C1 CRNNFEKVPRFZKJ-UHFFFAOYSA-N 0.000 description 2
- DQGSJTVMODPFBK-UHFFFAOYSA-N oxacyclotridecan-2-one Chemical compound O=C1CCCCCCCCCCCO1 DQGSJTVMODPFBK-UHFFFAOYSA-N 0.000 description 2
- XNLICIUVMPYHGG-UHFFFAOYSA-N pentan-2-one Chemical compound CCCC(C)=O XNLICIUVMPYHGG-UHFFFAOYSA-N 0.000 description 2
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 230000001376 precipitating effect Effects 0.000 description 2
- KRIOVPPHQSLHCZ-UHFFFAOYSA-N propiophenone Chemical compound CCC(=O)C1=CC=CC=C1 KRIOVPPHQSLHCZ-UHFFFAOYSA-N 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- KHXSJSBQIWAIEG-UHFFFAOYSA-N (4-chlorophenyl)-pyridin-2-ylmethanone Chemical compound C1=CC(Cl)=CC=C1C(=O)C1=CC=CC=N1 KHXSJSBQIWAIEG-UHFFFAOYSA-N 0.000 description 1
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- SYMAGJYJMLUEQE-ONEGZZNKSA-N (e)-3-ethoxyprop-2-enoic acid Chemical compound CCO\C=C\C(O)=O SYMAGJYJMLUEQE-ONEGZZNKSA-N 0.000 description 1
- DDNPADUKGZMCHV-CMDGGOBGSA-N (e)-3-phenyl-1-thiophen-2-ylprop-2-en-1-one Chemical compound C=1C=CSC=1C(=O)\C=C\C1=CC=CC=C1 DDNPADUKGZMCHV-CMDGGOBGSA-N 0.000 description 1
- YFKBXYGUSOXJGS-UHFFFAOYSA-N 1,3-Diphenyl-2-propanone Chemical compound C=1C=CC=CC=1CC(=O)CC1=CC=CC=C1 YFKBXYGUSOXJGS-UHFFFAOYSA-N 0.000 description 1
- JBGJVMVWYWUVOW-UHFFFAOYSA-N 1-(1-hydroxynaphthalen-2-yl)ethanone Chemical compound C1=CC=CC2=C(O)C(C(=O)C)=CC=C21 JBGJVMVWYWUVOW-UHFFFAOYSA-N 0.000 description 1
- LDUYRQGNBYKUAG-UHFFFAOYSA-N 1-(2,4-dimethoxyphenyl)propan-2-one Chemical compound COC1=CC=C(CC(C)=O)C(OC)=C1 LDUYRQGNBYKUAG-UHFFFAOYSA-N 0.000 description 1
- XHEUPKZIPXSSLU-UHFFFAOYSA-N 1-(2,5-dimethylphenyl)propan-2-one Chemical compound CC(=O)CC1=CC(C)=CC=C1C XHEUPKZIPXSSLU-UHFFFAOYSA-N 0.000 description 1
- YAEXJQJVTXDTJM-UHFFFAOYSA-N 1-(2,6-dichlorophenyl)propan-2-one Chemical compound CC(=O)CC1=C(Cl)C=CC=C1Cl YAEXJQJVTXDTJM-UHFFFAOYSA-N 0.000 description 1
- LAWHOFKPDMZDLJ-UHFFFAOYSA-N 1-(2,6-difluorophenyl)propan-2-one Chemical compound CC(=O)CC1=C(F)C=CC=C1F LAWHOFKPDMZDLJ-UHFFFAOYSA-N 0.000 description 1
- VUIOUIWZVKVFCI-UHFFFAOYSA-N 1-(2-hydroxynaphthalen-1-yl)ethanone Chemical compound C1=CC=C2C(C(=O)C)=C(O)C=CC2=C1 VUIOUIWZVKVFCI-UHFFFAOYSA-N 0.000 description 1
- WFBBDKXOGOJOKY-UHFFFAOYSA-N 1-(2-methoxynaphthalen-1-yl)ethanone Chemical compound C1=CC=CC2=C(C(C)=O)C(OC)=CC=C21 WFBBDKXOGOJOKY-UHFFFAOYSA-N 0.000 description 1
- CWILMKDSVMROHT-UHFFFAOYSA-N 1-(2-phenanthrenyl)ethanone Chemical compound C1=CC=C2C3=CC=C(C(=O)C)C=C3C=CC2=C1 CWILMKDSVMROHT-UHFFFAOYSA-N 0.000 description 1
- YJKHOUIVWKQRSL-UHFFFAOYSA-N 1-(3,5-dimethoxyphenyl)ethanone Chemical compound COC1=CC(OC)=CC(C(C)=O)=C1 YJKHOUIVWKQRSL-UHFFFAOYSA-N 0.000 description 1
- VCNYPJMEQHTAHS-UHFFFAOYSA-N 1-(3-chlorophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=CC(Cl)=C1 VCNYPJMEQHTAHS-UHFFFAOYSA-N 0.000 description 1
- RMMRRRLPDBJBQL-UHFFFAOYSA-N 1-(3-methoxyphenyl)propan-2-one Chemical compound COC1=CC=CC(CC(C)=O)=C1 RMMRRRLPDBJBQL-UHFFFAOYSA-N 0.000 description 1
- OWZZXUNOENOULT-UHFFFAOYSA-N 1-(3-methylphenyl)propan-2-one Chemical compound CC(=O)CC1=CC=CC(C)=C1 OWZZXUNOENOULT-UHFFFAOYSA-N 0.000 description 1
- WEJRYKSUUFKMBC-UHFFFAOYSA-N 1-(4-chlorophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C(Cl)C=C1 WEJRYKSUUFKMBC-UHFFFAOYSA-N 0.000 description 1
- DNAGWILBAXAZOH-UHFFFAOYSA-N 1-(4-ethylphenyl)propan-2-one Chemical compound CCC1=CC=C(CC(C)=O)C=C1 DNAGWILBAXAZOH-UHFFFAOYSA-N 0.000 description 1
- BKGIZYOJHJKFJP-UHFFFAOYSA-N 1-(4-methylnaphthalen-1-yl)ethanone Chemical compound C1=CC=C2C(C(=O)C)=CC=C(C)C2=C1 BKGIZYOJHJKFJP-UHFFFAOYSA-N 0.000 description 1
- NOXKUHSBIXPZBJ-UHFFFAOYSA-N 1-(4-methylphenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C(C)C=C1 NOXKUHSBIXPZBJ-UHFFFAOYSA-N 0.000 description 1
- GEWWCWZGHNIUBW-UHFFFAOYSA-N 1-(4-nitrophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C([N+]([O-])=O)C=C1 GEWWCWZGHNIUBW-UHFFFAOYSA-N 0.000 description 1
- AZXFCQJWAAKUBY-UHFFFAOYSA-N 1-(6-bromophenanthren-9-yl)ethanone Chemical compound BrC1=CC=C2C(C(=O)C)=CC3=CC=CC=C3C2=C1 AZXFCQJWAAKUBY-UHFFFAOYSA-N 0.000 description 1
- LIQJHDWTWXVXFT-UHFFFAOYSA-N 1-bromo-1-phenylbutan-2-one Chemical compound CCC(=O)C(Br)C1=CC=CC=C1 LIQJHDWTWXVXFT-UHFFFAOYSA-N 0.000 description 1
- QFIJADPMCNPDIS-UHFFFAOYSA-N 1-hydroxy-3-phenylbutan-2-one Chemical compound OCC(=O)C(C)C1=CC=CC=C1 QFIJADPMCNPDIS-UHFFFAOYSA-N 0.000 description 1
- JKVNPRNAHRHQDD-UHFFFAOYSA-N 1-phenanthren-3-ylethanone Chemical compound C1=CC=C2C3=CC(C(=O)C)=CC=C3C=CC2=C1 JKVNPRNAHRHQDD-UHFFFAOYSA-N 0.000 description 1
- NQUAUYFCIGKDFS-UHFFFAOYSA-N 1-phenanthren-4-ylethanone Chemical compound C1=CC=CC2=C3C(C(=O)C)=CC=CC3=CC=C21 NQUAUYFCIGKDFS-UHFFFAOYSA-N 0.000 description 1
- UIFAWZBYTTXSOG-UHFFFAOYSA-N 1-phenanthren-9-ylethanone Chemical compound C1=CC=C2C(C(=O)C)=CC3=CC=CC=C3C2=C1 UIFAWZBYTTXSOG-UHFFFAOYSA-N 0.000 description 1
- GKDLTXYXODKDEA-UHFFFAOYSA-N 1-phenylbutan-2-one Chemical compound CCC(=O)CC1=CC=CC=C1 GKDLTXYXODKDEA-UHFFFAOYSA-N 0.000 description 1
- OMYOJDLCFAUIHN-UHFFFAOYSA-N 1-pyrrol-1-ylethanone Chemical compound CC(=O)N1C=CC=C1 OMYOJDLCFAUIHN-UHFFFAOYSA-N 0.000 description 1
- MVOSYKNQRRHGKX-UHFFFAOYSA-N 11-Undecanolactone Chemical compound O=C1CCCCCCCCCCO1 MVOSYKNQRRHGKX-UHFFFAOYSA-N 0.000 description 1
- IUDIJIVSWGWJNV-UHFFFAOYSA-N 13-tridecanolide Chemical compound O=C1CCCCCCCCCCCCO1 IUDIJIVSWGWJNV-UHFFFAOYSA-N 0.000 description 1
- OJNBOHZOCFAFLT-UHFFFAOYSA-N 2,2,4-trichloronaphthalen-1-one Chemical compound ClC1(C(C2=CC=CC=C2C(=C1)Cl)=O)Cl OJNBOHZOCFAFLT-UHFFFAOYSA-N 0.000 description 1
- 125000003562 2,2-dimethylpentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000003660 2,3-dimethylpentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- JABKESJVYSQBGF-UHFFFAOYSA-N 2-(4-methoxyphenyl)-1-phenylethanone Chemical compound C1=CC(OC)=CC=C1CC(=O)C1=CC=CC=C1 JABKESJVYSQBGF-UHFFFAOYSA-N 0.000 description 1
- XSAYZAUNJMRRIR-UHFFFAOYSA-N 2-acetylnaphthalene Chemical compound C1=CC=CC2=CC(C(=O)C)=CC=C21 XSAYZAUNJMRRIR-UHFFFAOYSA-N 0.000 description 1
- PVHNKILPLXXDDZ-UHFFFAOYSA-N 2-phenylcycloheptan-1-one Chemical compound O=C1CCCCCC1C1=CC=CC=C1 PVHNKILPLXXDDZ-UHFFFAOYSA-N 0.000 description 1
- DRLVMOAWNVOSPE-UHFFFAOYSA-N 2-phenylcyclohexan-1-one Chemical compound O=C1CCCCC1C1=CC=CC=C1 DRLVMOAWNVOSPE-UHFFFAOYSA-N 0.000 description 1
- NPELEPAOYMNNRW-UHFFFAOYSA-N 2-phenylcyclopentan-1-one Chemical compound O=C1CCCC1C1=CC=CC=C1 NPELEPAOYMNNRW-UHFFFAOYSA-N 0.000 description 1
- RZPFVRFSYMUDJO-UHFFFAOYSA-N 2h-naphthalen-1-one Chemical compound C1=CC=C2C(=O)CC=CC2=C1 RZPFVRFSYMUDJO-UHFFFAOYSA-N 0.000 description 1
- WDNDMVGQGRDIBC-UHFFFAOYSA-N 3,4,5-Trimethoxyphenyl-2-propanone Chemical compound COC1=CC(CC(C)=O)=CC(OC)=C1OC WDNDMVGQGRDIBC-UHFFFAOYSA-N 0.000 description 1
- JQXBETDGCMQLMK-UHFFFAOYSA-N 3,4-Dihydroxyphenylacetone Chemical compound CC(=O)CC1=CC=C(O)C(O)=C1 JQXBETDGCMQLMK-UHFFFAOYSA-N 0.000 description 1
- QBCUUJGHWFKMDC-UHFFFAOYSA-N 3-Hydroxy-4-phenylbutan-2-one Chemical compound CC(=O)C(O)CC1=CC=CC=C1 QBCUUJGHWFKMDC-UHFFFAOYSA-N 0.000 description 1
- VVSQAKKEJGMRSP-UHFFFAOYSA-N 3-bromo-4-phenylbutan-2-one Chemical compound CC(=O)C(Br)CC1=CC=CC=C1 VVSQAKKEJGMRSP-UHFFFAOYSA-N 0.000 description 1
- PNDPCXFOHXLBRE-UHFFFAOYSA-N 3-chloro-4-phenylbutan-2-one Chemical compound CC(=O)C(Cl)CC1=CC=CC=C1 PNDPCXFOHXLBRE-UHFFFAOYSA-N 0.000 description 1
- NYTLQBYJPPTEQG-UHFFFAOYSA-N 3-hydroxy-1,3-diphenylbutan-2-one Chemical compound CC(O)(C(=O)Cc1ccccc1)c1ccccc1 NYTLQBYJPPTEQG-UHFFFAOYSA-N 0.000 description 1
- RFHRXUDEWBOJCW-UHFFFAOYSA-N 3-hydroxy-1-phenylbutan-2-one Chemical compound CC(O)C(=O)CC1=CC=CC=C1 RFHRXUDEWBOJCW-UHFFFAOYSA-N 0.000 description 1
- IBWFPYRVHYLXEU-UHFFFAOYSA-N 3-hydroxy-3-phenylbutan-2-one Chemical compound CC(=O)C(C)(O)C1=CC=CC=C1 IBWFPYRVHYLXEU-UHFFFAOYSA-N 0.000 description 1
- ZATIMOAGKJEVGN-UHFFFAOYSA-N 3-methyl-1-phenylbutan-2-one Chemical compound CC(C)C(=O)CC1=CC=CC=C1 ZATIMOAGKJEVGN-UHFFFAOYSA-N 0.000 description 1
- 125000003469 3-methylhexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- ROADCYAOHVSOLQ-UHFFFAOYSA-N 3-oxetanone Chemical compound O=C1COC1 ROADCYAOHVSOLQ-UHFFFAOYSA-N 0.000 description 1
- CVWMNAWLNRRPOL-UHFFFAOYSA-N 3-phenylbutan-2-one Chemical compound CC(=O)C(C)C1=CC=CC=C1 CVWMNAWLNRRPOL-UHFFFAOYSA-N 0.000 description 1
- JLDCLDZGNYDRKX-UHFFFAOYSA-N 3-phenylcycloheptan-1-one Chemical compound C1C(=O)CCCCC1C1=CC=CC=C1 JLDCLDZGNYDRKX-UHFFFAOYSA-N 0.000 description 1
- CJAUDSQXFVZPTO-UHFFFAOYSA-N 3-phenylcyclohexan-1-one Chemical compound C1C(=O)CCCC1C1=CC=CC=C1 CJAUDSQXFVZPTO-UHFFFAOYSA-N 0.000 description 1
- MFBXYJLOYZMFIN-UHFFFAOYSA-N 3-phenylcyclopentan-1-one Chemical compound C1C(=O)CCC1C1=CC=CC=C1 MFBXYJLOYZMFIN-UHFFFAOYSA-N 0.000 description 1
- FPHXYKLKNOEKTQ-UHFFFAOYSA-N 4,4-diphenylbutan-2-one Chemical compound C=1C=CC=CC=1C(CC(=O)C)C1=CC=CC=C1 FPHXYKLKNOEKTQ-UHFFFAOYSA-N 0.000 description 1
- TZJLGGWGVLADDN-UHFFFAOYSA-N 4-(3,4-Methylenedioxyphenyl)-2-butanone Chemical compound CC(=O)CCC1=CC=C2OCOC2=C1 TZJLGGWGVLADDN-UHFFFAOYSA-N 0.000 description 1
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 1
- LXZMKGWDPDBHSO-UHFFFAOYSA-N 4-hydroxy-3-phenylbutan-2-one Chemical compound CC(=O)C(CO)C1=CC=CC=C1 LXZMKGWDPDBHSO-UHFFFAOYSA-N 0.000 description 1
- MUVVFSVVVHYLMQ-UHFFFAOYSA-N 4-hydroxy-4-phenylbutan-2-one Chemical compound CC(=O)CC(O)C1=CC=CC=C1 MUVVFSVVVHYLMQ-UHFFFAOYSA-N 0.000 description 1
- VWMVAQHMFFZQGD-UHFFFAOYSA-N 4-hydroxyphenylacetone Chemical compound CC(=O)CC1=CC=C(O)C=C1 VWMVAQHMFFZQGD-UHFFFAOYSA-N 0.000 description 1
- FLCQYXWYKZDWKF-UHFFFAOYSA-N 4-methyl-1h-naphthalen-2-one Chemical compound C1=CC=C2C(C)=CC(=O)CC2=C1 FLCQYXWYKZDWKF-UHFFFAOYSA-N 0.000 description 1
- QCVMLXNTSUFLGI-UHFFFAOYSA-N 4-methylfluoren-9-one Chemical compound O=C1C2=CC=CC=C2C2=C1C=CC=C2C QCVMLXNTSUFLGI-UHFFFAOYSA-N 0.000 description 1
- VRJHQPZVIGNGMX-UHFFFAOYSA-N 4-piperidinone Chemical compound O=C1CCNCC1 VRJHQPZVIGNGMX-UHFFFAOYSA-N 0.000 description 1
- OZJPLYNZGCXSJM-UHFFFAOYSA-N 5-valerolactone Chemical compound O=C1CCCCO1 OZJPLYNZGCXSJM-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- OABDDDLGKQGKGV-UHFFFAOYSA-N C(C)C(=O)C.CN(C)CC1=CC=CC=C1 Chemical compound C(C)C(=O)C.CN(C)CC1=CC=CC=C1 OABDDDLGKQGKGV-UHFFFAOYSA-N 0.000 description 1
- BGGKQMMGRCLCDS-UHFFFAOYSA-N C(C)C(=O)C.N1=C(C=CC=C1)C Chemical compound C(C)C(=O)C.N1=C(C=CC=C1)C BGGKQMMGRCLCDS-UHFFFAOYSA-N 0.000 description 1
- VZCLQIQLNHCCBN-UHFFFAOYSA-N C1=CC=C2C(C(=O)C)=C(F)C3=CC=CC=C3C2=C1 Chemical compound C1=CC=C2C(C(=O)C)=C(F)C3=CC=CC=C3C2=C1 VZCLQIQLNHCCBN-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KMSNYNIWEORQDJ-UHFFFAOYSA-N Dihydro-2(3H)-thiophenone Chemical compound O=C1CCCS1 KMSNYNIWEORQDJ-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- MUCRYNWJQNHDJH-OADIDDRXSA-N Ursonic acid Chemical compound C1CC(=O)C(C)(C)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CC[C@@H](C)[C@H](C)[C@H]5C4=CC[C@@H]3[C@]21C MUCRYNWJQNHDJH-OADIDDRXSA-N 0.000 description 1
- HZSIFDFXFAXICF-UHFFFAOYSA-N acetolactone Chemical compound O=C1CO1 HZSIFDFXFAXICF-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 125000000777 acyl halide group Chemical group 0.000 description 1
- 150000001299 aldehydes Chemical group 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000001408 amides Chemical group 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- WUTGHUJGKDDNMJ-UHFFFAOYSA-N azacyclopentadecan-7-one Chemical compound C1CCCCNCCCCCCCCC1=O WUTGHUJGKDDNMJ-UHFFFAOYSA-N 0.000 description 1
- NMYDLIYKLDCLSU-UHFFFAOYSA-N azacyclotetradecan-7-one Chemical compound O=C1CCCCCCCNCCCCC1 NMYDLIYKLDCLSU-UHFFFAOYSA-N 0.000 description 1
- KQJMYBBTWBTGFZ-UHFFFAOYSA-N azacyclotetradecan-8-one Chemical compound O=C1CCCCCCNCCCCCC1 KQJMYBBTWBTGFZ-UHFFFAOYSA-N 0.000 description 1
- YNPHWXKBVDFVNH-UHFFFAOYSA-N azacyclotridecan-5-one Chemical compound O=C1CCCCCCCCNCCC1 YNPHWXKBVDFVNH-UHFFFAOYSA-N 0.000 description 1
- HVYNIIDMVFDCGB-UHFFFAOYSA-N azacyclotridecan-6-one Chemical compound O=C1CCCCCCCNCCCC1 HVYNIIDMVFDCGB-UHFFFAOYSA-N 0.000 description 1
- BFPYDKUUSZTVAV-UHFFFAOYSA-N azacyclotridecan-7-one Chemical compound O=C1CCCCCCNCCCCC1 BFPYDKUUSZTVAV-UHFFFAOYSA-N 0.000 description 1
- ZTSPWWDUWKMFLO-UHFFFAOYSA-N azacycloundecan-6-one Chemical compound O=C1CCCCCNCCCC1 ZTSPWWDUWKMFLO-UHFFFAOYSA-N 0.000 description 1
- XYGKKJHGZOGVBL-UHFFFAOYSA-N azepan-3-one Chemical compound O=C1CCCCNC1 XYGKKJHGZOGVBL-UHFFFAOYSA-N 0.000 description 1
- GMHPWGYTSXHHPI-UHFFFAOYSA-N azepan-4-one Chemical compound O=C1CCCNCC1 GMHPWGYTSXHHPI-UHFFFAOYSA-N 0.000 description 1
- 125000002785 azepinyl group Chemical group 0.000 description 1
- XPRVSYXHPUYSGF-UHFFFAOYSA-N azetidin-3-one Chemical compound O=C1CNC1 XPRVSYXHPUYSGF-UHFFFAOYSA-N 0.000 description 1
- ADHMWGUEFSNYCZ-UHFFFAOYSA-N azocan-4-one Chemical compound O=C1CCCCNCC1 ADHMWGUEFSNYCZ-UHFFFAOYSA-N 0.000 description 1
- JCEJTIOVIJRGIK-UHFFFAOYSA-N azocan-5-one Chemical compound O=C1CCCNCCC1 JCEJTIOVIJRGIK-UHFFFAOYSA-N 0.000 description 1
- VNHCCFQBSJDDSI-UHFFFAOYSA-N azonan-3-one Chemical compound O=C1CCCCCCNC1 VNHCCFQBSJDDSI-UHFFFAOYSA-N 0.000 description 1
- DWYSUKURKKCNKD-UHFFFAOYSA-N azonan-4-one Chemical compound O=C1CCCCCNCC1 DWYSUKURKKCNKD-UHFFFAOYSA-N 0.000 description 1
- ZDIHHVJNMXHNGF-UHFFFAOYSA-N azonan-5-one Chemical compound O=C1CCCCNCCC1 ZDIHHVJNMXHNGF-UHFFFAOYSA-N 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- WVXMLBXFHITUIT-UHFFFAOYSA-N benzo[a]fluoren-1-one Chemical compound C1=CC=CC2=CC3=C4C(=O)C=CC=C4C=CC3=C21 WVXMLBXFHITUIT-UHFFFAOYSA-N 0.000 description 1
- WRQZBCCMYPGFTM-UHFFFAOYSA-N benzo[a]fluoren-11-one 4-methylfluoren-9-one Chemical compound C1=CC=CC=2C1=C1C(C3=CC=CC=C3C1=CC2)=O.CC2=CC=CC=1C(C3=CC=CC=C3C21)=O WRQZBCCMYPGFTM-UHFFFAOYSA-N 0.000 description 1
- AGCQBGSDLGORJV-UHFFFAOYSA-N benzo[h]fluoren-1-one Chemical compound C1=CC=C2C=C3C4=CC=CC(=O)C4=CC3=CC2=C1 AGCQBGSDLGORJV-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- VEZXCJBBBCKRPI-UHFFFAOYSA-N beta-propiolactone Chemical compound O=C1CCO1 VEZXCJBBBCKRPI-UHFFFAOYSA-N 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- 238000009937 brining Methods 0.000 description 1
- HOWPNAXQBWCGOT-UHFFFAOYSA-N butan-2-one N,N-dipropylpropan-1-amine Chemical compound C(C)C(=O)C.C(CC)N(CCC)CCC HOWPNAXQBWCGOT-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical group 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000011208 chromatographic data Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- SHQSVMDWKBRBGB-UHFFFAOYSA-N cyclobutanone Chemical compound O=C1CCC1 SHQSVMDWKBRBGB-UHFFFAOYSA-N 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- MVEBDOSCXOQNAR-UHFFFAOYSA-N cyclobutyl(phenyl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1CCC1 MVEBDOSCXOQNAR-UHFFFAOYSA-N 0.000 description 1
- SXOZDDAFVJANJP-UHFFFAOYSA-N cyclodecanone Chemical compound O=C1CCCCCCCCC1 SXOZDDAFVJANJP-UHFFFAOYSA-N 0.000 description 1
- CGZZMOTZOONQIA-UHFFFAOYSA-N cycloheptanone Chemical compound O=C1CCCCCC1 CGZZMOTZOONQIA-UHFFFAOYSA-N 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- BMFYCFSWWDXEPB-UHFFFAOYSA-N cyclohexyl(phenyl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1CCCCC1 BMFYCFSWWDXEPB-UHFFFAOYSA-N 0.000 description 1
- IIRFCWANHMSDCG-UHFFFAOYSA-N cyclooctanone Chemical compound O=C1CCCCCCC1 IIRFCWANHMSDCG-UHFFFAOYSA-N 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- VYDIMQRLNMMJBW-UHFFFAOYSA-N cyclopentyl(phenyl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1CCCC1 VYDIMQRLNMMJBW-UHFFFAOYSA-N 0.000 description 1
- VBBRYJMZLIYUJQ-UHFFFAOYSA-N cyclopropanone Chemical compound O=C1CC1 VBBRYJMZLIYUJQ-UHFFFAOYSA-N 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- PJDFNFSTSCAKPC-UHFFFAOYSA-N cyclopropyl(2-thienyl)methanone Chemical compound C=1C=CSC=1C(=O)C1CC1 PJDFNFSTSCAKPC-UHFFFAOYSA-N 0.000 description 1
- PJRHFTYXYCVOSJ-UHFFFAOYSA-N cyclopropyl(phenyl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1CC1 PJRHFTYXYCVOSJ-UHFFFAOYSA-N 0.000 description 1
- FUGDHQXYVPQGLJ-UHFFFAOYSA-N cyclotetradecanone Chemical compound O=C1CCCCCCCCCCCCC1 FUGDHQXYVPQGLJ-UHFFFAOYSA-N 0.000 description 1
- VHUGWUBIUBBUAF-UHFFFAOYSA-N cyclotridecanone Chemical compound O=C1CCCCCCCCCCCC1 VHUGWUBIUBBUAF-UHFFFAOYSA-N 0.000 description 1
- UPOSSYJVWXLPTA-UHFFFAOYSA-N cycloundecanone Chemical compound O=C1CCCCCCCCCC1 UPOSSYJVWXLPTA-UHFFFAOYSA-N 0.000 description 1
- 238000010612 desalination reaction Methods 0.000 description 1
- RMMZGPSYPUIIRO-UHFFFAOYSA-N di(thiophen-3-yl)methanone Chemical compound C1=CSC=C1C(=O)C=1C=CSC=1 RMMZGPSYPUIIRO-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- OUQSAXROROGQEE-UHFFFAOYSA-N diphenyl-[2-(4-propan-2-yl-4,5-dihydro-1,3-oxazol-2-yl)phenyl]phosphane Chemical compound CC(C)C1COC(C=2C(=CC=CC=2)P(C=2C=CC=CC=2)C=2C=CC=CC=2)=N1 OUQSAXROROGQEE-UHFFFAOYSA-N 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000000909 electrodialysis Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 150000002085 enols Chemical class 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000012527 feed solution Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 238000009292 forward osmosis Methods 0.000 description 1
- 239000013505 freshwater Substances 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- JEGUKCSWCFPDGT-UHFFFAOYSA-N h2o hydrate Chemical compound O.O JEGUKCSWCFPDGT-UHFFFAOYSA-N 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 125000001183 hydrocarbyl group Chemical group 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000010808 liquid waste Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- SUCIUGVCEOVANL-UHFFFAOYSA-N oxacyclododecan-3-one Chemical compound O1CC(CCCCCCCCC1)=O SUCIUGVCEOVANL-UHFFFAOYSA-N 0.000 description 1
- KGGRNJOEDBFTSP-UHFFFAOYSA-N oxacyclododecan-4-one Chemical compound O1CCC(CCCCCCCC1)=O KGGRNJOEDBFTSP-UHFFFAOYSA-N 0.000 description 1
- CFYWUJKNUBMHGY-UHFFFAOYSA-N oxacyclododecan-5-one Chemical compound O1CCCC(CCCCCCC1)=O CFYWUJKNUBMHGY-UHFFFAOYSA-N 0.000 description 1
- FIZTZNPBLKWRDV-UHFFFAOYSA-N oxacyclododecan-6-one Chemical compound O1CCCCC(CCCCCC1)=O FIZTZNPBLKWRDV-UHFFFAOYSA-N 0.000 description 1
- VUBVZGULTULSRB-UHFFFAOYSA-N oxacyclododecan-7-one Chemical compound O1CCCCCC(CCCCC1)=O VUBVZGULTULSRB-UHFFFAOYSA-N 0.000 description 1
- LFSYLMRHJKGLDV-UHFFFAOYSA-N oxacyclopentadecan-2-one Chemical compound O=C1CCCCCCCCCCCCCO1 LFSYLMRHJKGLDV-UHFFFAOYSA-N 0.000 description 1
- ZDFFZJJWVFANBK-UHFFFAOYSA-N oxacyclopentadecan-3-one Chemical compound O=C1CCCCCCCCCCCCOC1 ZDFFZJJWVFANBK-UHFFFAOYSA-N 0.000 description 1
- REWPBZGACQVODN-UHFFFAOYSA-N oxacyclopentadecan-4-one Chemical compound O1CCC(CCCCCCCCCCC1)=O REWPBZGACQVODN-UHFFFAOYSA-N 0.000 description 1
- XWTZYLRYKXTJHO-UHFFFAOYSA-N oxacyclopentadecan-5-one Chemical compound O1CCCC(CCCCCCCCCC1)=O XWTZYLRYKXTJHO-UHFFFAOYSA-N 0.000 description 1
- RMKPGVRTEHCOFS-UHFFFAOYSA-N oxacyclopentadecan-6-one Chemical compound O1CCCCC(CCCCCCCCC1)=O RMKPGVRTEHCOFS-UHFFFAOYSA-N 0.000 description 1
- SVAURAVGAXMPKE-UHFFFAOYSA-N oxacyclopentadecan-7-one Chemical compound O1CCCCCC(CCCCCCCC1)=O SVAURAVGAXMPKE-UHFFFAOYSA-N 0.000 description 1
- MNEGDAZDRPEGIK-UHFFFAOYSA-N oxacyclopentadecan-8-one Chemical compound O1CCCCCCC(CCCCCCC1)=O MNEGDAZDRPEGIK-UHFFFAOYSA-N 0.000 description 1
- OROOIZMWNSKQQH-UHFFFAOYSA-N oxacyclotetradecan-3-one Chemical compound O=C1CCCCCCCCCCCOC1 OROOIZMWNSKQQH-UHFFFAOYSA-N 0.000 description 1
- VVTJOAFDCQVEIH-UHFFFAOYSA-N oxacyclotetradecan-4-one Chemical compound O1CCC(CCCCCCCCCC1)=O VVTJOAFDCQVEIH-UHFFFAOYSA-N 0.000 description 1
- PCZOIAVWJCLDPV-UHFFFAOYSA-N oxacyclotetradecan-5-one Chemical compound O1CCCC(CCCCCCCCC1)=O PCZOIAVWJCLDPV-UHFFFAOYSA-N 0.000 description 1
- MAUHJCCHDLCIAA-UHFFFAOYSA-N oxacyclotetradecan-6-one Chemical compound O1CCCCC(CCCCCCCC1)=O MAUHJCCHDLCIAA-UHFFFAOYSA-N 0.000 description 1
- RPEBCIDHWOZLOD-UHFFFAOYSA-N oxacyclotetradecan-7-one Chemical compound O=C1CCCCCCCOCCCCC1 RPEBCIDHWOZLOD-UHFFFAOYSA-N 0.000 description 1
- RFHJQALGFDMNEV-UHFFFAOYSA-N oxacyclotetradecan-8-one Chemical compound O1CCCCCCC(CCCCCC1)=O RFHJQALGFDMNEV-UHFFFAOYSA-N 0.000 description 1
- SUEDIHOALKJEPW-UHFFFAOYSA-N oxacyclotridecan-3-one Chemical compound O1CC(CCCCCCCCCC1)=O SUEDIHOALKJEPW-UHFFFAOYSA-N 0.000 description 1
- XVDUDYHRJRKWIZ-UHFFFAOYSA-N oxacyclotridecan-4-one Chemical compound O1CCC(CCCCCCCCC1)=O XVDUDYHRJRKWIZ-UHFFFAOYSA-N 0.000 description 1
- VUIJTEPYXZRTMI-UHFFFAOYSA-N oxacyclotridecan-5-one Chemical compound O1CCCC(CCCCCCCC1)=O VUIJTEPYXZRTMI-UHFFFAOYSA-N 0.000 description 1
- LYUTURWBUXHTSF-UHFFFAOYSA-N oxacyclotridecan-6-one Chemical compound O1CCCCC(CCCCCCC1)=O LYUTURWBUXHTSF-UHFFFAOYSA-N 0.000 description 1
- RYTAUJFRHPXPRS-UHFFFAOYSA-N oxacyclotridecan-7-one Chemical compound O1CCCCCC(CCCCCC1)=O RYTAUJFRHPXPRS-UHFFFAOYSA-N 0.000 description 1
- GHZRKQCHJFHJPX-UHFFFAOYSA-N oxacycloundecan-2-one Chemical compound O=C1CCCCCCCCCO1 GHZRKQCHJFHJPX-UHFFFAOYSA-N 0.000 description 1
- IGBKCWJHWBSRPY-UHFFFAOYSA-N oxacycloundecan-3-one Chemical compound O1CC(CCCCCCCC1)=O IGBKCWJHWBSRPY-UHFFFAOYSA-N 0.000 description 1
- UUIRPWJYOBMBGR-UHFFFAOYSA-N oxacycloundecan-4-one Chemical compound O1CCC(CCCCCCC1)=O UUIRPWJYOBMBGR-UHFFFAOYSA-N 0.000 description 1
- PKFCSFIKZMTGRQ-UHFFFAOYSA-N oxacycloundecan-5-one Chemical compound O=C1CCCCCCOCCC1 PKFCSFIKZMTGRQ-UHFFFAOYSA-N 0.000 description 1
- BKLCHLFZWGQVJP-UHFFFAOYSA-N oxacycloundecan-6-one Chemical compound O1CCCCC(CCCCC1)=O BKLCHLFZWGQVJP-UHFFFAOYSA-N 0.000 description 1
- URUUZIAJVSGYRC-UHFFFAOYSA-N oxan-3-one Chemical compound O=C1CCCOC1 URUUZIAJVSGYRC-UHFFFAOYSA-N 0.000 description 1
- JMJRYTGVHCAYCT-UHFFFAOYSA-N oxan-4-one Chemical compound O=C1CCOCC1 JMJRYTGVHCAYCT-UHFFFAOYSA-N 0.000 description 1
- YUIJSWMGTIISBE-UHFFFAOYSA-N oxepan-3-one Chemical compound O=C1CCCCOC1 YUIJSWMGTIISBE-UHFFFAOYSA-N 0.000 description 1
- CTLGXFASUHKSGN-UHFFFAOYSA-N oxepan-4-one Chemical compound O=C1CCCOCC1 CTLGXFASUHKSGN-UHFFFAOYSA-N 0.000 description 1
- 125000003585 oxepinyl group Chemical group 0.000 description 1
- BTLSLHNLDQCWKS-UHFFFAOYSA-N oxocan-2-one Chemical compound O=C1CCCCCCO1 BTLSLHNLDQCWKS-UHFFFAOYSA-N 0.000 description 1
- JNDCCAGMQAHZNT-UHFFFAOYSA-N oxocan-3-one Chemical compound O=C1CCCCCOC1 JNDCCAGMQAHZNT-UHFFFAOYSA-N 0.000 description 1
- IKPVWKOTNSETJG-UHFFFAOYSA-N oxocan-4-one Chemical compound O1CCC(CCCC1)=O IKPVWKOTNSETJG-UHFFFAOYSA-N 0.000 description 1
- CPFWITMGMTXZQJ-UHFFFAOYSA-N oxocan-5-one Chemical compound O=C1CCCOCCC1 CPFWITMGMTXZQJ-UHFFFAOYSA-N 0.000 description 1
- JLPJFSCQKHRSQR-UHFFFAOYSA-N oxolan-3-one Chemical compound O=C1CCOC1 JLPJFSCQKHRSQR-UHFFFAOYSA-N 0.000 description 1
- ZYDGQQTXLBNSGJ-UHFFFAOYSA-N oxonan-2-one Chemical compound O=C1CCCCCCCO1 ZYDGQQTXLBNSGJ-UHFFFAOYSA-N 0.000 description 1
- FMXFFKVLOJPMSL-UHFFFAOYSA-N oxonan-3-one Chemical compound O1CC(CCCCCC1)=O FMXFFKVLOJPMSL-UHFFFAOYSA-N 0.000 description 1
- RKPUYTIUGXZNED-UHFFFAOYSA-N oxonan-4-one Chemical compound O1CCC(CCCCC1)=O RKPUYTIUGXZNED-UHFFFAOYSA-N 0.000 description 1
- MLBGIDINXVPGSF-UHFFFAOYSA-N oxonan-5-one Chemical compound O1CCCC(CCCC1)=O MLBGIDINXVPGSF-UHFFFAOYSA-N 0.000 description 1
- NFGGQMYSOLVBLF-UHFFFAOYSA-N phenyl(1h-pyrrol-2-yl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CN1 NFGGQMYSOLVBLF-UHFFFAOYSA-N 0.000 description 1
- GCSHUYKULREZSJ-UHFFFAOYSA-N phenyl(pyridin-2-yl)methanone Chemical compound C=1C=CC=NC=1C(=O)C1=CC=CC=C1 GCSHUYKULREZSJ-UHFFFAOYSA-N 0.000 description 1
- RYMBAPVTUHZCNF-UHFFFAOYSA-N phenyl(pyridin-3-yl)methanone Chemical compound C=1C=CN=CC=1C(=O)C1=CC=CC=C1 RYMBAPVTUHZCNF-UHFFFAOYSA-N 0.000 description 1
- SKFLCXNDKRUHTA-UHFFFAOYSA-N phenyl(pyridin-4-yl)methanone Chemical compound C=1C=NC=CC=1C(=O)C1=CC=CC=C1 SKFLCXNDKRUHTA-UHFFFAOYSA-N 0.000 description 1
- DWYFUJJWTRPARQ-UHFFFAOYSA-N phenyl(thiophen-2-yl)methanone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CS1 DWYFUJJWTRPARQ-UHFFFAOYSA-N 0.000 description 1
- USISRUCGEISZIB-UHFFFAOYSA-N piperidin-3-one Chemical compound O=C1CCCNC1 USISRUCGEISZIB-UHFFFAOYSA-N 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- QGKLPGKXAVVPOJ-UHFFFAOYSA-N pyrrolidin-3-one Chemical compound O=C1CCNC1 QGKLPGKXAVVPOJ-UHFFFAOYSA-N 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- LISFMEBWQUVKPJ-UHFFFAOYSA-N quinolin-2-ol Chemical compound C1=CC=C2NC(=O)C=CC2=C1 LISFMEBWQUVKPJ-UHFFFAOYSA-N 0.000 description 1
- 229930185107 quinolinone Natural products 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000012764 semi-quantitative analysis Methods 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 238000013207 serial dilution Methods 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 238000002207 thermal evaporation Methods 0.000 description 1
- CFFCKUISNHPWOW-UHFFFAOYSA-N thian-2-one Chemical compound O=C1CCCCS1 CFFCKUISNHPWOW-UHFFFAOYSA-N 0.000 description 1
- ATAMXDLUUTYFKT-UHFFFAOYSA-N thian-3-one Chemical compound O=C1CCCSC1 ATAMXDLUUTYFKT-UHFFFAOYSA-N 0.000 description 1
- OVRJVKCZJCNSOW-UHFFFAOYSA-N thian-4-one Chemical compound O=C1CCSCC1 OVRJVKCZJCNSOW-UHFFFAOYSA-N 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- UCUOVADUXMCWOM-UHFFFAOYSA-N thiepan-2-one Chemical compound O=C1CCCCCS1 UCUOVADUXMCWOM-UHFFFAOYSA-N 0.000 description 1
- MEUUMPRYCUQAQB-UHFFFAOYSA-N thiepan-3-one Chemical compound O=C1CCCCSC1 MEUUMPRYCUQAQB-UHFFFAOYSA-N 0.000 description 1
- FQCDOWQBRJYXCP-UHFFFAOYSA-N thiepan-4-one Chemical compound O=C1CCCSCC1 FQCDOWQBRJYXCP-UHFFFAOYSA-N 0.000 description 1
- 125000003777 thiepinyl group Chemical group 0.000 description 1
- KYSLXZJXRBTXDF-UHFFFAOYSA-N thietan-2-one Chemical compound O=C1CCS1 KYSLXZJXRBTXDF-UHFFFAOYSA-N 0.000 description 1
- DQOHDRDDPZNSQI-UHFFFAOYSA-N thietan-3-one Chemical compound O=C1CSC1 DQOHDRDDPZNSQI-UHFFFAOYSA-N 0.000 description 1
- YEEYWNJJZHIZLF-UHFFFAOYSA-N thiiran-2-one Chemical compound O=C1CS1 YEEYWNJJZHIZLF-UHFFFAOYSA-N 0.000 description 1
- VVGBVIKEALGJBR-UHFFFAOYSA-N thiocan-2-one Chemical compound O=C1CCCCCCS1 VVGBVIKEALGJBR-UHFFFAOYSA-N 0.000 description 1
- ZWFMISBAECZWOX-UHFFFAOYSA-N thiocan-3-one Chemical compound O=C1CCCCCSC1 ZWFMISBAECZWOX-UHFFFAOYSA-N 0.000 description 1
- ZSGQKIFSLSBWLR-UHFFFAOYSA-N thiocan-4-one Chemical compound O=C1CCCCSCC1 ZSGQKIFSLSBWLR-UHFFFAOYSA-N 0.000 description 1
- JEAURVWXXNRTKE-UHFFFAOYSA-N thiocan-5-one Chemical compound O=C1CCCSCCC1 JEAURVWXXNRTKE-UHFFFAOYSA-N 0.000 description 1
- DSXFPRKPFJRPIB-UHFFFAOYSA-N thiolan-3-one Chemical compound O=C1CCSC1 DSXFPRKPFJRPIB-UHFFFAOYSA-N 0.000 description 1
- BXFVHTVEKYSYMP-UHFFFAOYSA-N thionan-2-one Chemical compound O=C1CCCCCCCS1 BXFVHTVEKYSYMP-UHFFFAOYSA-N 0.000 description 1
- VNXBHORBHKWBOQ-UHFFFAOYSA-N thionan-3-one Chemical compound S1CC(CCCCCC1)=O VNXBHORBHKWBOQ-UHFFFAOYSA-N 0.000 description 1
- ZFAZQLAFICLFCV-UHFFFAOYSA-N thionan-4-one Chemical compound S1CCC(CCCCC1)=O ZFAZQLAFICLFCV-UHFFFAOYSA-N 0.000 description 1
- VBEYRRXYDXTHIY-UHFFFAOYSA-N thionan-5-one Chemical compound O=C1CCCCSCCC1 VBEYRRXYDXTHIY-UHFFFAOYSA-N 0.000 description 1
- XKGLSKVNOSHTAD-UHFFFAOYSA-N valerophenone Chemical compound CCCCC(=O)C1=CC=CC=C1 XKGLSKVNOSHTAD-UHFFFAOYSA-N 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 238000004065 wastewater treatment Methods 0.000 description 1
- 238000003911 water pollution Methods 0.000 description 1
- PAPBSGBWRJIAAV-UHFFFAOYSA-N ε-Caprolactone Chemical compound O=C1CCCCCO1 PAPBSGBWRJIAAV-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01D—COMPOUNDS OF ALKALI METALS, i.e. LITHIUM, SODIUM, POTASSIUM, RUBIDIUM, CAESIUM, OR FRANCIUM
- C01D3/00—Halides of sodium, potassium or alkali metals in general
- C01D3/04—Chlorides
- C01D3/06—Preparation by working up brines; seawater or spent lyes
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/26—Treatment of water, waste water, or sewage by extraction
- C02F1/265—Desalination
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/68—Treatment of water, waste water, or sewage by addition of specified substances, e.g. trace elements, for ameliorating potable water
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/10—Inorganic compounds
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2103/00—Nature of the water, waste water, sewage or sludge to be treated
- C02F2103/08—Seawater, e.g. for desalination
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2305/00—Use of specific compounds during water treatment
- C02F2305/14—Additives which dissolves or releases substances when predefined environmental conditions are reached, e.g. pH or temperature
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A20/00—Water conservation; Efficient water supply; Efficient water use
- Y02A20/124—Water desalination
- Y02A20/131—Reverse-osmosis
Definitions
- the present disclosure relates to a salt recovery solution and to a process for separating a salt from an aqueous solution.
- the present disclosure also relates to a salt recovery solution and to its use to concentrate a salt or brine solution by recovering water therefrom.
- Zero liquid discharge is an ambitious wastewater management strategy that eliminates any liquid waste leaving the plant or facility boundary, with the majority of water being recovered for reuse. Achieving ZLD, however, is generally characterized by intensive use of energy and high cost. As a result, ZLD has long been considered technically but not economically viable and has been applied only in limited cases.
- RO Reverse osmosis
- ED electrodialysis
- FO forward osmosis
- MD membrane distillation
- the present invention provides a salt recovery solution suitable for recovering a salt from a first aqueous solution, the salt recovery solution comprising:
- the salt recovery solution comprises:
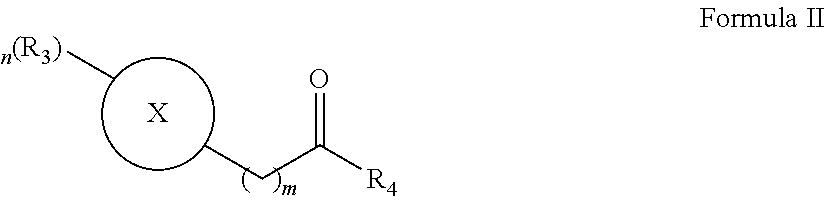
- the salt recovery solution comprises at least one enolisable carbonyl of Formula II,
- X is a 6-membered aromatic monocyclic.
- R 4 is a a C 1 -C 7 alkyl optionally substituted with a halo, a hydroxy, a —O—(C 1 -C 7 alkyl) or a C 3 -C 7 monocyclic.
- the enolisable carbonyl of Formula II is selected from acetophenone and 4-phenyl-2butanone.
- the present invention provides a process for recovering a salt from a first aqueous solution, the process including the step of
- the process is a zero-liquid discharge process.
- the process is a non-membrane process.
- the present invention provides a process for concentrating a salt containing first aqueous solution, the process comprising the steps of:
- the precipitated salt forms part of an aqueous layer distinct from the salt recovery solution.
- the process is a non-membrane process.
- the process is a non-osmotic process.
- the process is a non-membrane and non-osmotic process.
- the process concentrates the first aqueous solution by at least 20%. In other embodiments the process concentrates the first aqueous solution by at least 30%, or by at least 40%, or by at least 50% or by at least 60%, or by at least 70% or by at least 80% or by at least 90%.
- the process is a minimal discharge process.
- the process is a zero-liquid discharge process.
- FIG. 1 shows schematically a salt recovery process of the present invention.
- FIG. 2 shows gas chromatograms of the additive or regenerant solution overlaid with the additive or regenerant solution which has been used to remove water from a wet salt recovery solution.
- FIG. 4 shows a calibration curve of NaCl solutions against osmotic pressure measurements.
- FIG. 6 shows a graph of the osmotic pressure of NaCl solution after concentration using varying ratios of salt recovery solution (TEA:MEK).
- FIG. 7 shows a bar graph of the concentration of NaCl solution after concentration using varying ratios of N-Ethylpiperidine:Cyclohexanone salt recovery solution.
- FIG. 8 shows a graph of the osmotic pressure of NaCl solution after concentration using varying ratios of using salt recovery solution (N-Ethylpiperidine:Cyclohexanone).
- FIG. 10 shows a bar graph of the concentration of NH 4 SO 4 solution after concentration using varying ratios of salt recovery solution (TEA:MEK).
- the term “about” or “approximately” usually means within 20%, more preferably within 10%, and most preferably still within 5% of a given value or range. Alternatively, the term “about” means within a log (i.e., an order of magnitude) preferably within a factor of two of a given value.
- additive means an additive that releases water held within the salt recovery solution.
- a non-limiting example of a suitable additive that has been shown to release water bound within the salt recovery solution is citric acid or a citric acid solution.
- the word “regenerant” is also used interchangeably with the work “additive”. This is because the additive regenerates the salt recovery solution.
- brine or “brine solution” as used throughout the specification means an aqueous solution having a concentration of a salt in the water.
- the salt in the water could include sodium chloride, however, other salt solutions are envisaged, including ammonium sulfate or the like.
- concentration of the salt in the aqueous solution may range from about 3.5% (typical concentration of seawater) through to much higher concentrations, such as 25%, which would include a salt water solution used for brining foods.
- Other brine waste water solutions from textile processing, the semiconductor industry or oil and gas industry would also be applicable for use with the current salt recovery solution and processes defined herein.
- zero liquid discharge means a wastewater treatment process where no effluent, or discharge, is left over.
- C 1 -C 7 alkyl refers to a fully saturated branched or unbranched hydrocarbon moiety, which may be a straight or a branched chain of a particular range of 1-7 carbons.
- the alkyl comprises 1 to 7 carbon atoms, or 1 to 4 carbon atoms.
- C 1 -C 7 alkyl include, but are not limited to, methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl, 3-methylhexyl, 2,2-dimethylpentyl, 2,3-dimethylpentyl, n-heptyl, and the like.
- C 3 -C 7 monocyclic is a 3-, 4-, 5-, 6-, or 7-membered saturated or unsaturated monocyclic ring.
- Representative C 3 -C 7 monocyclic groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, phenyl, and cycloheptyl.
- 3- to 15-membered monocyclic ketone refers to a 3- to 15-membered non-aromatic monocyclic ring system containing a ketone functional group.
- Representative examples of a 3- to 15-membered monocyclic ketone include, but are not limited to cyclopropanone, cyclobutanone, cyclopentanone, cyclohexanone, cycloheptanone, cyclooctanone, cyclononanone, cyclodecanone, cycloundecanone, cyclododecanone, cyclotridecanone; cyclotetradecanone and cyclopentadecanone.
- the 3- to 15-membered monocyclic ketone may be substituted with one or more of the following groups -halo, —OH, —CN, —NO 2 , —C ⁇ CH, —SH, —C 1 -C 7 alkyl, —(C 1 -C 7 alkyl)-OH, —NH 2 , —NH(C 1 -C 7 alkyl), —NH(C 1 -C 7 alkyl) 2 , —O(C 1 -C 7 alkyl), —C(O)—O(—C 1 -C 7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C 1 -C 7 alkyl).
- groups -halo —OH, —CN, —NO 2 , —C ⁇ CH, —SH, —C 1 -C 7 alkyl, —(C 1 -C 7 alkyl)-OH, —NH 2
- 8-15 membered aromatic system optionally including one or more heteroatoms selected from S, N or O includes naphthaleneone, fluorenone, quinolinone and indanone ring systems, optionally substituted with halo, hydroxy, nitro, —SH, —C 1 -C 7 alkyl and —O(C 1 -C 7 alkyl).
- Such compounds of Formula I include, without limitation napthalenone, 2,2,4-trichloro-1(2h)-naphthalenone, 4-methyl-1h-naphthalen-2-one, 9-fluorenone, 9-fluorenone oxime, 2-nitro-9-fluorenone, 3-nitro-9-fluorenone, 4-nitro-9-fluorenone, 2,6-dinitro-9-fluorenone, 2,7-dinitro-9-fluorenone, 2,3,7-trinitro-9-fluorenone, 2-fluoro-9-fluorenone, 1-bromo-9-fluorenone, 2-bromo-9-fluorenone, 2,7-dichloro-9-fluorenone, 2,7-dibromo-9-fluorenone, 2-hydroxy-9-fluorenone, 4-hydroxy-9-fluorenone, 1-methylfluoren-9-one, 4-methylfluoren-9-one 11-benzo(a)fluorenone, benzo(b)fluorenone, 1h-
- 5-8 membered aromatic monocyclic optionally containing a heteroatom includes without limitation 5-membered rings such as pyrrolyl, furanyl, thiophenyl; 6-membered rings such as phenyl, pyridinyl, pyranyl, thiopyranyl and 7-membered rings such as azepinyl, oxepinyl, thiepinyl.
- 3- to 15-membered monocyclic heterocyclic ketone refers to: (i) a 3- or 4-membered non-aromatic monocyclic cycloalkyl in which 1 of the ring carbon atoms has been replaced with an N, O or S atom; or (ii) a 5- to 15-membered non-aromatic monocyclic cycloalkyl in which 1-4 of the ring carbon atoms have been independently replaced with a N, O or S atom.
- a 3- to 15-membered monocyclic heterocyclic ketone having one N, O or S atom include, but are not limited to oxiran-2-one, thiiran-2-one, oxetan-2-one, oxetan-3-one, azetidin-3-one, thietan-2-one, thietan-3-one, dihydrofuran-2(3H)-one, dihydrofuran-3(2H)-one, pyrrolidin-3-one, dihydrothiophen-3(2H)-one, dihydrothiophen-2(3H)-one, tetrahydro-2H-pyran-2-one, dihydro-2H-pyran-3(4H)-one, dihydro-2H-pyran-4(3H)-one, piperidin-3-one, piperidin-4-one, tetrahydro-2H-thiopyran-2-one, dihydro-2H-thiopyran-3(4H)-
- the 3- to 15-membered monocyclic heterocyclic ketone group may be substituted with one or more of the following groups -halo, —OH, —CN, —NO 2 , —C ⁇ CH, —SH, —C 1 -C 6 lower alkyl, —(C 1 -C 7 alkyl)-OH, —NH 2 , —NH(C 1 -C 7 alkyl), —N(C 1 -C 7 alkyl) 2 , —O(C 1 -C 7 alkyl), —C(O)—O(—C 1 -C 7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C 1 -C 7 alkyl).
- the 3-5 membered monocyclic heterocyclic ketone does not include any amide groups where the ketone enolisable carbonyl group is adjacent a N atom in the cyclic structure
- halo refers to —F, —Cl, —Br or —I.
- an enolisable carbonyl means a compound that has one or more carbonyl functional groups and wherein at least one of the carbonyl functional groups has alpha hydrogens (H ⁇ ) that may be removed by a base to form an enolate and then an enol as shown in the reaction scheme below.
- enolisable carbonyl does not include a compound having solely an aldehyde functional group, a compound having solely a carboxylic acid functional group, a compound having solely an amide functional group, a compound having solely an acyl halide functional group or acetylacetone.
- the enolisable carbonyls of the invention include without limitation those exemplified in the specification and defined by Formulae I and II, and without limitation also include the following enolisable carbonyls: 1-acetonapthone, 2-acetonaphthone, 4-methyl-1-acetonaphthone, 1′-hydroxy-2′-acetonaphthone, 2′-hydroxy-1′-acetonaphthone, 2-methoxy-1-acetonaphthone, 4-fluoro-1-acetonapthone; 2-acetylphenanthrene, 3-acetylphenanthrene, 4-acetylphenanthrene, 9-acetylphenanthrene, 6-bromo-9-acetylphenanthrene, 9-fluoro-10-acetylphenanthrene, 9-fluorenone, 9-fluorenone oxime, 2-nitro-9-fluorenone, 3-nitro-9-fluorenone, 4-nitro-9-flu
- tertiary amine containing compound preferably means a compound having at least one tertiary amine group, but it is to be appreciated that the compound may have more than one tertiary amine group or further may be a mixture of tertiary amine containing compounds.
- the tertiary amine containing compound is a base, such as a Lewis base. If the base is a Lewis base, it is envisaged that a Lewis adduct may be formed with the enolisable carbonyl.
- the tertiary amine containing compound is immiscible with water at or above 20 degrees Celsius under one standard atmosphere of pressure.
- the solution may include a combination of more than one tertiary amine containing compound.
- the tertiary amine containing compound may be aliphatic, conjugated, asymmetric or cyclic or a combination thereof.
- Suitable tertiary amine containing compounds include the following:
- the tertiary amine containing compound is selected from 1-ethyl pyrrolidine, 2-methylpyridine, N-methylpiperidine and 4-ethylmorpholine.
- the tertiary amine containing compound is selected from a —N(C 1 -C 7 alkyl) 3 . In another embodiment the tertiary amine containing compound is selected from a —N(C 1 -C 4 alkyl) 3 . In yet a further embodiment the tertiary amine containing compound is —N(C 2 alkyl) 3 (triethylamine).
- the present invention is directed to a salt recovery solution suitable for recovering a salt from a first aqueous solution, the salt recovery solution comprising:
- the process is a minimal liquid discharge process.
- the process includes the further step of adding an additive to the salt recovery solution to further release water held within the salt recovery solution.
- the additive is citric acid.
- the citric acid is a concentrated solution of citric acid comprising between about 200-450 gms of citric acid per litre of water.
- the citric acid is anhydrous citric acid added directly to the salt recovery solution.
- the molar ratio of the tertiary amine containing compound to the enolisable carbonyl may vary widely and may be from about 1:99 or 99:1; or from about 1:50 or 50:1 or from about 1:10 or 10:1 or from about 1:5 or 5:1 or from about 1:3 or from about 3:1 or from about 1:2 or from about 2:1. In a preferred embodiment the molar ratio is about 1:1.
- a chemistry technician would be able to routinely determine the most suitable molar ratio depending on the purpose for which the salt recovery solution is to be employed.
- the first aqueous solution is salt water or a brine solution.
- a salt recovery solution was prepared using triethylamine (TEA) and methyl ethyl ketone (MEK) in the molar ratio of 0.5:1.
- TAA triethylamine
- MEK methyl ethyl ketone
- a saturated sodium chloride salt solution was introduced to the salt recovery solution—see step 1 of FIG. 1 .
- the water formed the aqueous layer with salt precipitate at the bottom of the vial.
- the salt recovery solution along with the aqueous layer was decanted and the salt was then dried. This demonstrated the recovery of salt from a saturated salt solution using a salt recovery solution and with no liquid discharge.
- the input salt concentrations were measured, and the dry weights of samples recorded containing 0.2 mL, 0.5 mL and 1 mL of both 50 000 ppm NaCl solution and 100 000 ppm NaCl solution respectively.
- the input salt results are shown in (Table 3) and the percent salt recovery results are shown in Table 4.
- the output salt concentrations were also determined as follows.
- Volume Volume Dry weight Salt recovery % of salt of brine of salt Dry weight of recovery solution precipitated* salt output/Dry solution added (output) weight of salt Brine solution (mL) (mL) avg in mg input ⁇ 100 50 000 ppm NaCl 10.0000 0.2000 0.0112 112.0000 solution 50 000 ppm NaCl 15.0000 0.4000 0.0209 104.5000 solution 50 000 ppm NaCl 10.0000 0.2000 0.0107 107.0000 solution 100 000 ppm NaCl 10.0000 0.2000 0.0243 121.5000 solution 100 000 ppm NaCl 10.0000 0.2000 0.0206 103.0000 solution 100 000 ppm NaCl 10.0000 0.4000 0.0507 126.7500 solution *The dry weights are the average values of each set of two trials.
- Salt recovery From the above results, it can be seen that it is possible to achieve around 100% salt and water recovery from the brine solution when the volume of brine added is just below the maximum absorption capacity of the salt recovery solution. The deviation in salt recoveries can be attributed to the unavoidable error to the small scale of the experiments.
- the salt rejection percentage was estimated to show that only water was being absorbed by the salt recovery solution from the brine solution and that the salt recovery solution was rejecting substantially all of the salt from the brine.
- the conductivity of pure water, the neat brine solution and the concentrated salt recovery solution were measured.
- the salt rejection % was calculated by the following formula:
- the salt rejection % of over 99 is indicative of the fact that there is complete precipitation of salt from the brine solution and there are no residual salts going into the salt recovery solution.
- the salt recovery solution can achieve close to 100% salt and water recovery by minimizing the volume of feed solution in a thermo-efficient way unlike the traditional treatment systems.
- the lab scale results are indicating that there are no salts going into the salt recovery solution during the absorption process and that the purity of absorbed water is relatively high.
- Brine Sample 1 has the highest capacity at 0.81 mL (+/ ⁇ 0.04) and Brine Sample 2 having the lowest at 0.15 mL (+/ ⁇ 0.03).
- This reflects the differing salt compositions and concentrations of each of the commercial brine samples and demonstrates the importance of knowing what the composition of the brine is before salt recovery treatment.
- the salt recovery solution would be diluted and would be unable to absorb water and precipitate salts effectively. It is to be appreciated that for large scale processes, it would be desirable to utilize higher ratios of salt recovery solution relative to the brine solution to ensure a total absorption of water and avoid brine carry-over.
- the brine and the salt recovery solution were mixed and resulted in the salt precipitating out.
- the Samples were left for 20 mins to ensure full separation of the solid salts.
- the now wet salt recovery solution was decanted into a clean vial using a 5 mL syringe to carefully remove the wet salt recovery solution from the precipitated salts without transferring any salts into the fresh vial (approx. 17 mL, measuring the actual quantity in each individual case).
- the salt slurry left behind was heated at 110° C. for 1 hour to remove the left-over salt recovery material.
- the dried salt was cooled and then weighed.
- the weight of the clean vial was deducted to obtain the weight of salt recovered. The results are tabulated in Table 8.
- the salt recovery percentage was determined by the following formula.
- Table 8 above details the raw data collected to calculate the percentage of salt recovered.
- the “salt expected” was calculated by evaporating the same amount of raw brine as was added to each sample, in a heat block at 110° C. This measured how much salt was in each brine sample and allowed for the calculation of what percentage of this initial brine was collected (see Table 9).
- the decanted and wet salt recovery material was regenerated using a regenerating agent/regenerant (0.3-0.5 mL) using the following method.
- the regenerating agent which was a concentrated solution of citric acid (412 g/L) or anhydrous citric acid was added to the diluted/wet salt recovery solution from the previous step to separate the water absorbed by the salt recovery solution. It has been found, and gas chromatographic data has shown that some of the salt recovery solution is dissolved into the regenerant. With reference to FIG. 2 , it can be seen that the gas chromatogram of fresh regenerant shows a small peak at 2.8 mins and a larger slightly trailing peak at 3.0 mins. This larger peak is water which is used as the solvent for the regenerant.
- the tail is caused by the regenerant material bonding with water molecules.
- the trailing peak at 4.1 mins is highly characteristic of the salt recovery solutions and clearly indicates that some of the salt recovery solution has leached into the regenerant. Because of the trailing shape of this peak and the overlap of the first peaks, it is not possible to accurately quantify the amount of salt recovery solution which has crossed over into the regenerant, however, semi-quantitative analysis of multiple samples has suggested that up to about 15% of the salt recovery solution has crossed over into the regenerant. To minimise this cross over, the amount of regenerating agent used is selected to be as little as possible. In order to determine the amount of water recovered from the process a known volume of regenerating agent was added to the diluted salt recovery solution.
- the regenerating agent removed the water from the diluted salt recovery solution to leave a concentrated salt recovery solution. Because of the difference in density between the regenerating agent and the concentrated salt recovery solution, the concentrated salt recovery solution (now non-aqueous or dry) rises to the top while the now diluted regenerating agent sinks to the bottom of the vial. The bottom aqueous layer was carefully removed to a measuring cylinder and the volume recovered was recorded in Table 10. The volume of the aqueous layer was measured using the following formula.
- volume of water recovered (mL) Volume of aqueous phase (mL) ⁇ Volume of regenerant (mL)
- the water recovery % was calculated by the formula (Table 10):
- Table 11 shows the average water and salt recoveries across three samples for each of the brines tested. The majority of the salt recoveries are over 100%. This perhaps indicates some interaction between the salts in the brine and the salt recovery solution which results in the salts binding with the salt recovery solution, resulting in an increase in weight compared to when water is removed by evaporation alone.
- FIG. 3 shows an example gas chromatograph of Brine Sample 1 focusing on the water peak.
- the black (solid line) trace is the salt recovery solution before the addition of the brine sample.
- the second trace (long dash line) shows the water content after the brine sample has been added and shows a clear increase in intensity.
- the third trace (dotted line) shows the water content after the regenerant has been used to release the water from or dry the salt recovery solution, and clearly shows the water content has largely returned to the same prior to brine sample addition. This shows that the salt recovery solution has been regenerated and is ready for reuse.
- Parameter Setting Injection Volume 1.0 ⁇ L Injection temperature 250° C.
- Injection mode Split Split ratio 100.0 Carrier gas He Carrier gas pressure 53.1 kPa Column flow 0.93 mL/min Liner velocity 22.0 cm/s Column length 30.0 m Column inner diameter 0.32 Column method Isocratic Column temperature 163.0° C. Total time 9 min Detector TCD TCD sample rate 40 ms TCD current 70 mA Makeup gas He Makeup flow 8.0 mL/min TCD temperature 200° C.
- Table 12 shows the quantity of brine water (measured by GC) in the salt recovery solution after it has been used to absorb water and the quantity of brine water left in the salt recovery solution after it has been regenerated. This provides a measure of how effective the regenerant has been at removing water from the salt recovery solution. As can be seen from Table 12, the quantity of water remaining in the salt recovery solution after regeneration is very small. This shows that the citric acid regenerant has been very effective in removing most of the brine water from the salt recovery solution.
- salt recovery solutions were made up with amine(s) and ketone(s) in various specified molar ratios (see Tables 13-17 below).
- brine 300 000 ppm NaCl was slowly added to 5 mL of the selected salt recovery solution.
- the brine was absorbed by the salt recovery solution and the salts in the brine precipitated out.
- the addition of the brine was stopped as soon as a separate phase started to form at the bottom of the vial after shaking. The presence of the separate phase shows that the maximum absorption capacity of the salt recovery solution has been reached. At this point the volume of the brine, which has been added to the salt recovery solution is recorded.
- Table 13 summarises the maximum absorbing capacity of the salt recovery solution, along with the water recovery and the salts recovery of the different salt recovery solutions containing different amines with 2-butanone. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 13. The salt and water recovery results listed in this table record one-off tests.
- the following table 14 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing triethylamine combined with different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following table 14. The salt and water recovery results listed in this table record one-off tests.
- the following table 15 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to three different amines combined with cyclohexanone. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 15. The salt and water recovery results listed in this table record one-off tests.
- the following table 16 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to two different amines combined with up to two different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 16. The salt and water recovery results listed in this table record one-off tests.
- the following table 17 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to three different amines combined with up to three different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 16. The salt and water recovery results listed in this table record one-off tests.
- Salt recovery solutions were prepared using: Triethylamine (TEA) and methyl ethyl ketone (MEK), N-Ethylpiperidine and cyclohexanone in the molar ratio of 0.5:1.
- Example 5 The experiment described in Example 5 was repeated, except the 5% NaCl solution was replaced with a 20% (w/w) ammonium sulfate (NH 4 SO 4 ) solution.
- Salt recovery solutions were prepared using: Triethylamine (TEA) and methylethylketone (MEK).
- the results shown in FIG. 10 and Table 20 show that the salt recovery solution (TEA:MEK) is capable of concentrating a salt solution to a higher final concentration. Essentially water has been recovered from the initial NH 4 SO 4 solution and the water has moved into the salt recovery solution effectively leading to the concentration of the initial NH 4 SO 4 solution.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Environmental & Geological Engineering (AREA)
- Water Supply & Treatment (AREA)
- Life Sciences & Earth Sciences (AREA)
- Hydrology & Water Resources (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Extraction Or Liquid Replacement (AREA)
- Hydrogenated Pyridines (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
Abstract
Description
- The present disclosure relates to a salt recovery solution and to a process for separating a salt from an aqueous solution. The present disclosure also relates to a salt recovery solution and to its use to concentrate a salt or brine solution by recovering water therefrom.
- The extraction of salts from an aqueous solution is typically a high energy and time consuming process, requiring removal of water and salt crystallisation. As reported in 2016 by Tong et al. American Chemical Society 6846 DOI: 10.1021/acs.est.6b01000 Environ. Sci. Technol. 2016, 50, 6846-6855, Zero liquid discharge (ZLD) is an ambitious wastewater management strategy that eliminates any liquid waste leaving the plant or facility boundary, with the majority of water being recovered for reuse. Achieving ZLD, however, is generally characterized by intensive use of energy and high cost. As a result, ZLD has long been considered technically but not economically viable and has been applied only in limited cases. In recent years, greater recognition of the dual challenges of water scarcity and pollution of aquatic environments has revived global interest in ZLD. More stringent regulations, rising expenses for wastewater disposal, and increasing value of freshwater are driving ZLD to become a beneficial or even a necessary option for wastewater management. The global market for ZLD is estimated to reach an annual investment of at least $100-200 million spreading rapidly from developed countries in North America and Europe to emerging economies such as China and India. Early ZLD systems were based on stand-alone thermal processes, where wastewater was typically evaporated in a brine concentrator followed by a brine crystallizer or an evaporation pond. The condensed distillate water in ZLD systems is collected for reuse, while the produced solids are either sent to a landfill or recovered as valuable salt by-products. Such systems, which have been in successful operation for 40 years and are still being built, require considerable energy and capital. Reverse osmosis (RO), a membrane-based technology widely applied in desalination, has been incorporated into ZLD systems to improve energy and cost efficiencies. However, RO, although much more energy efficient than thermal evaporation, can be applied only to feedwaters with a limited salinity range. Accordingly, other salt-concentrating technologies that can treat higher salinity feedwaters, such as electrodialysis (ED), forward osmosis (FO), and membrane distillation (MD), have emerged recently as alternative ZLD technologies to further concentrate wastewater beyond RO. Although ZLD holds great promise to reduce water pollution and augment water supply, its viability is determined by a balance among the benefits associated with ZLD, energy consumption, and capital/operation costs.
- It is an object of the present invention to provide a solution that overcomes these difficulties or to at least provide a useful alternative.
- In a first aspect, the present invention provides a salt recovery solution suitable for recovering a salt from a first aqueous solution, the salt recovery solution comprising:
-
- a) at least one tertiary amine containing compound; and
- b) at least one enolisable carbonyl.
- In one embodiment the salt recovery solution comprises:
-
- a) at least one tertiary amine containing compound; and
- b) at least one enolisable carbonyl of Formula I,
- wherein
-
- c) R1 and R2 are independently selected from a —C1-C7 alkyl or a —C3-C7 monocyclic; or
- d) one of R1 or R2 is selected from a —O—(C1-C7 alkyl) and the other is selected from a —C1-C7 alkyl, or
- e) R1 and R2 together, with the carbonyl of Formula I, form a 3-15 membered monocyclic ketone or a 3-15 membered monocyclic heterocyclic ketone.
- In one embodiment, the salt recovery solution comprises:
-
- a) at least one tertiary amine containing compound; and
- b) at least one enolisable carbonyl of Formula I,
- wherein
- R1 and R2 together with the carbonyl of Formula I form an 8-15 membered aromatic system optionally including one or more heteroatoms selected from S, N or O.
- In another embodiment the salt recovery solution comprises at least one enolisable carbonyl of Formula II,
-
- wherein X is a 5-8 membered aromatic monocyclic optionally containing a heteroatom selected from S, N or O;
- each R3, if present, is selected independently from a halo, a hydroxy, a —NO2, a C1-C7 alkyl, or a —O—(C1-C7 alkyl);
- R4 is selected from a halo, a —O—(C1-C7 alkyl); a C1-C7 alkyl optionally substituted with a halo, a hydroxy, a —O—(C1-C7 alkyl) or a C3-C7 monocyclic; or a C3-C7 monocyclic optionally substituted with a halo, a hydroxy, a C1-C7 alkyl, or a —O—(C1-C7 alkyl);
- m is 0, 1, 2 or 3 carbons, each carbon being optionally substituted with one or more substituents selected from a halo, hydroxy, a C1-C7 alkyl optionally substituted with a halo or a hydroxy; a C1-C7 alkyl, a —O—(C1-C7 alkyl) or a C3-C7 monocyclic; and
- n is 0, 1, 2 or 3.
- In one embodiment, X is a 6-membered aromatic monocyclic.
- In one embodiment R4 is a a C1-C7 alkyl optionally substituted with a halo, a hydroxy, a —O—(C1-C7 alkyl) or a C3-C7 monocyclic.
- In one embodiment the enolisable carbonyl of Formula II is selected from acetophenone and 4-phenyl-2butanone.
- In another aspect, the present invention provides a process for recovering a salt from a first aqueous solution, the process including the step of
-
- (a) adding the salt containing first aqueous solution to a salt recovery solution; and
- (b) allowing the salt to precipitate on passage through the salt recovery solution.
- In one embodiment the process is a zero-liquid discharge process.
- Wherein the process is a non-membrane process.
- Wherein the process is a non-osmotic process.
- Wherein use of the salt recovery solution is in a non-membrane and non-osmotic process.
- In another aspect, the present invention provides a process for concentrating a salt containing first aqueous solution, the process comprising the steps of:
-
- (a) adding the salt containing first aqueous solution to a salt recovery solution as defined above; and
- (b) allowing water from the salt containing aqueous solution to pass into the salt recovery solution.
- In one embodiment the precipitated salt forms part of an aqueous layer distinct from the salt recovery solution.
- In one embodiment the process is a non-membrane process.
- In one embodiment the process is a non-osmotic process.
- In one embodiment the process is a non-membrane and non-osmotic process.
- In one embodiment the process concentrates the first aqueous solution by at least 20%. In other embodiments the process concentrates the first aqueous solution by at least 30%, or by at least 40%, or by at least 50% or by at least 60%, or by at least 70% or by at least 80% or by at least 90%.
- In one embodiment the process is a minimal discharge process.
- In one embodiment the process is a zero-liquid discharge process.
- The foregoing brief summary broadly describes the features and technical advantages of certain embodiments of the present invention. Further technical advantages will be described in the detailed description of the invention and examples that follows.
- Novel features that are believed to be characteristic of the invention will be better understood from the detailed description of the invention when considered in connection with any accompanying figures and examples. However, the figures and examples provided herein are intended to help illustrate the invention or assist with developing an understanding of the invention, and are not intended to limit the invention's scope.
-
FIG. 1 : shows schematically a salt recovery process of the present invention. -
FIG. 2 shows gas chromatograms of the additive or regenerant solution overlaid with the additive or regenerant solution which has been used to remove water from a wet salt recovery solution. -
FIG. 3 shows a series of overlaid gas chromatograms of the salt recovery solution before brine addition, with absorbed water after brine addition and then after the salt recovery solution has been regenerated. -
FIG. 4 shows a calibration curve of NaCl solutions against osmotic pressure measurements. -
FIG. 5 shows a bar graph of the concentration of NaCl solution after concentration using varying ratios of salt recovery solution (TEA:MEK). -
FIG. 6 shows a graph of the osmotic pressure of NaCl solution after concentration using varying ratios of salt recovery solution (TEA:MEK). -
FIG. 7 shows a bar graph of the concentration of NaCl solution after concentration using varying ratios of N-Ethylpiperidine:Cyclohexanone salt recovery solution. -
FIG. 8 shows a graph of the osmotic pressure of NaCl solution after concentration using varying ratios of using salt recovery solution (N-Ethylpiperidine:Cyclohexanone). -
FIG. 9 shows a calibration curve of NH4SO4 solutions against osmotic pressure measurement. -
FIG. 10 shows a bar graph of the concentration of NH4SO4 solution after concentration using varying ratios of salt recovery solution (TEA:MEK). - The following description sets forth numerous exemplary configurations, parameters, and the like. It should be recognised, however, that such description is not intended as a limitation on the scope of the present invention, but is instead provided as a description of exemplary embodiments.
- In each instance herein, in descriptions, embodiments, and examples of the present invention, the terms “comprising”, “including”, etc., are to be read expansively, without limitation. Thus, unless the context clearly requires otherwise, throughout the description and the claims, the words “comprise”, “comprising”, and the like are to be construed in an inclusive sense as to opposed to an exclusive sense, that is to say in the sense of “including but not limited to”.
- The term “about” or “approximately” usually means within 20%, more preferably within 10%, and most preferably still within 5% of a given value or range. Alternatively, the term “about” means within a log (i.e., an order of magnitude) preferably within a factor of two of a given value.
- The term “additive” as used herein means an additive that releases water held within the salt recovery solution. A non-limiting example of a suitable additive that has been shown to release water bound within the salt recovery solution is citric acid or a citric acid solution. Throughout the specification, the word “regenerant” is also used interchangeably with the work “additive”. This is because the additive regenerates the salt recovery solution.
- The term “minimal discharge” as used herein, means a salt water or brine treatment process where minimal effluent or discharge is left over.
- The term “brine” or “brine solution” as used throughout the specification means an aqueous solution having a concentration of a salt in the water. The salt in the water could include sodium chloride, however, other salt solutions are envisaged, including ammonium sulfate or the like. The concentration of the salt in the aqueous solution may range from about 3.5% (typical concentration of seawater) through to much higher concentrations, such as 25%, which would include a salt water solution used for brining foods. Other brine waste water solutions from textile processing, the semiconductor industry or oil and gas industry would also be applicable for use with the current salt recovery solution and processes defined herein.
- As used herein, the term zero liquid discharge, as used throughout the specification, means a wastewater treatment process where no effluent, or discharge, is left over.
- As used herein, the term “C1-C7 alkyl” refers to a fully saturated branched or unbranched hydrocarbon moiety, which may be a straight or a branched chain of a particular range of 1-7 carbons. Preferably the alkyl comprises 1 to 7 carbon atoms, or 1 to 4 carbon atoms. Representative examples of C1-C7alkyl include, but are not limited to, methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl, 3-methylhexyl, 2,2-dimethylpentyl, 2,3-dimethylpentyl, n-heptyl, and the like. For example, the expression C1-C4-alkyl includes, but is not limited to, methyl, ethyl, propyl, butyl, isopropyl, tert-butyl and isobutyl. In one embodiment the C1-C7 alkyl group may be substituted with one or more of the following groups: -halo, —OH, —CN, —NO2, —C≡CH, —SH, alkyl, —(C1-C7 alkyl)-OH, —NH2, —NH(C1-C7 alkyl), —NH(C1-C7 alkyl)2, —O(C1-C7 alkyl), —C(O)—O(—C1-C7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C1-C7 alkyl).
- The term “C3-C7 monocyclic” as used herein is a 3-, 4-, 5-, 6-, or 7-membered saturated or unsaturated monocyclic ring. Representative C3-C7 monocyclic groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, phenyl, and cycloheptyl. In one embodiment, the C3-C7 monocyclic cycloalkyl group may be substituted with one or more of the following groups: -halo, —OH, —CN, —NO2, —C≡CH, —SH, alkyl, —(C1-C7 alkyl)-OH, —NH2, —NH(C1-C7 alkyl), —N(C1-C7 alkyl)2, —O(C1-C7 alkyl), —C(O)—O(—C1-C7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C1-C7 alkyl).
- The term “3- to 15-membered monocyclic ketone” refers to a 3- to 15-membered non-aromatic monocyclic ring system containing a ketone functional group. Representative examples of a 3- to 15-membered monocyclic ketone include, but are not limited to cyclopropanone, cyclobutanone, cyclopentanone, cyclohexanone, cycloheptanone, cyclooctanone, cyclononanone, cyclodecanone, cycloundecanone, cyclododecanone, cyclotridecanone; cyclotetradecanone and cyclopentadecanone.
- In one embodiment, the 3- to 15-membered monocyclic ketone may be substituted with one or more of the following groups -halo, —OH, —CN, —NO2, —C≡CH, —SH, —C1-C7 alkyl, —(C1-C7 alkyl)-OH, —NH2, —NH(C1-C7 alkyl), —NH(C1-C7 alkyl)2, —O(C1-C7 alkyl), —C(O)—O(—C1-C7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C1-C7 alkyl).
- The term “8-15 membered aromatic system optionally including one or more heteroatoms selected from S, N or O” of Formula I includes naphthaleneone, fluorenone, quinolinone and indanone ring systems, optionally substituted with halo, hydroxy, nitro, —SH, —C1-C7 alkyl and —O(C1-C7 alkyl). Representative examples of such compounds of Formula I include, without limitation napthalenone, 2,2,4-trichloro-1(2h)-naphthalenone, 4-methyl-1h-naphthalen-2-one, 9-fluorenone, 9-fluorenone oxime, 2-nitro-9-fluorenone, 3-nitro-9-fluorenone, 4-nitro-9-fluorenone, 2,6-dinitro-9-fluorenone, 2,7-dinitro-9-fluorenone, 2,3,7-trinitro-9-fluorenone, 2-fluoro-9-fluorenone, 1-bromo-9-fluorenone, 2-bromo-9-fluorenone, 2,7-dichloro-9-fluorenone, 2,7-dibromo-9-fluorenone, 2-hydroxy-9-fluorenone, 4-hydroxy-9-fluorenone, 1-methylfluoren-9-one, 4-methylfluoren-9-one 11-benzo(a)fluorenone, benzo(b)fluorenone, 1h-benzo(a)fluoren-1-one, 3,4-dihydro-2(1h)-quinolinone, 7-hydroxy-3,4-dihydro-2(1h)-quinolinone, 6-hydroxy-3,4-dihydro-2(1h)-quinolinone, 3-butyl-4-hydroxy-1-methyl-2(1h)-quinolinone, 8-bromo-2,3-dihydro-4(1h)-quinolinone, 6-fluoro-4,4-dimethyl-3,4-dihydro-2(1h)-quinolinone, 8-fluoro-4,4-dimethyl-3,4-dihydro-2(1h)-quinolinone, 2,6-dimethyl-4(1h)-quinolinone and 3-butyl-4-hydroxy-1-methyl-2(1h)-quinolinone 1-indanone,5,6-dimethoxy-1-indanone, 6-bromo-1-indanone, 6-methoxy-1-indanone, 2-bromo-1-indanone, 4-bromo-1-indanone, 5-bromo-1-indanone, 5-chloro-1-indanone, 6-chloro-1-indanone, 4,7-dimethyl-1-indanone, 2-methyl-1-indanone, 4-methyl-1-indanone, 5-fluoro-1-indanone, 6-fluoro-1-indanone, 6-(trifluoromethyl)-1-indanone, 4-methoxy-1-indanone, 3,5-dimethoxy-1-indanone, 4,7-dimethoxy-1-indanone, 5-hydroxy-1-indanone, 4-hydroxy-1-indanone, 7-hydroxy-1-indanone, 2-indanone oxime and 2,2-di(methylthio)-1-indanone.
- The term “5-8 membered aromatic monocyclic optionally containing a heteroatom” includes without limitation 5-membered rings such as pyrrolyl, furanyl, thiophenyl; 6-membered rings such as phenyl, pyridinyl, pyranyl, thiopyranyl and 7-membered rings such as azepinyl, oxepinyl, thiepinyl.
- The term “3- to 15-membered monocyclic heterocyclic ketone” refers to: (i) a 3- or 4-membered non-aromatic monocyclic cycloalkyl in which 1 of the ring carbon atoms has been replaced with an N, O or S atom; or (ii) a 5- to 15-membered non-aromatic monocyclic cycloalkyl in which 1-4 of the ring carbon atoms have been independently replaced with a N, O or S atom. Representative examples of a 3- to 15-membered monocyclic heterocyclic ketone having one N, O or S atom include, but are not limited to oxiran-2-one, thiiran-2-one, oxetan-2-one, oxetan-3-one, azetidin-3-one, thietan-2-one, thietan-3-one, dihydrofuran-2(3H)-one, dihydrofuran-3(2H)-one, pyrrolidin-3-one, dihydrothiophen-3(2H)-one, dihydrothiophen-2(3H)-one, tetrahydro-2H-pyran-2-one, dihydro-2H-pyran-3(4H)-one, dihydro-2H-pyran-4(3H)-one, piperidin-3-one, piperidin-4-one, tetrahydro-2H-thiopyran-2-one, dihydro-2H-thiopyran-3(4H)-one, dihydro-2H-thiopyran-4(3H)-one, oxepan-2-one, oxepan-3-one, oxepan-4-one, thiepan-2-one, thiepan-3-one, thiepan-4-one, azepan-3-one, azepan-4-one, oxocan-2-one, oxocan-3-one, oxocan-4-one, oxocan-5-one, thiocan-2-one, thiocan-3-one, thiocan-4-one, thiocan-5-one, azocan-3-one, azocan-3-one, azocan-4-one, azocan-5-one, azonan-3-one, azonan-4-one, azonan-5-one, oxonan-2-one, oxonan-3-one, oxonan-4-one, oxonan-5-one, thionan-2-one, thionan-3-one, thionan-4-one, thionan-5-one, oxacycloundecan-2-one, oxacycloundecan-3-one, oxacycloundecan-4-one, oxacycloundecan-5-one, oxacycloundecan-6-one, azacycloundecan-3-one, azacycloundecan-4-one, azacycloundecan-5-one, azacycloundecan-6-one, thiacycloundecan-2-one, thiacycloundecan-3-one, thiacycloundecan-4-one, thiacycloundecan-5-one, thiacycloundecan-6-one, oxacyclododecan-2-one, oxacyclododecan-3-one, oxacyclododecan-4-one, oxacyclododecan-5-one, oxacyclododecan-6-one, oxacyclododecan-7-one, azacyclododecan-3-one, azacyclododecan-4-one, azacyclododecan-5-one, azacyclododecan-6-one, azacyclododecan-7-one, thiacyclododecan-2-one, thiacyclododecan-3-one, thiacyclododecan-4-one, thiacyclododecan-5-one, thiacyclododecan-6-one, thiacyclododecan-7-one, oxacyclotridecan-2-one, oxacyclotridecan-3-one, oxacyclotridecan-4-one, oxacyclotridecan-5-one, oxacyclotridecan-6-one, oxacyclotridecan-7-one, azacyclotridecan-3-one, azacyclotridecan-4-one, azacyclotridecan-5-one, azacyclotridecan-6-one, azacyclotridecan-7-one, thiacyclotridecan-2-one, thiacyclotridecan-3-one, thiacyclotridecan-4-one, thiacyclotridecan-5-one, thiacyclotridecan-6-one, thiacyclotridecan-7-one, oxacyclotetradecan-2-one, oxacyclotetradecan-3-one, oxacyclotetradecan-4-one, oxacyclotetradecan-5-one, oxacyclotetradecan-6-one, oxacyclotetradecan-7-one, oxacyclotetradecan-8-one, azacyclotetradecan-3-one, azacyclotetradecan-4-one, azacyclotetradecan-5-one, azacyclotetradecan-6-one, azacyclotetradecan-7-one, azacyclotetradecan-8-one, thiacyclotetradecan-2-one, thiacyclotetradecan-3-one, thiacyclotetradecan-4-one, thiacyclotetradecan-5-one, thiacyclotetradecan-6-one, thiacyclotetradecan-7-one, thiacyclotetradecan-8-one, oxacyclopentadecan-2-one, oxacyclopentadecan-3-one, oxacyclopentadecan-4-one, oxacyclopentadecan-5-one, oxacyclopentadecan-6-one, oxacyclopentadecan-7-one, oxacyclopentadecan-8-one, azacyclopentadecan-3-one, azacyclopentadecan-4-one, azacyclopentadecan-5-one, azacyclopentadecan-6-one, azacyclopentadecan-7-one, azacyclopentadecan-8-one, thiacyclopentadecan-2-one, thiacyclopentadecan-3-one, thiacyclopentadecan-4-one, thiacyclopentadecan-5-one, thiacyclopentadecan-6-one, thiacyclopentadecan-7-one, thiacyclopentadecan-8-one. In one embodiment, the 3- to 15-membered monocyclic heterocyclic ketone group may be substituted with one or more of the following groups -halo, —OH, —CN, —NO2, —C≡CH, —SH, —C1-C6 lower alkyl, —(C1-C7 alkyl)-OH, —NH2, —NH(C1-C7 alkyl), —N(C1-C7 alkyl)2, —O(C1-C7 alkyl), —C(O)—O(—C1-C7 alkyl), —C(O)OH; —C(O)—H, or —C(O)—(C1-C7 alkyl). For the avoidance of doubt, the 3-5 membered monocyclic heterocyclic ketone does not include any amide groups where the ketone enolisable carbonyl group is adjacent a N atom in the cyclic structure.
- The term “halo” as used herein refers to —F, —Cl, —Br or —I.
- The term “an enolisable carbonyl” means a compound that has one or more carbonyl functional groups and wherein at least one of the carbonyl functional groups has alpha hydrogens (Hα) that may be removed by a base to form an enolate and then an enol as shown in the reaction scheme below.
- It is to be understood that the term enolisable carbonyl as used in the specification does not include a compound having solely an aldehyde functional group, a compound having solely a carboxylic acid functional group, a compound having solely an amide functional group, a compound having solely an acyl halide functional group or acetylacetone. The enolisable carbonyls of the invention include without limitation those exemplified in the specification and defined by Formulae I and II, and without limitation also include the following enolisable carbonyls: 1-acetonapthone, 2-acetonaphthone, 4-methyl-1-acetonaphthone, 1′-hydroxy-2′-acetonaphthone, 2′-hydroxy-1′-acetonaphthone, 2-methoxy-1-acetonaphthone, 4-fluoro-1-acetonapthone; 2-acetylphenanthrene, 3-acetylphenanthrene, 4-acetylphenanthrene, 9-acetylphenanthrene, 6-bromo-9-acetylphenanthrene, 9-fluoro-10-acetylphenanthrene, 9-fluorenone, 9-fluorenone oxime, 2-nitro-9-fluorenone, 3-nitro-9-fluorenone, 4-nitro-9-fluorenone, 2,6-dinitro-9-fluorenone, 2,7-dinitro-9-fluorenone, 2,3,7-trinitro-9-fluorenone, 2-fluoro-9-fluorenone, 1-bromo-9-fluorenone, 2-bromo-9-fluorenone, 2,7-dichloro-9-fluorenone, 2,7-dibromo-9-fluorenone, 2-hydroxy-9-fluorenone, 4-hydroxy-9-fluorenone; 1-methylfluoren-9-one; 4-methylfluoren-9-one; 3,4-dihydro-2(1H)-quinolinone, 7-hydroxy-3,4-dihydro-2(1H)-quinolinone, 6-hydroxy-3,4-dihydro-2(1H)-quinolinone, 8-bromo-2,3-dihydro-4(1H)-quinolinone, 3-butyl-4-hydroxy-1-methyl-2(1H)-quinolinone, 6-fluoro-4,4-dimethyl-3,4-dihydro-2(1H)-quinolinone, 8-fluoro-4,4-dimethyl-3,4-dihydro-2(1H)-quinolinone, 2,6-dimethyl-4(1H)-quinolinone, 3-butyl-4-hydroxy-1-methyl-2(1H)-quinolinone, 1-indanone, 5,6-dimethoxy-1-indanone, 6-bromo-1-indanone, 6-methoxy-1-indanone, 2-bromo-1-indanone, 4-bromo-1-indanone, 5-bromo-1-indanone, 5-chloro-1-indanone, 6-chloro-1-indanone, 4,7-dimethyl-1-indanone, 2-methyl-1-indanone, 4-methyl-1-indanone, 5-fluoro-1-indanone, 6-fluoro-1-indanone, 6-(trifluoromethyl)-1-indanone, 4-methoxy-1-indanone, 3,5-dimethoxy-1-indanone, 4,7-dimethoxy-1-indanone, 5-hydroxy-1-indanone, 4-hydroxy-1-indanone, 7-hydroxy-1-indanone, 2-indanone oxime, 2,2-di(methylthio)-1-indanone, (2,4-dimethoxyphenyl)acetone, 3,5-dimethoxyacetophenone, 4-(4-methoxyphenyl)-2-butanone, 3-methoxyphenylacetone, 4-methoxy acetophenone, 4-methoxy-2-phenylacetophenone, 2,5-dimethylphenylacetone, 3,4,5-trimethoxyphenylacetone, 4-hydroxy-3-phenylbutan-2-one, 3-hydroxy-4-phenylbutan-2-one, 3-hydroxy-3-phenylbutan-2-one, 4-hydroxy-4-phenylbutan-2-one, 1-hydroxy-3-phenylbutan-2-one, 3-hydroxy-1-phenylbutan-2-one, 3-hydroxy-1,3-diphenylbutan-2-one, 4-hydroxyphenylacetone, 3,4-dihydroxyphenylacetone, 4-nitrophenylacetone, acetophenone, 4-methyl acetophenone, benzylacetone, 3-methylphenylacetone, 4-methylphenylacetone, 4-ethylphenylacetone, 1-phenylbutan-2-one, 3-phenylbutan-2-one, 4-phenylbutan-2-one, 1-bromo-4-phenylbutan-2-one, 3-methyl-1-phenylbutan-2-one, 3-methly-4-phenylbutan-2-one, ethyl phenyl ketone, butyl phenyl ketone, cyclopropyl phenyl ketone, cyclopentyl phenyl ketone, cyclobutyl phenyl ketone, cyclohexyl phenyl ketone, 2-phenylcyclopentanone, 3-phenylcyclopentanone, 2-phenylcyclohexanone, 3-phenylcyclohexanone, 2-phenylcycloheptanone,3-phenylcycloheptanone, 4-chlorophenyl acetone, 4-chloro-2-phenylacetophenone, 2,6-dichlorophenylacetone, 3-chlorophenylacetone, 2,6-difluorophenylacetone, 1-bromo-1-phenylbutan-2-one, 3-bromo-4-phenylbutan-2-one, 1-bromo-4-phenylbutan-2-one, 3-chloro-4-phenylbutan-2-one, 2-acetylthiophene, cyclopropyl-2-thienyl ketone, 2-acetylfuran, 2-furyl methyl ketone, 1-acetylpyrrole, 2-acetylpyrrole, 4-methyl-2-phenylacetophenone, 1,3-diphenylacetone, 4,4-diphenylbutan-2-one, benzophenone, 4-napthyl phenyl ketone, 2-benzoylpyridine, 3-benzoylpyridine, 4-benzoylpyridine, 2-(4-chlorobenzoyl) pyridine, 2-benzoylthiophene, 2-benzoylpyrrole, di(3-thiophenyl) methanone, 3-phenyl-1-(2-thienyl)-2-propen-1-one, and piperonyl acetone.
- The term “tertiary amine containing compound” preferably means a compound having at least one tertiary amine group, but it is to be appreciated that the compound may have more than one tertiary amine group or further may be a mixture of tertiary amine containing compounds. Preferably the tertiary amine containing compound is a base, such as a Lewis base. If the base is a Lewis base, it is envisaged that a Lewis adduct may be formed with the enolisable carbonyl. In one embodiment it is preferred that the tertiary amine containing compound is immiscible with water at or above 20 degrees Celsius under one standard atmosphere of pressure. The solution may include a combination of more than one tertiary amine containing compound. The tertiary amine containing compound may be aliphatic, conjugated, asymmetric or cyclic or a combination thereof.
- Examples of suitable tertiary amine containing compounds include the following:
- In one embodiment the tertiary amine containing compound is selected from 1-ethyl pyrrolidine, 2-methylpyridine, N-methylpiperidine and 4-ethylmorpholine.
- In one embodiment the tertiary amine containing compound is selected from a —N(C1-C7 alkyl)3. In another embodiment the tertiary amine containing compound is selected from a —N(C1-C4 alkyl)3. In yet a further embodiment the tertiary amine containing compound is —N(C2 alkyl)3 (triethylamine).
- It will be appreciated that the above listed tertiary amine containing compounds are simple enough for production on an industrial scale.
- The present invention is directed to a salt recovery solution suitable for recovering a salt from a first aqueous solution, the salt recovery solution comprising:
-
- a) at least one tertiary amine containing compound; and
- b) at least one enolisable carbonyl.
- In one embodiment the salt recovery solution comprises:
-
- a) at least one tertiary amine containing compound; and
- b) at least one enolisable carbonyl of Formula I,
- wherein
-
- c) R1 and R2 are independently selected from a —C1-C7 alkyl or a —C3-C7 monocyclic; or
- d) one of R1 or R2 is selected from a —O—(C1-C7 alkyl) and the other is selected from a —C1-C7 alkyl, or
- e) R1 and R2 together, with the carbonyl of Formula I, form a 3-15 membered monocyclic ketone or a 3-15 membered monocyclic heterocyclic ketone.
- In another embodiment the salt recovery solution comprises at least one enolisable carbonyl of Formula II,
-
- wherein X is a 5-8 membered aromatic monocyclic optionally containing a heteroatom selected from S, N or O;
- each R3, if present, is selected independently from a halo, a hydroxy, a —NO2, a C1-C7 alkyl, or a —O—(C1-C7 alkyl);
- R4 is selected from a halo, a —O—(C1-C7 alkyl); a C1-C7 alkyl optionally substituted with a halo, a hydroxy, a —O—(C1-C7 alkyl) or a C3-C7 monocyclic; or a C3-C7 monocyclic optionally substituted with a halo, a hydroxy, a C1-C7 alkyl, or a —O—(C1-C7 alkyl);
- m is 0, 1, 2 or 3 carbons, each carbon being optionally substituted with one or more substituents selected from a halo, hydroxy, a C1-C7 alkyl optionally substituted with a halo or a hydroxy; a C1-C7 alkyl, a —O—(C1-C7 alkyl) or a C3-C7 monocyclic; and
- n is 0, 1, 2 or 3.
- In another aspect, the present invention provides a process for recovering a salt from a first aqueous solution, the process including the step of:
-
- (a) adding the salt containing first aqueous solution to a salt recovery solution; and
- (b) allowing the salt to precipitate on passage through the salt recovery solution.
- Wherein the process is a non-membrane process.
- Wherein the process is a non-osmotic process.
- Wherein use of the salt recovery solution is in a non-membrane and non-osmotic process.
- In one embodiment the process is a minimal liquid discharge process.
- In one embodiment the process is a zero liquid discharge process.
- In a further embodiment the process includes the further step of adding an additive to the salt recovery solution to further release water held within the salt recovery solution. In one embodiment the additive is citric acid. In one embodiment the citric acid is a concentrated solution of citric acid comprising between about 200-450 gms of citric acid per litre of water. In another embodiment the citric acid is anhydrous citric acid added directly to the salt recovery solution.
- In a further embodiment the salt recovery solution is recovered. In a further embodiment the salt recovery solution is recycled for use in a further separation process. In a further embodiment the process is a continuous process.
- It is to be appreciated that the molar ratio of the tertiary amine containing compound to the enolisable carbonyl may vary widely and may be from about 1:99 or 99:1; or from about 1:50 or 50:1 or from about 1:10 or 10:1 or from about 1:5 or 5:1 or from about 1:3 or from about 3:1 or from about 1:2 or from about 2:1. In a preferred embodiment the molar ratio is about 1:1. A chemistry technician would be able to routinely determine the most suitable molar ratio depending on the purpose for which the salt recovery solution is to be employed.
- In a further embodiment the first aqueous solution is salt water or a brine solution.
- The examples described herein are provided for the purpose of illustrating specific embodiments of the invention and are not intended to limit the invention in any way. Persons of ordinary skill can utilise the disclosures and teachings herein to produce other embodiments and variations without undue experimentation. All such embodiments and variations are considered to be part of this invention.
- In a first example and with reference to
FIG. 1 , a salt recovery solution was prepared using triethylamine (TEA) and methyl ethyl ketone (MEK) in the molar ratio of 0.5:1. A saturated sodium chloride salt solution was introduced to the salt recovery solution—seestep 1 ofFIG. 1 . After adding a few drops of saturated salt solution, it was observed that salt crystals separated out from water and settled at the bottom instantly—seestep 2 ofFIG. 1 . The water formed the aqueous layer with salt precipitate at the bottom of the vial. The salt recovery solution along with the aqueous layer was decanted and the salt was then dried. This demonstrated the recovery of salt from a saturated salt solution using a salt recovery solution and with no liquid discharge. It was also recognised that despite there being a separate layer of water formed at the bottom of the vial, there was a small quantity of water that had been absorbed by the salt recovery solution. Accordingly, a weak acid, citric acid, was used as an additive to help desorb the water from the salt recovery solution. This step (seeStep 3 inFIG. 1 ) was required to re-establish a system that would desorb all the water from the salt recovery solution and reinstate the capability of the salt recovery solution to separate the salt from water for use in a further separation step or in a continuous separation process. It was observed that upon adding as few as 5 drops of saturated citric acid, a thin layer of water became apparent at the bottom of the vial. Furthermore, it has been established that the citric acid got washed through to the aqueous phase rather than being retained in the salt recovery solution phase. Thus, the water was extracted from the salt recovery solution by using a citric acid solution. The last step (not shown inFIG. 1 ) was to check if the ability of the recycled salt recovery solution to salt-out was restored. The salt recovery solution was removed from the salt solution and then re-tested to see if the salt recovery solution could be re-used to cause additional saturated salt solution that was dropped into the salt recovery solution to precipitate out salt. It was found that the recycled salt recovery solution could effectively separate out salt again from a second saturated salt solution introduced to the salt recovery solution. The salt again precipitated to the bottom of the test vial after passage through the salt recovery solution. A similar aqueous/precipitate layer formed under the non-aqueous salt recovery solution layer in the vial. - A salt recovery solution was prepared using triethylamine (TEA) and methyl ethyl ketone (MEK) in the molar ratio of 0.5:1. Two brine solutions were prepared. One brine solution comprised sodium chloride at a concentration of 50 grams per litre (50,000 ppm NaCl) and the other brine solution comprised sodium chloride at a concentration of 100 grams per litre (100,000 ppm NaCl). The brine solutions were separately introduced to 10 mL of the salt recovery solution. The salts were observed to precipitate immediately. The addition of the brine solution to the salt recovery solution was stopped as soon as a thin aqueous phase was observed at the bottom of the vial. This indicated that the maximum absorption capacity of the salt recovery solution had been reached. The volume of brine solution added once the aqueous layer started to develop was recorded.
- The results for the 50 000 ppm NaCl brine solution are shown in Table 1.
-
TABLE 1 50 000 ppm NaCl solution Samples-Maximum Absorption Capacity Experiments Volume of salt Volume of brine Volume recovery solution added for of excess solution complete absorption brine (approx) (V1) in mL in mL in mL 10 0.25 0.0000 10 0.30 0.0000 10 0.35 0.0000 10 0.40 0.0050 - From Table 1, the ratio between the salt recovery solution and the 50 000 ppm NaCl brine solution for complete water absorption was approximately 29:1.
- The results for the 100 000 ppm NaCl brine solution are shown in Table 2.
-
TABLE 2 100 000 ppm NaCl solution Samples-Maximum Absorption Capacity Experiments Volume of dry Volume of brine Volume of excess salt recovery added for complete brine (approx.) solution (V1) in mL absorption in mL in mL 10 0.25 0.0000 10 0.35 0.0000 10 0.45 0.0000 10 0.50 0.0010 - From Table 2, the ratio between the salt recovery solution and the 100 000 ppm NaCl solution for complete water absorption was approximately 22:1.
- In the case of a larger scale process, it would be recommended that the process operate at higher ratios of salt recovery solution to brine solution to ensure a total absorption of water and avoid brine carry-over.
- The input salt concentrations were measured, and the dry weights of samples recorded containing 0.2 mL, 0.5 mL and 1 mL of both 50 000 ppm NaCl solution and 100 000 ppm NaCl solution respectively. The input salt results are shown in (Table 3) and the percent salt recovery results are shown in Table 4.
- The output salt concentrations were also determined as follows.
-
- 0.20 mL and 0.40 mL of brine were added to 10 mL of the salt recovery solution.
- The water from the brine was absorbed by the salt recovery solution and the salts precipitated.
- The precipitated salts were allowed to settle to the bottom of the glass vial overnight.
- Diluted salt recovery solution (which absorbed the water from the brine) was filtered into a clean vial
- The dry weight of the salt (output) was measured and recorded once completely dried in the block heater operating at 150° C. (Table 3).
- Salt recovery % was determined by the formula:
-
- Results
-
TABLE 3 Dry weights of initial salt concentration Brine Volume of brine Dry weight of salt solution solution in mL input (avg) in mg 50 000 ppm 0.20 10.00 NaCl solution 0.50 25.00 1.00 50.00 100 000 ppm 0.20 20.00 NaCl solution 0.50 50.00 1.00 100.00 -
TABLE 4 Salt recovery % of the salt recovery material. Volume Volume Dry weight Salt recovery % = of salt of brine of salt Dry weight of recovery solution precipitated* salt output/Dry solution added (output) weight of salt Brine solution (mL) (mL) avg in mg input × 100 50 000 ppm NaCl 10.0000 0.2000 0.0112 112.0000 solution 50 000 ppm NaCl 15.0000 0.4000 0.0209 104.5000 solution 50 000 ppm NaCl 10.0000 0.2000 0.0107 107.0000 solution 100 000 ppm NaCl 10.0000 0.2000 0.0243 121.5000 solution 100 000 ppm NaCl 10.0000 0.2000 0.0206 103.0000 solution 100 000 ppm NaCl 10.0000 0.4000 0.0507 126.7500 solution *The dry weights are the average values of each set of two trials. - Salt recovery: From the above results, it can be seen that it is possible to achieve around 100% salt and water recovery from the brine solution when the volume of brine added is just below the maximum absorption capacity of the salt recovery solution. The deviation in salt recoveries can be attributed to the unavoidable error to the small scale of the experiments.
- At the maximum absorption capacity of the salt recovery solution: The decrease in salt recovery % maybe due to sub-optimal crystallization and loss of mass during the filtering process. With the excess brine developing, the calculations become harder due to small volumes and experimental errors.
- Salt Rejection Percentage
- The salt rejection percentage was estimated to show that only water was being absorbed by the salt recovery solution from the brine solution and that the salt recovery solution was rejecting substantially all of the salt from the brine. The conductivity of pure water, the neat brine solution and the concentrated salt recovery solution were measured. For 10 mL of the concentrated salt recovery solution, different volumes of brine solution were added and the conductivity of the diluted salt recovery solution (concentrated salt recovery solution+water absorbed) was measured (Table 5).
- The conductivity of the brine solution and dry concentrated salt recovery solution were compared.
- The salt rejection % was calculated by the following formula:
-
-
- Conductivity of pure water in μS/cm=0.0
- Conductivity of concentrated salt recovery solution in μS/cm=0.4
- Conductivity of concentrated salt recovery solution with distilled water μS/cm=37.0
-
TABLE 5 Conductivity measurements of the salt recovery solution along with absorbed water and salt rejection % calculations Conductivity of Conductivity of salt recovery Salt 50 000 ppm NaCl material with absorbed water rejection solution in μS/cm μS/cm % 78,600 9.1 99.99 8.2 99.99 6.1 99.99 Conductivity of Conductivity of salt recovery Salt 100 000 ppm NaCl material with absorbed water rejection solution in μS/cm μS/cm % 148,600 10.8 99.99 11.6 99.99 9.6 99.99 - The salt rejection % of over 99 is indicative of the fact that there is complete precipitation of salt from the brine solution and there are no residual salts going into the salt recovery solution.
- We can conclude that the water absorbed from the brine solution is free of salts because the conductivity measurements of the salt recovery solution with water absorbed from the brine is equivalent to that of salt recovery solution with distilled water.
- From the above examples we can conclude that we are able to absorb all the water from both 50 000 ppm NaCl solution and 100 000 ppm NaCl solution solutions when the volumetric ratio of the slat recovery solution to brine is above 25. For large scale process it is recommended to run at higher ratios to avoid formation of saturated brine solution and to obtain a 100% salt recovery by absorbing all the water.
- From the results of Example 2, we can see that the recovery percentages are close to 100% when the volume of brine added is below the maximum capacity of the salt recovery solution. Factors such as formation of hydrates, experimental and instrumental errors are to be taken into consideration.
- Based on the conductivity results, an average salt rejection percentage of 99.99 is observed in both the brine solutions which means that we are completely precipitating out the salts from the brine solution.
- The salt recovery solution can achieve close to 100% salt and water recovery by minimizing the volume of feed solution in a thermo-efficient way unlike the traditional treatment systems. The lab scale results are indicating that there are no salts going into the salt recovery solution during the absorption process and that the purity of absorbed water is relatively high.
- Brine solutions from industrial operations (
Brine Samples -
TABLE 6 Brine Sample Compositions Brine Brine Brine Sample 1 Sample 2Sample 3Concentration Concentration Concentration Analyte (mg/L) (mg/L) (mg/L) Alkalinity, Bicarbonate 293 100 376 as CaCO3 Chloride 1950 126000 34600 Sulfate 5950 910 ND Barium 0.012 6.21 240 Calcium 501 14500 1790 Magnesium 359 2110 180 Manganese 0.011 8 4 Potassium 3.62 1010 142 Sodium 3100 47500 19000 Strontium 6.93 1270 209 Boron 30.7 ND 25 Iron ND ND 181 Total Dissolved Solids 12300 215000 55400 - Brine having the compositions of
Sample 1,Sample 2 andSample 3 was each separately and slowly added to 10 mL of the salt recovery solution as defined above, specifically using triethylamine (TEA) and methyl ethyl ketone (MEK) in the molar ratio of 1:2. The water from the brine was absorbed by the salt recovery solution and the salts began to precipitate. The addition of each brine solution continued until bubbles of a separate phase could be observed at the bottom of the vial after shaking. These bubbles of separate phase comprise water, which is no longer being absorbed by the salt recovery solution, and which indicates that the maximum water absorption capacity of the salt recovery solution had been reached. The volume at which the aqueous layer started to form for each sample (repeated 5 times) was recorded and the results are shown in Table 7. -
TABLE 7 Volume Volume Volume Volume of brine of brine of brine of dry salt Sample 1 Sample 2Sample 3recovery added added for added for solution for maximum maximum maximum Sample (V1) absorption absorption absorption number in mL in mL in mL in mL 1.1 10 0.75 0.20 0.35 1.2 10 0.78 0.15 0.38 1.3 10 0.8 0.12 0.36 1.4 10 0.85 0.13 0.41 1.5 10 0.85 0.13 0.42 Average — 0.81 0.15 0.38 Std Dev — 0.04 0.03 0.03 - From Table 7, it can be seen that quite a large variation between the three brine samples exists in terms of absorption capacity.
Brine Sample 1 has the highest capacity at 0.81 mL (+/−0.04) andBrine Sample 2 having the lowest at 0.15 mL (+/−0.03). This reflects the differing salt compositions and concentrations of each of the commercial brine samples and demonstrates the importance of knowing what the composition of the brine is before salt recovery treatment. Once the average maximum absorption capacity for 10 mL of salt recovery solution was met, the salt recovery solution would be diluted and would be unable to absorb water and precipitate salts effectively. It is to be appreciated that for large scale processes, it would be desirable to utilize higher ratios of salt recovery solution relative to the brine solution to ensure a total absorption of water and avoid brine carry-over. - Salt and Water Recovery Capacity of Salt Recovery Solution
- To ensure full water absorption when calculating the salt and water recovery capacity, between 80-85% of the maximum brine absorption capacity was used in the following experiments. This ensured that the quantity of brine sample which was added to the salt recovery solution (triethylamine (TEA) and methyl ethyl ketone (MEK) in the molar ratio of 1:2) was sufficiently under the maximum absorption concentration to ensure full adsorption of the water with no brine carry-over.
- The quantity of brine added to 20 mL of salt recovery solution, in a pre-weighed vial, was adjusted to be slightly below maximum water absorption as follows:
-
- Brine Sample 1: 1.4 mL
- Brine Sample 2: 0.24 mL
- Brine Sample 3: 0.65 mL
- The brine and the salt recovery solution were mixed and resulted in the salt precipitating out. The Samples were left for 20 mins to ensure full separation of the solid salts.
- The now wet salt recovery solution was decanted into a clean vial using a 5 mL syringe to carefully remove the wet salt recovery solution from the precipitated salts without transferring any salts into the fresh vial (approx. 17 mL, measuring the actual quantity in each individual case).
- The salt slurry left behind was heated at 110° C. for 1 hour to remove the left-over salt recovery material. The dried salt was cooled and then weighed. The weight of the clean vial was deducted to obtain the weight of salt recovered. The results are tabulated in Table 8.
-
TABLE 8 Raw data from the salt recovery experiments. Vial weight Salt Brine Vial tare Brine after collected Salt % salt Sample weight (g) (mL) drying (g) (g) expected (g) recovered Brine Sample 115.1620 1.4 15.2188 0.0568 0.05025 113 15.0258 1.4 15.0859 0.0601 0.05025 120 15.0369 1.4 15.0957 0.0588 0.05025 117 Brine Sample 215.1319 0.24 15.1886 0.0567 0.0578 98 15.0513 0.24 15.1101 0.0588 0.0578 102 14.9817 0.24 15.0396 0.0579 0.0578 100 Brine Sample 315.146 0.65 15.1929 0.0469 0.0475 99 13.5146 0.65 13.572 0.0574 0.0475 121 13.2655 0.65 13.3221 0.0566 0.0475 119 - The salt recovery percentage was determined by the following formula.
-
- Table 8 above details the raw data collected to calculate the percentage of salt recovered. The “salt expected” was calculated by evaporating the same amount of raw brine as was added to each sample, in a heat block at 110° C. This measured how much salt was in each brine sample and allowed for the calculation of what percentage of this initial brine was collected (see Table 9).
-
TABLE 9 Calculating the dry weight of brine for the ‘salt expected’ column. Brine Dry Weight Average Brine Sample 1 0.0482 0.0505 0.0523 (+/−0.0023) 0.0483 0.0534 Brine Sample 20.0576 0.0559 0.0581 (+/−0.0019) 0.054 0.0539 Brine Sample 30.043 0.0446 0.052 (+/−0.0043) 0.0414 0.0419 - The decanted and wet salt recovery material was regenerated using a regenerating agent/regenerant (0.3-0.5 mL) using the following method. The regenerating agent, which was a concentrated solution of citric acid (412 g/L) or anhydrous citric acid was added to the diluted/wet salt recovery solution from the previous step to separate the water absorbed by the salt recovery solution. It has been found, and gas chromatographic data has shown that some of the salt recovery solution is dissolved into the regenerant. With reference to
FIG. 2 , it can be seen that the gas chromatogram of fresh regenerant shows a small peak at 2.8 mins and a larger slightly trailing peak at 3.0 mins. This larger peak is water which is used as the solvent for the regenerant. It is considered that the tail is caused by the regenerant material bonding with water molecules. The trailing peak at 4.1 mins is highly characteristic of the salt recovery solutions and clearly indicates that some of the salt recovery solution has leached into the regenerant. Because of the trailing shape of this peak and the overlap of the first peaks, it is not possible to accurately quantify the amount of salt recovery solution which has crossed over into the regenerant, however, semi-quantitative analysis of multiple samples has suggested that up to about 15% of the salt recovery solution has crossed over into the regenerant. To minimise this cross over, the amount of regenerating agent used is selected to be as little as possible. In order to determine the amount of water recovered from the process a known volume of regenerating agent was added to the diluted salt recovery solution. The regenerating agent removed the water from the diluted salt recovery solution to leave a concentrated salt recovery solution. Because of the difference in density between the regenerating agent and the concentrated salt recovery solution, the concentrated salt recovery solution (now non-aqueous or dry) rises to the top while the now diluted regenerating agent sinks to the bottom of the vial. The bottom aqueous layer was carefully removed to a measuring cylinder and the volume recovered was recorded in Table 10. The volume of the aqueous layer was measured using the following formula. -
Volume of water recovered (mL)=Volume of aqueous phase (mL)−Volume of regenerant (mL) - The water recovery % was calculated by the formula (Table 10):
-
-
TABLE 10 Water recovery % of the salt recovery solution (All values are averaged values from two trials.) Gravimetric data and analysis Salt Recovery Water Salt Solution Brine Regenerant collected % water collected % salt Brine (mL) (mL) (mL) (mL) recovered (g) recovered Brine 20 1.40 0.35 1.05 95 0.0568 113 Sample 20 1.40 0.35 1.01 91 0.0601 120 1 20 1.40 0.35 1.01 91 0.0588 117 Brine 20 0.24 0.35 0.20 107 0.0567 98 Sample 20 0.24 0.50 0.18 95 0.0588 102 2 20 0.24 0.35 0.18 96 0.0579 100 Brine 20 0.65 0.50 0.56 102 0.0469 99 Sample 20 0.65 0.30 0.53 104 0.0574 121 3 20 0.65 0.30 0.53 104 0.0566 119 - Table 11, below, shows the average water and salt recoveries across three samples for each of the brines tested. The majority of the salt recoveries are over 100%. This perhaps indicates some interaction between the salts in the brine and the salt recovery solution which results in the salts binding with the salt recovery solution, resulting in an increase in weight compared to when water is removed by evaporation alone.
-
TABLE 11 Percentage water and salt recovery (average of 3 samples) % water % salt Brine recovered recovered Brine Sample 93 117 1 Brine Sample 99 100 2 Brine Sample 103 113 3 - From the above results, it can be seen that it is possible to achieve 100% salt and water recovery from the brine solution when the volume of brine added is just below the maximum absorption capacity of the salt recovery solution. It is to be appreciated that the deviation seen in the salt recoveries can be attributed to the hydration of salts and the unavoidable error due to the small scale of these experiments.
- To support the gravimetric data presented above, an example of gas chromatogram traces is shown in
FIG. 3 .FIG. 3 shows an example gas chromatograph ofBrine Sample 1 focusing on the water peak. The black (solid line) trace is the salt recovery solution before the addition of the brine sample. The second trace (long dash line) shows the water content after the brine sample has been added and shows a clear increase in intensity. The third trace (dotted line) shows the water content after the regenerant has been used to release the water from or dry the salt recovery solution, and clearly shows the water content has largely returned to the same prior to brine sample addition. This shows that the salt recovery solution has been regenerated and is ready for reuse. - All GC data was collected on a SHIMADZU Nexis 2030 gas chromatograph fitted with a SUPELCO WATERCOL 1910 column. The GC parameters were set up as shown below:
-
Parameter Setting Injection Volume 1.0 μL Injection temperature 250° C. Injection mode Split Split ratio 100.0 Carrier gas He Carrier gas pressure 53.1 kPa Column flow 0.93 mL/min Liner velocity 22.0 cm/s Column length 30.0 m Column inner diameter 0.32 Column method Isocratic Column temperature 163.0° C. Total time 9 min Detector TCD TCD sample rate 40 ms TCD current 70 mA Makeup gas He Makeup flow 8.0 mL/ min TCD temperature 200° C. - Table 12 below shows the quantity of brine water (measured by GC) in the salt recovery solution after it has been used to absorb water and the quantity of brine water left in the salt recovery solution after it has been regenerated. This provides a measure of how effective the regenerant has been at removing water from the salt recovery solution. As can be seen from Table 12, the quantity of water remaining in the salt recovery solution after regeneration is very small. This shows that the citric acid regenerant has been very effective in removing most of the brine water from the salt recovery solution.
-
TABLE 12 water absorbed by the salt recovery solution and water left in the salt recovery solution after regeneration, calculated from GC data. Wet Salt Recovery Solution Dry Salt Recovery Solution Water Water in Salt absorbed by Recovery Salt GC Peak Recovery Solution after Brine GC Peak GC Peak Solution GC Peak GC Peak regeneration sample position area (mL) position area (mL) 1 3.005 128022 1.10 3.001 99323 0.27 3.006 127162 1.08 3.006 93709 0.20 2 3.001 97534 0.55 3.004 100351 0.35 3.003 72778 0.18 3.004 69319 −0.08 3 3.004 100270 0.61 3.000 72322 0.05 3.005 102327 0.65 3.000 68313 0.05 - Various salt recovery solutions were made up with amine(s) and ketone(s) in various specified molar ratios (see Tables 13-17 below). For each of the salt recovery solutions, brine 300 000 ppm NaCl, was slowly added to 5 mL of the selected salt recovery solution. Upon addition of the brine, the water from the brine was absorbed by the salt recovery solution and the salts in the brine precipitated out. The addition of the brine was stopped as soon as a separate phase started to form at the bottom of the vial after shaking. The presence of the separate phase shows that the maximum absorption capacity of the salt recovery solution has been reached. At this point the volume of the brine, which has been added to the salt recovery solution is recorded.
- The salt and water recovery from the various salt recovery solutions tested in Tables 13-17 below were calculated as described above for the brine samples tested in Examples 2 and 3 above. The additive/regenerant used in this Example was concentrated citric acid solution 0.3 ml.
- The following table, Table 13, summarises the maximum absorbing capacity of the salt recovery solution, along with the water recovery and the salts recovery of the different salt recovery solutions containing different amines with 2-butanone. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 13. The salt and water recovery results listed in this table record one-off tests.
-
TABLE 13 Absorption capacity per 5 Water Ratio mL of salt recov- Salts of recovery ery recov- Amine: solution % ery Amine Ketone Ketone (mL) (vol.) % TEA 2-Butanone 0.5:1 0.55 91 113 Tripropylamine 2-Butanone 0.5:1 0.15 433 45 Dimethylbenzylamine 2-Butanone 0.5:1 0.2 325 128 1-Ethylpiperidine 2-Butanone 0.5:1 0.4 143 91 1-Ethylpyrrolidine 2-Butanone 0.5:1 0.6 83 91 2-Methylpyridine 2-Butanone 0.5:1 0.55 45 119 4-ethylmorpholine 2-Butanone 0.5:1 0.6 83 108 • TEA = triethylamine - The following table 14 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing triethylamine combined with different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following table 14. The salt and water recovery results listed in this table record one-off tests.
-
TABLE 14 Absorption capacity per 5 Water mL of salt recovery Salts recovery % recovery Amine Ketone Ratio solution (mL) (vol.) % TEA Butanone 0.5:1 0.6 92 94 TEA Pentan-3-one 0.5:1 0.15 167 110 TEA Pentan-2-one 0.5:1 0.15 133 136 TEA Cyclopentanone 0.5:1 0.3 100 117 TEA Cyclohexanone 0.5:1 0.25 120 109 TEA Acetophenone 0.5:1 0.15 233 109 TEA 4-Phenyl-2-butanone 0.5:1 0.15 200 108 • TEA = triethylamine - The following table 15 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to three different amines combined with cyclohexanone. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 15. The salt and water recovery results listed in this table record one-off tests.
-
TABLE 15 Ratio of Absorption Amine 1: capacity per 5 Amine 2: mL of salt Water Salts Amine Amine Amine Amine 3: recovery recovery recovery 1 2 3 Ketone Ketone solution (mL) % (vol.) % TEA N-MP* 4-EM CH 0.3: 0.1: 0.1: 1 0.4 75 101 TEA N-MP 4-EM CH 0.3: 0.1: 0.1: 1 0.4 110 108 TEA, N-MP — CH 0.3: 0.2: 1 0.35 86 109 TEA, N-MP — CH 0.3: 0.2: 1 0.35 133 99 TEA, — 4-EM CH 0.3: 0.2: 1 0.4 88 106 — N-MP 4-EM CH 0.3: 0.2: 1 0.5 80 107 TEA = triethylamine N-MP = N-methylpiperidine 4-EM = 4-ethylmorpholine CH = cyclohexanone - The following table 16 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to two different amines combined with up to two different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 16. The salt and water recovery results listed in this table record one-off tests.
-
TABLE 16 Absorption capacity per Ratio of Amine 1: 5 mL of salt Water Salts Amine Amine Ketone Ketone Amine 2: Ketone recovery recovery recovery 1 2 1 2 1: Ketone 2solution (mL) % (vol.) % TEA 4-EM 4-MCH 2-B 0.3: 0.2: 0.5: 0.5 0.3 117 113 TEA, — 4-MCH 2-B 0.5: 0.5: 0.5 0.3 100 107 — 4-EM 4-MCH 2-B 0.5: 0.5: 0.5 0.55 100 118 TEA N-MP 4-MCH 2-B 0.3: 0.2: 0.5: 0.5 0.3 117 106 — N-MP 4-MCH 2-B 0.5: 0.5: 0.5 0.4 120 110 N-MP 4-EM 4-MCH 2-B 0.2: 0.3: 0.5: 0.5 0.45 107 112 N-MP = N-methylpiperidine 4-EM = 4-ethylmorpholine 4-MCH = 4-methylcyclohexanone 2-B = 2-Butanone - The following table 17 summarises the maximum absorbing capacity of the salt recovery solution along with the water recovery and the salts recovery of the different salt recovery solutions containing up to three different amines combined with up to three different ketones. 300 000 ppm NaCl solution was passed through each combination of the salt recovery solution outlined in the following Table 16. The salt and water recovery results listed in this table record one-off tests.
-
TABLE 17 Ratio of Amine Absorption 1: Amine 2: capacity per Amine 3: Ketone 5 mL of salt Water Salts 1: Ketone 2: recovery recovery recovery A1 A2 A3 K1 K2 K3 Ketone 3 (mL) % % TEA N-MP 4-EM 4-MCH 2-B CH 0.3: 0.1: 0.1: 0.4: 0.35 88 107 0.3: 0.3 TEA — — 4-MCH 2-B CH 0.5: 0.4: 0.3: 0.3 0.25 120 114 — N-MP — 4-MCH 2-B CH 0.5: 0.4: 0.3: 0.3 0.35 71 109 — — 4-EM 4-MCH 2-B CH 0.5: 0.4: 0.3: 0.3 0.4 110 109 TEA N-MP — 4-MCH 2-B CH 0.3: 0.2: 0.4: 0.3: 0.25 100 115 0.3 N-MP 4-EM 4-MCH 2-B CH 0.3: 0.2: 0.4: 0.3: 0.35 86 110 0.3 TEA — 4-EM 4-MCH 2-B CH 0.3: 0.2: 0.4: 0.3: 0.3 100 113 0.3 TEA = trimethylamine N-MP = N-methylpiperidine 4-EM = 4-ethylmorpholine 4-MCH = 4-methylcyclohexanone CH = cyclohexanone 2-B = 2-Butanone - It is to be appreciated that from the results shown in Tables 13 to 17 a variety of combinations of amines and ketones can be used successfully as a salt recovery solution.
- An initial 5% NaCl solution (w/w) was made up and serial dilutions of 5, 2.5, 1.25 and 0.625% were made from the initial solution. The osmotic pressure of these dilutions was measured, and a calibration curve was plotted as shown in
FIG. 4 . A trendline equation and R2 value were determined from the calibration plot, as shown onFIG. 4 . The equation was used to calculate the unknown concentrations of NaCl after concentrating a brine solution using the salt recovery solution as described below. - Salt recovery solutions were prepared using: Triethylamine (TEA) and methyl ethyl ketone (MEK), N-Ethylpiperidine and cyclohexanone in the molar ratio of 0.5:1.
- 1 mL of 5% NaCl brine solution (the initial NaCl solution prepared above) was added to the salt recovery solution in ratios (by vol.) varying from 1:1 to 1:20. These samples separated into two layers. The top layer was salt recovery solution and the bottom layer was concentrated brine. The salt concentration in the aqueous layer increased with varying ratios of the salt recovery solution. The concentrated brine layer was separated, and the NaCl concentration was measured using the osmometer. The results for the TEA:MEK salt recovery solution are tabulated in Table 18 and shown in
FIGS. 5 and 6 . -
TABLE 18 Concentration of NaCl brine after concentrating using salt recovery solution (TEA:MEK). Ratio of Brine:Salt Osmotic Pressure Wt. % recovery solution (mOsm/kg) NaCl 1:1 2690 9 1:5 3740 12 1:10 5570 18 1:15 6430 21 1:20 13430 43 - The results for the N-ethylpiperidine:cyclohexanone salt recovery solution are tabulated in Table 19 and shown in
FIGS. 7 and 8 . -
TABLE 19 Concentration of NaCl brine after concentrating using salt recovery solution (N- Ethlpiperidine: Cyclohexanone). Ratio of Brine:Salt Osmotic Pressure Wt. % recovery solution (mOsm/kg) NaCl 1:1 2350 7 1:5 3150 10 1:10 3870 12 1:15 6120 20 1:20 12870 41 - The results shown in Tables 18 and 19 and in
FIGS. 4-8 show the extent to which the original 5% NaCl solution has been concentrated by the two different salt recovery solutions. Essentially water has been recovered from the initial 5% NaCl solution and the water has moved into the salt recovery solution effectively leading to the concentration of the initial 5% NaCl solution. - The experiment described in Example 5 was repeated, except the 5% NaCl solution was replaced with a 20% (w/w) ammonium sulfate (NH4SO4) solution.
- An initial 20% NH4SO4 solution (w/w) was made up and four standard samples were made from the initial solution. The osmotic pressure of these samples was measured, and a calibration curve was plotted as shown in
FIG. 9 . A trendline equation and R2 value were determined from the calibration plot, as shown onFIG. 9 . The equation was used to calculate the unknown concentrations of NH4SO4 after concentrating a brine solution using the salt recovery solution as described below. - Salt recovery solutions were prepared using: Triethylamine (TEA) and methylethylketone (MEK).
- 1 mL of 20% NH4SO4 brine solution (the initial 20% NH4SO4 solution prepared above) was added to the salt recovery solution in ratios (by vol.) varying from 1:5 to 1:30. These samples separated into two layers. The top layer was salt recovery solution and the bottom layer was concentrated brine. The salt concentration in the aqueous layer increased with varying ratios of the salt recovery solution. The concentrated brine layer was separated, and the 20% NH4SO4 concentration was measured using the osmometer. The results for the TEA:MEK salt recovery solution are tabulated in Table 20 and shown in
FIG. 10 . -
TABLE 20 Concentration of NH4SO4 brine after concentrating using salt recovery solution (TEA:MEK). Ratio of Brine:Salt Osmotic Pressure Wt. % recovery solution (mOsm/kg) NH4SO4 1:5 5266.96 31.42 1:10 5753.01 34.35 1:15 6011.97 35.91 1:20 6304.80 37.67 1:35 6478.10 38.71 - The results shown in
FIG. 10 and Table 20 show that the salt recovery solution (TEA:MEK) is capable of concentrating a salt solution to a higher final concentration. Essentially water has been recovered from the initial NH4SO4 solution and the water has moved into the salt recovery solution effectively leading to the concentration of the initial NH4SO4 solution. - The present invention and its embodiments have been described in detail. However, the scope of the present invention is not intended to be limited to the particular embodiments of any process, manufacture, composition of matter, compounds, means, methods, and/or steps described in the specification. Various modifications, substitutions, and variations can be made to the disclosed material without departing from the spirit and/or essential characteristics of the present invention. Accordingly, one of ordinary skill in the art will readily appreciate from the disclosure that later modifications, substitutions, and/or variations performing substantially the same function or achieving substantially the same result as embodiments described herein may be utilized according to such related embodiments of the present invention. Thus, the following claims are intended to encompass within their scope modifications, substitutions, and variations to combinations, kits, compounds, means, methods, and/or steps disclosed herein.
Claims (22)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/753,263 US20200308023A1 (en) | 2017-10-03 | 2018-10-03 | Salt recovery solution and processes of use thereof |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762567545P | 2017-10-03 | 2017-10-03 | |
| PCT/NZ2018/050135 WO2019070134A2 (en) | 2017-10-03 | 2018-10-03 | A salt recovery solution and processes of use thereof |
| US16/753,263 US20200308023A1 (en) | 2017-10-03 | 2018-10-03 | Salt recovery solution and processes of use thereof |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/NZ2018/050135 A-371-Of-International WO2019070134A2 (en) | 2017-10-03 | 2018-10-03 | A salt recovery solution and processes of use thereof |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US18/093,263 Division US11987506B2 (en) | 2017-10-03 | 2023-01-04 | Salt recovery solution and processes of use thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20200308023A1 true US20200308023A1 (en) | 2020-10-01 |
Family
ID=65994968
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/753,263 Abandoned US20200308023A1 (en) | 2017-10-03 | 2018-10-03 | Salt recovery solution and processes of use thereof |
| US18/093,263 Active US11987506B2 (en) | 2017-10-03 | 2023-01-04 | Salt recovery solution and processes of use thereof |
| US18/646,430 Pending US20240368001A1 (en) | 2017-10-03 | 2024-04-25 | Salt recovery solution and processes of use thereof |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US18/093,263 Active US11987506B2 (en) | 2017-10-03 | 2023-01-04 | Salt recovery solution and processes of use thereof |
| US18/646,430 Pending US20240368001A1 (en) | 2017-10-03 | 2024-04-25 | Salt recovery solution and processes of use thereof |
Country Status (21)
| Country | Link |
|---|---|
| US (3) | US20200308023A1 (en) |
| EP (1) | EP3691996A4 (en) |
| JP (1) | JP7291690B2 (en) |
| KR (1) | KR102611749B1 (en) |
| CN (2) | CN111225875B (en) |
| AU (1) | AU2018346086B2 (en) |
| BR (1) | BR112020006722A2 (en) |
| CA (1) | CA3075922A1 (en) |
| CL (1) | CL2020000891A1 (en) |
| CO (1) | CO2020004614A2 (en) |
| EA (1) | EA202090823A1 (en) |
| IL (1) | IL273653B2 (en) |
| MA (1) | MA52603A (en) |
| MX (1) | MX2020003514A (en) |
| PE (1) | PE20210450A1 (en) |
| PH (1) | PH12020500517A1 (en) |
| SA (1) | SA520411670B1 (en) |
| SG (1) | SG11202002320XA (en) |
| TW (1) | TW201922681A (en) |
| WO (1) | WO2019070134A2 (en) |
| ZA (1) | ZA202001746B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11020706B2 (en) | 2016-10-04 | 2021-06-01 | Aquafortus Technologies Limited | Thermo-responsive solution, and method of use therefor |
| US11987506B2 (en) | 2017-10-03 | 2024-05-21 | Aquafortus Technologies Limited | Salt recovery solution and processes of use thereof |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW202104480A (en) * | 2019-04-03 | 2021-02-01 | 紐西蘭商艾克福特士技術有限公司 | A solvent drying composition and processes therfor |
| WO2021247354A1 (en) * | 2020-06-01 | 2021-12-09 | Magna Imperio Systems Corp. | Liquid crystallizer salting out solution |
| MX2022016324A (en) * | 2020-07-10 | 2023-03-15 | Aquafortus Tech Limited | A solvent drying solution and processes therefor. |
| BR112023000494A2 (en) * | 2020-07-10 | 2023-03-28 | Aquafortus Tech Limited | SALT RECOVERY SOLUTION AND USE PROCESSES |
| TWI745208B (en) * | 2020-12-30 | 2021-11-01 | 財團法人工業技術研究院 | System and method of treating waste water |
Family Cites Families (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3077500A (en) * | 1955-06-23 | 1963-02-12 | Celanese Corp | Aldol condensation reactions |
| US3130156A (en) | 1960-12-13 | 1964-04-21 | Ray A Neff | Solvent extractor |
| BE615339A (en) * | 1961-03-29 | |||
| FR2460276A1 (en) * | 1979-07-03 | 1981-01-23 | Rhone Poulenc Ind | PROCESS FOR TREATING RARE EARTH OXIDES AND GALLIUM MIXTURES |
| JPS5935373B2 (en) | 1979-07-06 | 1984-08-28 | ダイセル化学工業株式会社 | Acetic acid recovery method |
| JPS5912705A (en) * | 1982-07-13 | 1984-01-23 | Daicel Chem Ind Ltd | Extracting method |
| CA1208134A (en) * | 1983-12-23 | 1986-07-22 | Kenzo Arizono | Pranoprofen gelled ointment |
| US5346620A (en) * | 1993-08-31 | 1994-09-13 | Basf Corporation | Process for waste water treatment by removal of sodium sulfate |
| US5705074A (en) * | 1996-11-19 | 1998-01-06 | Merichem Company | Removal of acidic organic contaminants from refinery waste water |
| DE69839323T2 (en) | 1997-12-09 | 2009-04-09 | National Institute Of Advanced Industrial Science And Technology | Stimulus-responsive polymer via keto-enol tautomerism |
| US6307087B1 (en) | 1998-07-10 | 2001-10-23 | Massachusetts Institute Of Technology | Ligands for metals and improved metal-catalyzed processes based thereon |
| TW508891B (en) | 2000-02-21 | 2002-11-01 | Misubishi Electric Corp | Stator iron core of electric motor, manufacturing method thereof, electric motor, and compresor |
| US7560029B2 (en) | 2001-02-01 | 2009-07-14 | Yale University | Osmotic desalination process |
| US9839879B2 (en) | 2010-04-30 | 2017-12-12 | Woongjin Chemical Co., Ltd. | Forward osmosis membrane for seawater desalination and method for preparing the same |
| WO2011136465A2 (en) | 2010-04-30 | 2011-11-03 | 웅진케미칼 주식회사 | Forward osmosis membrane for seawater desalination, and method for manufacturing same |
| WO2012043669A1 (en) | 2010-09-29 | 2012-04-05 | 富士フイルム株式会社 | Forward osmosis device, and forward osmosis method |
| EP2651992B1 (en) | 2010-12-15 | 2020-01-22 | Queen's University At Kingston | Method for use of water with switchable ionic strength |
| KR101220261B1 (en) | 2011-07-27 | 2013-02-07 | 서울대학교산학협력단 | Thermal responsive draw agent for forward osmosis desalination and the forward osmosis desalination method using the draw agent |
| ES2374249B1 (en) * | 2011-10-24 | 2012-12-20 | Institut Químic De Sarrià Cets Fundació Privada | ADHESIVE COMPOSITION. |
| WO2013065293A1 (en) | 2011-10-31 | 2013-05-10 | Jfeエンジニアリング株式会社 | Method and device for preparing fresh water |
| US9447239B2 (en) | 2012-03-19 | 2016-09-20 | Samsung Electronics Co., Ltd. | Thermosensitive copolymers, forward osmosis water treatment devices including the same, and methods of producing and using the same |
| WO2013175380A1 (en) | 2012-05-21 | 2013-11-28 | Re10 Ltd. | Methods and systems for water recovery |
| CN105026019A (en) | 2012-11-16 | 2015-11-04 | Oasys水有限公司 | Draw solutions and draw solute recovery for osmotically driven membrane processes |
| EP2928586A1 (en) | 2012-12-04 | 2015-10-14 | McGinnis, Robert | Signal responsive solutes |
| US20150360973A1 (en) | 2013-01-28 | 2015-12-17 | Aharon Eyal | Methods and systems for water recovery |
| JP6330230B2 (en) | 2013-03-11 | 2018-05-30 | ユニヴァーシティー オブ ノートル ダム デュ ラック | Multi-block copolymer and method of use |
| US10384167B2 (en) | 2013-11-21 | 2019-08-20 | Oasys Water LLC | Systems and methods for improving performance of osmotically driven membrane systems |
| WO2016094835A1 (en) | 2014-12-12 | 2016-06-16 | Artesion, Inc. | A membrane and gas recycling system for forward osmosis water treatment systems using switchable polar solvents |
| AU2016220549B2 (en) | 2015-02-17 | 2021-09-23 | Nanyang Technological University | Regenerable draw solute for osmotically driven processes |
| US10195543B2 (en) | 2016-06-09 | 2019-02-05 | Battelle Energy Alliance, Llc | Methods and systems for treating a switchable polarity material, and related methods of liquid treatment |
| TWI757350B (en) | 2016-10-04 | 2022-03-11 | 紐西蘭商艾克福特士技術有限公司 | A thermo-responsive solution, and method of use therefor |
| US10933377B2 (en) | 2017-09-29 | 2021-03-02 | Aquafortus Technologies Limited | Apparatus and apparatus assembly |
| US20200308023A1 (en) * | 2017-10-03 | 2020-10-01 | Aquafortus Technologies Limited | Salt recovery solution and processes of use thereof |
| EP3935035A4 (en) | 2019-03-08 | 2022-12-07 | Aquafortus Technologies Limited | An alcohol recovery solution, and method of use therefor |
| TW202104480A (en) | 2019-04-03 | 2021-02-01 | 紐西蘭商艾克福特士技術有限公司 | A solvent drying composition and processes therfor |
-
2018
- 2018-10-03 US US16/753,263 patent/US20200308023A1/en not_active Abandoned
- 2018-10-03 IL IL273653A patent/IL273653B2/en unknown
- 2018-10-03 BR BR112020006722-9A patent/BR112020006722A2/en not_active Application Discontinuation
- 2018-10-03 CN CN201880063567.XA patent/CN111225875B/en active Active
- 2018-10-03 WO PCT/NZ2018/050135 patent/WO2019070134A2/en active Application Filing
- 2018-10-03 AU AU2018346086A patent/AU2018346086B2/en active Active
- 2018-10-03 MX MX2020003514A patent/MX2020003514A/en unknown
- 2018-10-03 EP EP18864958.6A patent/EP3691996A4/en active Pending
- 2018-10-03 MA MA052603A patent/MA52603A/en unknown
- 2018-10-03 KR KR1020207012734A patent/KR102611749B1/en active IP Right Grant
- 2018-10-03 JP JP2020517910A patent/JP7291690B2/en active Active
- 2018-10-03 CN CN202310281930.8A patent/CN116282078A/en active Pending
- 2018-10-03 TW TW107134912A patent/TW201922681A/en unknown
- 2018-10-03 EA EA202090823A patent/EA202090823A1/en unknown
- 2018-10-03 PE PE2020000882A patent/PE20210450A1/en unknown
- 2018-10-03 SG SG11202002320XA patent/SG11202002320XA/en unknown
- 2018-10-03 CA CA3075922A patent/CA3075922A1/en active Pending
-
2020
- 2020-03-13 PH PH12020500517A patent/PH12020500517A1/en unknown
- 2020-03-19 ZA ZA2020/01746A patent/ZA202001746B/en unknown
- 2020-03-31 SA SA520411670A patent/SA520411670B1/en unknown
- 2020-04-02 CL CL2020000891A patent/CL2020000891A1/en unknown
- 2020-04-16 CO CONC2020/0004614A patent/CO2020004614A2/en unknown
-
2023
- 2023-01-04 US US18/093,263 patent/US11987506B2/en active Active
-
2024
- 2024-04-25 US US18/646,430 patent/US20240368001A1/en active Pending
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11020706B2 (en) | 2016-10-04 | 2021-06-01 | Aquafortus Technologies Limited | Thermo-responsive solution, and method of use therefor |
| US11826704B2 (en) | 2016-10-04 | 2023-11-28 | Aquafortus Technologies Limited | Thermo-responsive solution, and method of use therefor |
| US11987506B2 (en) | 2017-10-03 | 2024-05-21 | Aquafortus Technologies Limited | Salt recovery solution and processes of use thereof |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11987506B2 (en) | Salt recovery solution and processes of use thereof | |
| CN104071940A (en) | Treatment method of butanol and octanol waste alkali liquor | |
| WO2009115888A1 (en) | Process for the recovery of water and valuable organics from wastewater in the production of aromatic carboxylic acids | |
| CN113891760A (en) | Solvent drying compositions and related methods | |
| EP2334631A1 (en) | Method for separating acrylic acid, contained as the main constituent, and glyoxal, contained as a by-product, from a gaseous product mixture of a c3 precursor compound of acrylic acid | |
| WO2012045738A1 (en) | Process for inhibiting unwanted free-radical polymerization of acrylic acid present in a liquid phase p | |
| BR112016001554B1 (en) | method for the continuous recovery of (meth) acrylic acid and apparatus for the method | |
| JP2018524332A5 (en) | ||
| NZ761109B2 (en) | A salt recovery solution and processes of use thereof | |
| JP7016366B2 (en) | Method for preventing polymer precipitation in the method for purifying (meth) acrylic acid | |
| EA045660B1 (en) | SOLUTION FOR EXTRACTION OF SALT AND METHODS OF ITS USE | |
| JP2003160530A (en) | Method for purifying (meth)acrylic acid | |
| JP2014050834A (en) | Treatment method of waste water flow generated in post-treatment of triacetone amine-containing reaction mixture | |
| US20230286833A1 (en) | A salt recovery solution and processes of use thereof | |
| KR100916286B1 (en) | Selective separatin of xylene isomer from xylene mixture by inclusion using guanidinium biphenyl¡ª2,2¡¯¡ªdiyldimethanesulfonate host compound | |
| CA3185722A1 (en) | Liquid crystallizer salting out solution | |
| WO2002010097A1 (en) | A process for separating o- and p-substituted benzene compounds | |
| KR100949140B1 (en) | Method for selective separation of xylene isomer by selcective inclusion with guanidinium 4?chlorobenzene sulfonate | |
| WO2012118196A1 (en) | Method for concentrating aqueous 3-hydroxy-2,2-dimethylpropanal solution | |
| CN106854196A (en) | A kind of synthesis technique of organic synthesis intermediate cyclohexanone ethylene ketal |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: AQUAFORTUS TECHNOLOGIES LIMITED, NEW ZEALAND Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:BRIGGS, DARYL JOSEPH;PRAKASH, CHAITRA;REEL/FRAME:053750/0186 Effective date: 20200701 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: APPLICATION DISPATCHED FROM PREEXAM, NOT YET DOCKETED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |