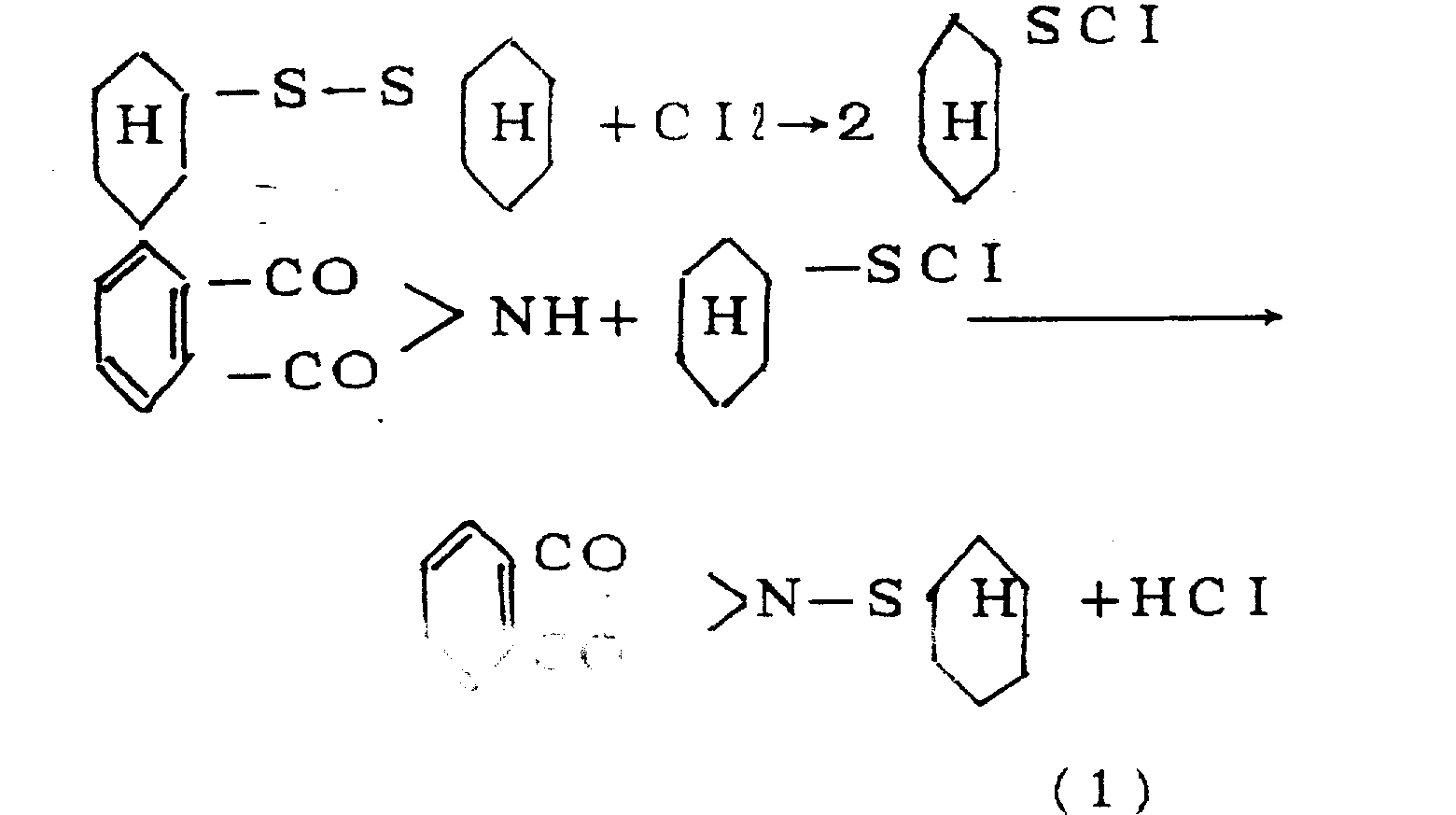CN1023120C - Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide - Google Patents
Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide Download PDFInfo
- Publication number
- CN1023120C CN1023120C CN 90106379 CN90106379A CN1023120C CN 1023120 C CN1023120 C CN 1023120C CN 90106379 CN90106379 CN 90106379 CN 90106379 A CN90106379 A CN 90106379A CN 1023120 C CN1023120 C CN 1023120C
- Authority
- CN
- China
- Prior art keywords
- reaction
- cyclohexyl
- chlorocyclohexane
- chlorine
- mole ratio
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Indole Compounds (AREA)
Abstract
The present invention relates to a cyclohexane chlorination method for preparing chemical N-cyclohexyl thiophthalimide, namely a coking preventing agent CTP, of N-cyclohexyl thiophthalimide coking preventing agent CTP. The structural formula is *. The coking preventing agent CTP is a tried and true additive for preventing the coking in the process of the processing of rubber, and is widely applied to the processes of the processing and the production of the rubber to have the definite function for preventing rubber materials from being coked.
Description
N-cyclohexyl thio phthalimide (abbreviation anti-scorching agent CTP)
Structural formula:
Molecular formula: C14H15O2SN
Anti-scorching agent CTP is the effective additive that prevents incipient scorch in the Rubber processing process, report in 73 pages of the 6th phases of rubber bibliography 1974: anti-scorching agent CTP can be used for natural rubber and synthetic rubber, add a spot of anti-scorching agent CTP and just can prevent sizing material generation scorching (being incipient scorch) effectively, anti-scorching agent CTP has been widely used in the Rubber processing production process, particularly in the process that modern age, rubber processing developed towards the high temperature and high speed direction, the incipient scorch of sizing material is a recurrent problem in the Rubber processing process, the application of anti-scorching agent CTP can be satisfied technologic requirement, the incipient scorch that prevents sizing material has been played certain effect, vulcanization characteristics and cured properties to sizing material have no adverse effects, and can improve the package stability of rubber unvulcanizate.1976 the 20th phase 26-34 of american industrial report page or leaf report: the at first synthetic anti-scorching agent CTPs of U.S. Monsanto Company in 1970 are also gone into operation, and trade name is PVI.Fr1, and 500,844(1976) middle report; The method of producing anti-scorching agent CTP is to be the intermediate that raw material is produced CTP with cyclohexylmercaptan or hexalin, and its reaction equation is:
Adopt aforesaid method that following weak point is arranged:
1, the odd flavor of smelling of cyclohexylmercaptan.
The mole ratio of hexalin and hydrochloric acid consumption is 1: 4 when 2, being the synthetic chlorocyclohexane of raw material with hexalin and hydrochloric acid, and excessive hydrochloric acid has brought three-waste pollution and equipment corrosion problem.
3, be that raw material is to be catalyzer with ZnCl2 when producing chlorocyclohexane with the hexalin, make complicated operating processization, and strengthened cost.
4, hair ring hexanol or cyclohexylmercaptan are that the synthetic chlorocyclohexane yield of raw material is lower, have only 30-40%, and the hexalin price are more expensive.
The objective of the invention is to solve the environmental pollution problem, simplify technology, reduce cost, improve feed stock conversion and yield in order to solve etching problem.
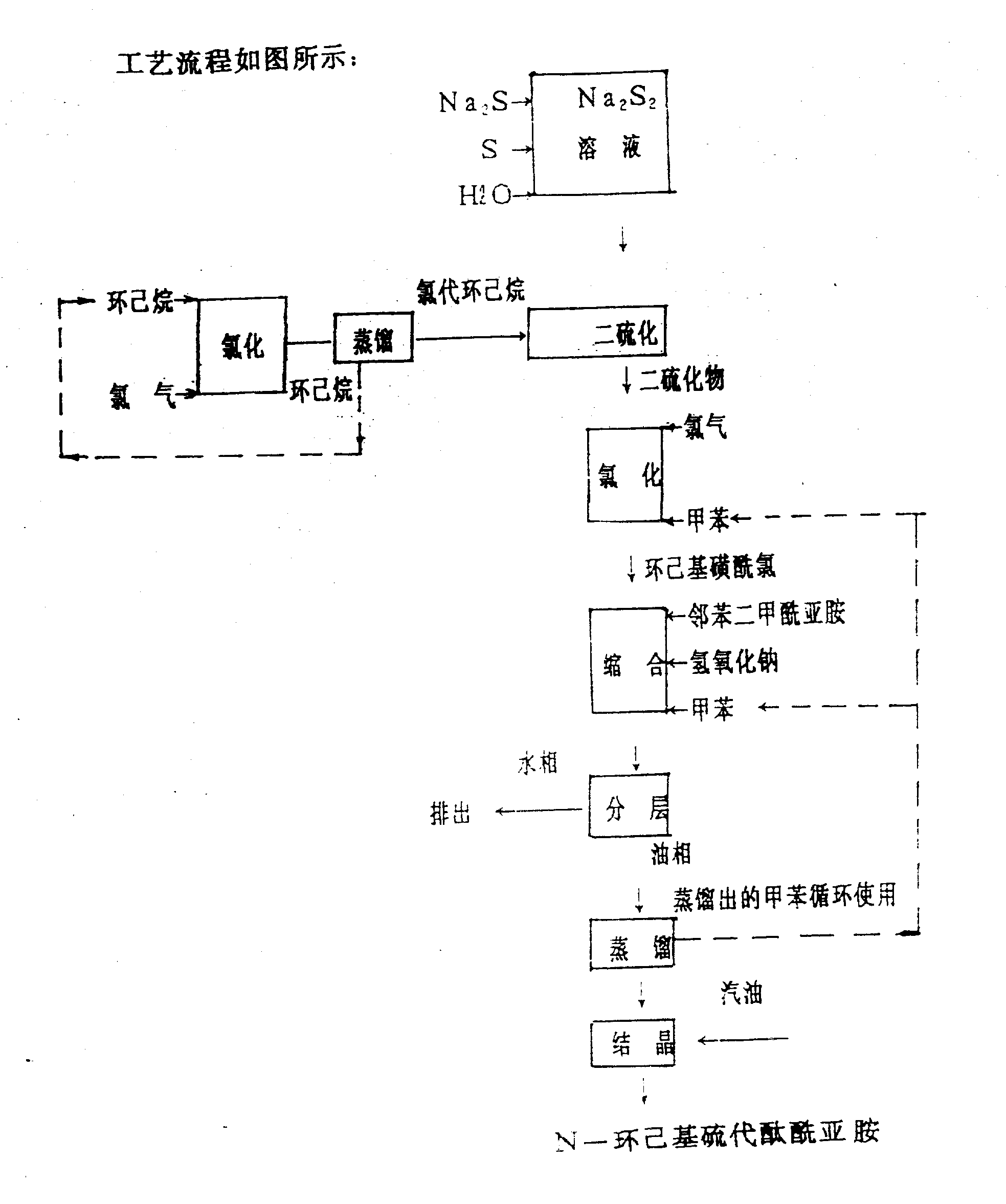
The present invention is that the employing hexanaphthene is that raw material is produced CTP's.The present invention realizes as follows:
At first make the raw material hexanaphthene in the chlorination reaction jar in 50-90 ℃ down and chlorine carry out chlorination reaction and make chlorocyclohexane, react in the curing still with sodium disulfide again and make dicyclohexyl bisulfide, reaction equation is as follows:
Dicyclohexyl bisulfide in the chlorination reaction jar in 10-40 ℃ down and chlorine carry out chlorination reaction, obtain cyclohexyl sulphur muriate, in the condensation reaction jar, under 40-80 ℃, carry out condensation reaction with phthalic imidine again and obtain N-cyclohexyl thio phthalimide (being anti-scorching agent CTP).Its reaction equation is as follows:
Characteristics of the present invention are:
1, hexanaphthene generates chlorocyclohexane through chlorination reaction, the hexanaphthene of the reaction of not participating in to recycle after distillation is reclaimed.
2, the chloride process of dicyclohexyl bisulfide and condensation operation adopt with a kind of solvent among the present invention, have simplified technical process, make aftertreatment simple and convenient, easy handling.Also help simultaneously the recovery and the recycle of solvent, help reducing cost.And being two operations, old technology uses two kinds of different solvents respectively.
3, gained anti-scorching agent CTP color and luster is a yellowish white.
Example 1
First part
Get 100 milliliters of hexanaphthenes and in reactor, carry out chlorination reaction generation chlorocyclohexane at 50-90 ℃ of following logical chlorine, reach 70% when above with the content of gas chromatographic analysis chlorocyclohexane, get final product stopped reaction, generally after reaction two hours, conversion of cyclohexane can reach more than 70%, the hexanaphthene that the reaction of not participating in is reclaimed in reaction mass rectifying, chlorocyclohexane that generates and Sodium Sulphide are with 1: an amount of water of the molar ratio adding of 0.5-0.7 is made solvent and is carried out curing reaction generation dicyclohexyl bisulfide, aqueous-phase material is removed in layering, distillation is removed foreshot and is got purified dicyclohexyl bisulfide, yield 90%, content 98%.
Second section
1, chlorination; Get 1 molar disulphide and the molar chlorine of 1-1.2 at first carries out chlorination reaction, 10-40 ℃ down reaction generated cyclohexyl sulphur muriate in 8-15 minute.
2, condensation: in there-necked flask, add 2 molar phthalic imidines, add an amount of toluene solvant, the cyclohexyl sulphur muriate and the alkaline solution of chlorination gained are added in the there-necked flask, in 40-80 ℃ of following condensation reaction after 2 hours, condensation reaction solution is moved on to static layering in the funnel, tell aqueous-phase material, toluene solvant is removed in the oil phase thing underpressure distillation that will contain CTP, and recovered solvent recycles.The solution that contains CTP after concentrating is added boiling range 70-90 ℃ gas and oil under 0-20 ℃, the CTP crystallization is separated out, the CTP that after suction filtration, drying, gets product, yield 85%, product are the oyster white crystal, content 99%.
Claims (1)
1, the preparation method of N-cyclohexyl thio phthalimide, it is characterized in that adopting hexanaphthene is raw material, under 50-90 ℃, carry out chlorination reaction with chlorine and produce chlorocyclohexane, do not recycle after the hexanaphthene distillation that does not transform to reclaim, when dicyclohexyl bisulfide is produced in chlorocyclohexane and sodium disulfide reaction, the mole ratio of chlorocyclohexane and sodium disulfide consumption is 1: 0.5-0.7, reaction is to be solvent with water, carry out under the normal temperature, dicyclohexyl bisulfide in 10-40 ℃ down and chlorine reaction produce cyclohexyl sulphur muriate, the mole ratio of disulphide and chlorine is 1: 1-1.2, the mole ratio that N-cyclohexyl thio phthalimide is produced in dicyclohexyl bisulfide and phthalic imidine condensation is 1: 1, temperature of reaction is 40-80 ℃, the chlorination of dicyclohexyl bisulfide and condensation operation are all used with a kind of solvent, can be benzene, toluene, dimethylbenzene, the N-cyclohexyl thio phthalimide crude product that makes makes elaboration with the gasoline of 70-90 ℃ of boiling point in 0-20 ℃ of following recrystallization.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 90106379 CN1023120C (en) | 1990-09-18 | 1990-09-18 | Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 90106379 CN1023120C (en) | 1990-09-18 | 1990-09-18 | Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1059906A CN1059906A (en) | 1992-04-01 |
| CN1023120C true CN1023120C (en) | 1993-12-15 |
Family
ID=4880020
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 90106379 Expired - Fee Related CN1023120C (en) | 1990-09-18 | 1990-09-18 | Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1023120C (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101070296B (en) * | 2006-05-09 | 2012-12-05 | 东丽精密化学株式会社 | Preparation method of dicyclohexyldisulfide |
| CN102906069B (en) * | 2010-08-26 | 2014-05-28 | 东丽精细化工株式会社 | Preparation method of dicyclohexyl disulfide |
| CN102382021A (en) * | 2011-09-19 | 2012-03-21 | 张迎宾 | Method for preparing dicyclohexyl disulfide |
| CN102993069A (en) * | 2011-09-19 | 2013-03-27 | 河南省汤阴县永新助剂厂 | Automatic salt separation technique and device in dicyclohexyl disulfide reaction |
| CN108083970A (en) * | 2017-12-19 | 2018-05-29 | 山东戴瑞克新材料有限公司 | A kind of method that anti-scorching agent CTP intermediate dicyclohexyl bisulfide is continuously synthesizing to |
-
1990
- 1990-09-18 CN CN 90106379 patent/CN1023120C/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN1059906A (en) | 1992-04-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4730034A (en) | Recovering poly(arylene sulfide) oligomers from a poly(arylene sulfide) reaction mixture | |
| CN1023120C (en) | Cyclohexane Chlorination process for obtaining N-cyclohexyl thiophthalimide | |
| CN1064674A (en) | The preparation method of forulic acid | |
| CN109705347B (en) | Process method for separating oligomer from polyphenylene sulfide resin synthetic slurry | |
| CN103508938A (en) | Preparation method of N, N'-bis(2, 2, 6, 6-tetramethyl-4-piperidyl)-1, 3-benzenedicarboxamide | |
| US3907854A (en) | Dialkyl thionocarbamate method | |
| CN1060205C (en) | Process for refining diesel oil by solvent extraction and denitrification | |
| CN1026233C (en) | Process for production of lower dialkyl disulphides | |
| CN114957049B (en) | Synthesis method of N-phenyl-N-trichloromethylthiobenzene sulfonamide | |
| CN112707558B (en) | Method for recycling waste water | |
| CN1217326A (en) | Process for preparing dimethyl thio-ether and methylthio-alcohol | |
| CN1166699C (en) | Recovery of modifier compounds and polar organic compounds from a poly (arylene sulfide) | |
| CN1927851A (en) | Synthesis method of allyl glycidyl ether | |
| CN1246843A (en) | Method for separating imine from mixture containing amine and imine | |
| US3975264A (en) | Flotation of copper sulfide ores with improved thionocarbamates | |
| CN105037082A (en) | Method for recycling paradichlorobenzene in polyphenylene sulphide production mother liquor | |
| US2635120A (en) | Obtaining purified phenols and cresols of petroleum origin | |
| CN110003266B (en) | Environment-friendly production method of high-quality 3-octanoyl thio-1-propyltriethoxysilane | |
| CA2025242A1 (en) | Polar aprotic catalysts for formation of fluorosilicone fluids | |
| US4239885A (en) | Process for the preparation of diacetone acrylamide | |
| KR920019815A (en) | Method for preparing α-aspartyl-L-phenylalanine methyl ester | |
| CN112707876A (en) | Preparation method of 2,2' -di (4-hydroxycyclohexyl) propane glycidyl ether | |
| CN1377375A (en) | Methanol extraction polar organic compounds and modifier compounds from poly (arylene sulfide) polymer and oligomer streams | |
| CN1812969A (en) | Method for the production of trimeric ketone peroxide solutions | |
| KR100590416B1 (en) | Purification of Thiophene |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C06 | Publication | ||
| PB01 | Publication | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |