Here is a recording of the first BGA23 workshop session on BRAKER. If learning by watching videos is easy for you, consider watching that: https://www.youtube.com/watch?v=UXTkJ4mUkyg
BRAKER3 is now in https://usegalaxy.eu/
TSEBRA & BRAKER3 related:
- Lars Gabriel, University of Greifswald, Germany, [email protected]
BRAKER & AUGUSTUS related:
- Katharina J. Hoff, University of Greifswald, Germany, [email protected], +49 3834 420 4624,
GeneMark related:
-
Mark Borodovsky, Georgia Tech, U.S.A., [email protected]
-
Tomas Bruna, Joint Genome Institute, U.S.A., [email protected]
-
Alexandre Lomsazde, Georgia Tech, U.S.A., [email protected]
[a] University of Greifswald, Institute for Mathematics and Computer Science, Walther-Rathenau-Str. 47, 17489 Greifswald, Germany
[b] University of Greifswald, Center for Functional Genomics of Microbes, Felix-Hausdorff-Str. 8, 17489 Greifswald, Germany
[c] Joint Georgia Tech and Emory University Wallace H Coulter Department of Biomedical Engineering, 30332 Atlanta, USA
[d] School of Computational Science and Engineering, 30332 Atlanta, USA
[e] Moscow Institute of Physics and Technology, Moscow Region 141701, Dolgoprudny, Russia
Figure 1: Current BRAKER authors, from left to right: Mario Stanke, Alexandre Lomsadze, Katharina J. Hoff, Tomas Bruna, Lars Gabriel, and Mark Borodovsky. We acknowledge that a larger community of scientists contributed to the BRAKER code (e.g. via pull requests).
The development of BRAKER1, BRAKER2, and BRAKER3 was supported by the National Institutes of Health (NIH) [GM128145 to M.B. and M.S.]. Development of BRAKER3 was partially funded by Project Data Competency granted to K.J.H. and M.S. by the government of Mecklenburg-Vorpommern, Germany.
The Transcript Selector for BRAKER (TSEBRA) is available at https://github.com/Gaius-Augustus/TSEBRA .
GeneMark-ETP, one of the gene finders at the core of BRAKER, is available at https://github.com/gatech-genemark/GeneMark-ETP .
AUGUSTUS, the second gene finder at the core of BRAKER, is available at https://github.com/Gaius-Augustus/Augustus .
GALBA, a BRAKER pipeline spin-off for using Miniprot or GenomeThreader to generate training genes, is available at https://github.com/Gaius-Augustus/GALBA .
- Authors
- Funding
- What is BRAKER?
- Keys to successful gene prediction
- Overview of modes for running BRAKER
- Container
- Installation
- Running BRAKER
- Output of BRAKER
- Example data
- Starting BRAKER on the basis of previously existing BRAKER runs
- Bug reporting
- Citing BRAKER and software called by BRAKER
- License
The rapidly growing number of sequenced genomes requires fully automated methods for accurate gene structure annotation. With this goal in mind, we have developed BRAKER1R1R0, a combination of GeneMark-ET R2 and AUGUSTUS R3, R4, that uses genomic and RNA-Seq data to automatically generate full gene structure annotations in novel genome.
However, the quality of RNA-Seq data that is available for annotating a novel genome is variable, and in some cases, RNA-Seq data is not available, at all.
BRAKER2 is an extension of BRAKER1 which allows for fully automated training of the gene prediction tools GeneMark-ES/ET/EP/ETP R14, R15, R17, F1 and AUGUSTUS from RNA-Seq and/or protein homology information, and that integrates the extrinsic evidence from RNA-Seq and protein homology information into the prediction.
In contrast to other available methods that rely on protein homology information, BRAKER2 reaches high gene prediction accuracy even in the absence of the annotation of very closely related species and in the absence of RNA-Seq data.
BRAKER3 is the latest pipeline in the BRAKER suite. It enables the usage of RNA-seq and protein data in a fully automated pipeline to train and predict highly reliable genes with GeneMark-ETP and AUGUSTUS. The result of the pipeline is the combined gene set of both gene prediction tools, which only contains genes with very high support from extrinsic evidence.
In this user guide, we will refer to BRAKER1, BRAKER2, and BRAKER3 simply as BRAKER because they are executed by the same script (braker.pl).
-
Use a high quality genome assembly. If you have a huge number of very short scaffolds in your genome assembly, those short scaffolds will likely increase runtime dramatically but will not increase prediction accuracy.
-
Use simple scaffold names in the genome file (e.g.
>contig1will work better than>contig1my custom species namesome putative function /more/information/ and lots of special characters %&!*(){}). Make the scaffold names in all your fasta files simple before running any alignment program. -
In order to predict genes accurately in a novel genome, the genome should be masked for repeats. This will avoid the prediction of false positive gene structures in repetitive and low complexitiy regions. Repeat masking is also essential for mapping RNA-Seq data to a genome with some tools (other RNA-Seq mappers, such as HISAT2, ignore masking information). In case of GeneMark-ES/ET/EP/ETP and AUGUSTUS, softmasking (i.e. putting repeat regions into lower case letters and all other regions into upper case letters) leads to better results than hardmasking (i.e. replacing letters in repetitive regions by the letter
Nfor unknown nucleotide). -
Many genomes have gene structures that will be predicted accurately with standard parameters of GeneMark-ES/ET/EP/ETP and AUGUSTUS within BRAKER. However, some genomes have clade-specific features, i.e. special branch point model in fungi, or non-standard splice-site patterns. Please read the options section [options] in order to determine whether any of the custom options may improve gene prediction accuracy in the genome of your target species.
-
Always check gene prediction results before further usage! You can e.g. use a genome browser for visual inspection of gene models in context with extrinsic evidence data. BRAKER supports the generation of track data hubs for the UCSC Genome Browser with MakeHub for this purpose.
BRAKER mainly features semi-unsupervised, extrinsic evidence data (RNA-Seq and/or protein spliced alignment information) supported training of GeneMark-ES/ET/EP/ETP[F1] and subsequent training of AUGUSTUS with integration of extrinsic evidence in the final gene prediction step. However, there are now a number of additional pipelines included in BRAKER. In the following, we give an overview of possible input files and pipelines:
- Genome file, only. In this mode, GeneMark-ES is trained on the genome sequence, alone. Long genes predicted by GeneMark-ES are selected for training AUGUSTUS. Final predictions by AUGUSTUS are ab initio. This approach will likely yield lower prediction accuracy than all other here described pipelines. (see Figure 2),
Figure 2: BRAKER pipeline A: training GeneMark-ES on genome data, only; ab initio gene prediction withAUGUSTUS
- Genome and RNA-Seq file from the same species (see figure 3); this approach is suitable for short read RNA-Seq libraries with a good coverage of the transcriptome, important: this approach requires that each intron is covered by many alignments, i.e. it does not work with assembled transcriptome mappings. In principle, also alignments of long read RNA-Seq data may lead to sufficient data for running BRAKER, but only if each transcript that will go into training was sequenced and aligned to the genome multiple times. Please be aware that at the current point in time, BRAKER does not officially support the integration of long read RNA-Seq data, yet.
Figure 3: BRAKER pipeline B: training GeneMark-ET supported by RNA-Seq spliced alignment information, prediction with AUGUSTUS with that same spliced alignment information.
- Genome file and database of proteins that may be of unknown evolutionary distance to the target species (see Figure 4); this approach is particularly suitable if no RNA-Seq data is available. This method will work better with proteins from species that are rather close to the target species, but accuracy will drop only very little if the reference proteins are more distant from the target species. Important: This approach requires a database of protein families, i.e. many representatives of each protein family must be present in the database. BRAKER has been tested with OrthoDB R19, successfully. The ProtHint R18 protein mapping pipeline for generating required hints for BRAKER is available for download at https://github.com/gatech-genemark/ProtHint, the software on how to prepare the OrthoDB input proteins is available at https://github.com/tomasbruna/orthodb-clades. You may add proteins of a closely related species to the OrthoDB fasta file in order to incorporate additional evidence into gene prediction. We provide pre-partitioned OrthoDB v.11 clades for download at https://bioinf.uni-greifswald.de/bioinf/partitioned_odb11/ , and OrthoDB v.12 clades at https://bioinf.uni-greifswald.de/bioinf/partitioned_odb12/.
Figure 4: BRAKER pipeline C: training GeneMark-EP+ on protein spliced alignment, start and stop information, prediction with AUGUSTUS with that same information, in addition chained CDSpart hints. Proteins used here can be of any evolutionary distance to the target organism.
- Genome file and RNA-Seq set(s) from the same species, and proteins that may be of unknown evolutionary distance to the target species (see figure 5); important: this approach requires a database of protein families, i.e. many representatives of each protein family must be present in the database, e.g. OrthoDB is suitable. (You may add proteins of a closely related species to the OrthoDB fasta file in order to incorporate additional evidence into gene prediction.)
Figure 5: BRAKER pipeline D: If necessary, download and alignment of RNA-Seq sets for the target species. Training of GeneMark-ETP supported by the RNA-Seq alignments and a large protein database (proteins can be of any evolutionary distance). Subsequently, AUGUSTUS training and prediction using the same extrinsic information together with the GeneMark-ETP results. The final prediction is the TSEBRA combination of the AUGUSTUS and GeneMark-ETP results.
We are aware that the "manual" installation of BRAKER3 and all its dependencies is tedious and really challenging without root permissions. Therefore, we provide a Docker container that has been developed to be run with Singularity. All information on this container can be found at https://hub.docker.com/r/teambraker/braker3
In short, build it as follows:
singularity build braker3.sif docker:https://teambraker/braker3:latest
Execute with:
singularity exec braker3.sif braker.pl
Test with:
singularity exec -B $PWD:$PWD braker3.sif cp /opt/BRAKER/example/singularity-tests/test1.sh .
singularity exec -B $PWD:$PWD braker3.sif cp /opt/BRAKER/example/singularity-tests/test2.sh .
singularity exec -B $PWD:$PWD braker3.sif cp /opt/BRAKER/example/singularity-tests/test3.sh .
export BRAKER_SIF=/your/path/to/braker3.sif # may need to modify
bash test1.sh
bash test2.sh
bash test3.sh
Few users want to run their analysis inside Docker (since root permissions are required). However, if that's your goal, you can run and test the container as follows
sudo docker run --user 1000:100 --rm -it teambraker/braker3:latest bash
bash /opt/BRAKER/example/docker-tests/test1.sh # BRAKER1
bash /opt/BRAKER/example/docker-tests/test2.sh # BRAKER2
bash /opt/BRAKER/example/docker-tests/test3.sh # BRAKER3
Good luck ;-)
$PATH variable might lead to unforeseen interferences, causing program failures. Please move all older GeneMark versions out of your $PATH (also e.g. the GeneMark in ProtHint/dependencies).
At the time of release, this BRAKER version was tested with:
-
AUGUSTUS 3.5.0 F2
-
GeneMark-ETP (source see Dockerfile)
-
BAMTOOLS 2.5.1R5
-
SAMTOOLS 1.7-4-g93586edR6
-
DIAMOND 0.9.24
-
cdbfasta 0.99
-
cdbyank 0.981
-
GUSHR 1.0.0
-
SRA Toolkit 3.00 R14
-
HISAT2 2.2.1 R15
-
BEDTOOLS 2.30 R16
-
StringTie2 2.2.1 R17
-
GFFRead 0.12.7 R18
-
compleasm 0.2.5 R27
Running BRAKER requires a Linux-system with bash and Perl. Furthermore, BRAKER requires the following CPAN-Perl modules to be
installed:
-
File::Spec::Functions -
Hash::Merge -
List::Util -
MCE::Mutex -
Module::Load::Conditional -
Parallel::ForkManager -
POSIX -
Scalar::Util::Numeric -
YAML -
Math::Utils -
File::HomeDir
For GeneMark-ETP, used when protein and RNA-Seq is supplied:
YAML::XSData::DumperThread::Queuethreads
On Ubuntu, for example, install the modules with CPANminusF4: sudo cpanm Module::Name, e.g. sudo cpanm Hash::Merge.
BRAKER also uses a Perl module helpMod_braker.pm that is not available on CPAN. This module is part of the BRAKER release and does not require separate installation.
If you do not have root permissions on the Linux machine, try setting up an Anaconda (https://www.anaconda.com/distribution/) environment as follows:
wget https://repo.anaconda.com/archive/Anaconda3-2018.12-Linux-x86_64.sh
bash bin/Anaconda3-2018.12-Linux-x86_64.sh # do not install VS (needs root privileges)
conda install -c anaconda perl
conda install -c anaconda biopython
conda install -c bioconda perl-app-cpanminus
conda install -c bioconda perl-file-spec
conda install -c bioconda perl-hash-merge
conda install -c bioconda perl-list-util
conda install -c bioconda perl-module-load-conditional
conda install -c bioconda perl-posix
conda install -c bioconda perl-file-homedir
conda install -c bioconda perl-parallel-forkmanager
conda install -c bioconda perl-scalar-util-numeric
conda install -c bioconda perl-yaml
conda install -c bioconda perl-class-data-inheritable
conda install -c bioconda perl-exception-class
conda install -c bioconda perl-test-pod
conda install -c bioconda perl-file-which # skip if you are not comparing to reference annotation
conda install -c bioconda perl-mce
conda install -c bioconda perl-threaded
conda install -c bioconda perl-list-util
conda install -c bioconda perl-math-utils
conda install -c bioconda cdbtools
conda install -c eumetsat perl-yaml-xs
conda install -c bioconda perl-data-dumper
Subsequently install BRAKER and other software "as usual" while being in your conda environment. Note: There is a bioconda braker package, and a bioconda augustus package. They work. But they are usually lagging behind the development code of both tools on github. We therefore recommend manual installation and usage of lastest sources.
BRAKER is a collection of Perl and Python scripts and a Perl module. The main script that will be called in order to run BRAKER is braker.pl. Additional Perl and Python components are:
-
align2hints.pl -
filterGenemark.pl -
filterIntronsFindStrand.pl -
startAlign.pl -
helpMod_braker.pm -
findGenesInIntrons.pl -
downsample_traingenes.pl -
ensure_n_training_genes.py -
get_gc_content.py -
get_etp_hints.py
All scripts (files ending with *.pl and *.py) that are part of BRAKER must be executable in order to run BRAKER. This should already be the case if you download BRAKER from GitHub. Executability may be overwritten if you e.g. transfer BRAKER on a USB-stick to another computer. In order to check whether required files are executable, run the following command in the directory that contains BRAKER Perl scripts:
ls -l *.pl *.py
The output should be similar to this:
-rwxr-xr-x 1 katharina katharina 18191 Mai 7 10:25 align2hints.pl
-rwxr-xr-x 1 katharina katharina 6090 Feb 19 09:35 braker_cleanup.pl
-rwxr-xr-x 1 katharina katharina 408782 Aug 17 18:24 braker.pl
-rwxr-xr-x 1 katharina katharina 5024 Mai 7 10:25 downsample_traingenes.pl
-rwxr-xr-x 1 katharina katharina 5024 Mai 7 10:23 ensure_n_training_genes.py
-rwxr-xr-x 1 katharina katharina 4542 Apr 3 2019 filter_augustus_gff.pl
-rwxr-xr-x 1 katharina katharina 30453 Mai 7 10:25 filterGenemark.pl
-rwxr-xr-x 1 katharina katharina 5754 Mai 7 10:25 filterIntronsFindStrand.pl
-rwxr-xr-x 1 katharina katharina 7765 Mai 7 10:25 findGenesInIntrons.pl
-rwxr-xr-x 1 katharina katharina 1664 Feb 12 2019 gatech_pmp2hints.pl
-rwxr-xr-x 1 katharina katharina 2250 Jan 9 13:55 log_reg_prothints.pl
-rwxr-xr-x 1 katharina katharina 4679 Jan 9 13:55 merge_transcript_sets.pl
-rwxr-xr-x 1 katharina katharina 41674 Mai 7 10:25 startAlign.pl
It is important that the x in -rwxr-xr-x is present for each script. If that is not the case, run
`chmod a+x *.pl *.py`
in order to change file attributes.
You may find it helpful to add the directory in which BRAKER perl scripts reside to your $PATH environment variable. For a single bash session, enter:
PATH=/your_path_to_braker/:$PATH
export PATH
To make this $PATH modification available to all bash sessions, add the above lines to a startup script (e.g.~/.bashrc).
BRAKER calls upon various bioinformatics software tools that are not part of BRAKER. Some tools are obligatory, i.e. BRAKER will not run at all if these tools are not present on your system. Other tools are optional. Please install all tools that are required for running BRAKER in the mode of your choice.
Download GeneMark-ETPF1 from https://github.com/gatech-genemark/GeneMark-ETP or https://topaz.gatech.edu/GeneMark/etp.for_braker.tar.gz. Unpack and install GeneMark-ETP as described in GeneMark-ETP’s README file.
If already contained in your $PATH variable, BRAKER will guess the location of gmes_petap.pl or gmetp.pl automatically. Otherwise, BRAKER can find GeneMark-ES/ET/EP/ETP executables either by locating them in an environment variable GENEMARK_PATH, or by taking a command line argument (--GENEMARK_PATH=/your_path_to_GeneMark_executables/).
In order to set the environment variable for your current Bash session, type:
export GENEMARK_PATH=/your_path_to_GeneMark_executables/
Add the above lines to a startup script (e.g. ~/.bashrc) in order to make it available to all bash sessions.
Perl scripts within GeneMark-ES/ET/EP/ETP are configured with default Perl location at /usr/bin/perl.
If you are running GeneMark-ES/ET/EP/ETP in an Anaconda environment (or want to use Perl from the $PATH variable for any other reason), modify the shebang of all GeneMark-ES/ET/EP/ETP scripts with the following command located inside GeneMark-ES/ET/EP/ETP folder:
perl change_path_in_perl_scripts.pl "/usr/bin/env perl"
You can check whether GeneMark-ES/ET/EP is installed properly by running the check_install.bash and/or executing examples in GeneMark-E-tests directory.
GeneMark-ETP is downward compatible, i.e. it covers the functionality of GeneMark-EP and GeneMark-ET in BRAKER, too.
Download AUGUSTUS from its master branch at https://github.com/Gaius-Augustus/Augustus. Unpack AUGUSTUS and install AUGUSTUS according to AUGUSTUS README.TXT. Do not use outdated AUGUSTUS versions from other sources, e.g. Debian package or Bioconda package! BRAKER highly depends in particular on an up-to-date Augustus/scripts directory, and other sources are often lagging behind.
You should compile AUGUSTUS on your own system in order to avoid problems with versions of libraries used by AUGUSTUS. Compilation instructions are provided in the AUGUSTUS README.TXT file (Augustus/README.txt).
AUGUSTUS consists of augustus, the gene prediction tool, additional C++ tools located in Augustus/auxprogs and Perl scripts located in Augustus/scripts. Perl scripts must be executable (see instructions in section BRAKER components.
The C++ tool bam2hints is an essential component of BRAKER when run with RNA-Seq. Sources are located in Augustus/auxprogs/bam2hints. Make sure that you compile bam2hints on your system (it should be automatically compiled when AUGUSTUS is compiled, but in case of problems with bam2hints, please read troubleshooting instructions in Augustus/auxprogs/bam2hints/README).
Since BRAKER is a pipeline that trains AUGUSTUS, i.e. writes species specific parameter files, BRAKER needs writing access to the configuration directory of AUGUSTUS that contains such files (Augustus/config/). If you install AUGUSTUS globally on your system, the config folder will typically not be writable by all users. Either make the directory where config resides recursively writable to users of AUGUSTUS, or copy the config/ folder (recursively) to a location where users have writing permission.
AUGUSTUS will locate the config folder by looking for an environment variable $AUGUSTUS_CONFIG_PATH. If the $AUGUSTUS_CONFIG_PATH
environment variable is not set, then BRAKER will look in the path ../config relative to the directory in which it finds an AUGUSTUS
executable. Alternatively, you can supply the variable as a command line argument to BRAKER (--AUGUSTUS_CONFIG_PATH=/your_path_to_AUGUSTUS/Augustus/config/). We recommend that you export the variable e.g. for your current bash
session:
export AUGUSTUS_CONFIG_PATH=/your_path_to_AUGUSTUS/Augustus/config/
In order to make the variable available to all Bash sessions, add the above line to a startup script, e.g. ~/.bashrc.
Please have a look at the Dockerfile in case you want to install AUGUSTUS as Debian package. A number of scripts needs to be patched, then.
BRAKER expects the entire config directory of AUGUSTUS at $AUGUSTUS_CONFIG_PATH, i.e. the subfolders species with its contents (at least generic) and extrinsic! Providing a writable but empty folder at $AUGUSTUS_CONFIG_PATH will not work for BRAKER. If you need
to separate augustus binary and $AUGUSTUS_CONFIG_PATH, we recommend that you recursively copy the un-writable config contents to a writable location.
If you have a system-wide installation of AUGUSTUS at /usr/bin/augustus, an unwritable copy of config sits at /usr/bin/augustus_config/. The folder /home/yours/ is writable to you. Copy with the following command (and additionally set the then required variables):
cp -r /usr/bin/Augustus/config/ /home/yours/
export AUGUSTUS_CONFIG_PATH=/home/yours/augustus_config
export AUGUSTUS_BIN_PATH=/usr/bin
export AUGUSTUS_SCRIPTS_PATH=/usr/bin/augustus_scripts
Adding directories of AUGUSTUS binaries and scripts to your $PATH variable enables your system to locate these tools,
automatically. It is not a requirement for running BRAKER to do this, because BRAKER will try to guess them from the location of another
environment variable ($AUGUSTUS_CONFIG_PATH), or both directories can be supplied as command line arguments to braker.pl, but we recommend to add them to your $PATH variable. For your current bash session, type:
PATH=:/your_path_to_augustus/bin/:/your_path_to_augustus/scripts/:$PATH
export PATH
For all your BASH sessions, add the above lines to a startup script (e.g.~/.bashrc).
On Ubuntu, Python3 is usually installed by default, python3 will be in your $PATH variable, by default, and BRAKER will automatically locate it. However, you have the option to specify the python3 binary location in two other ways:
-
Export an environment variable
$PYTHON3_PATH, e.g. in your~/.bashrcfile:export PYTHON3_PATH=/path/to/python3/ -
Specify the command line option
--PYTHON3_PATH=/path/to/python3/tobraker.pl.
Download BAMTOOLS (e.g. git clone https://github.com/pezmaster31/bamtools.git). Install
BAMTOOLS by typing the following in your shell:
cd your-bamtools-directory mkdir build cd build cmake .. make
If already in your $PATH variable, BRAKER will find bamtools, automatically. Otherwise, BRAKER can locate the bamtools binary either
by using an environment variable $BAMTOOLS_PATH, or by taking a command line argument (--BAMTOOLS_PATH=/your_path_to_bamtools/bin/F6). In order to set the environment variable e.g. for your current bash session, type:
export BAMTOOLS_PATH=/your_path_to_bamtools/bin/
Add the above line to a startup script (e.g. ~/.bashrc) in order to set the environment variable for all bash sessions.
You can use either NCBI BLAST+ or DIAMOND for removal of redundant training genes. You do not need both tools. If DIAMOND is present, it will be preferred because it is much faster.
Obtain and unpack DIAMOND as follows:
wget https://github.com/bbuchfink/diamond/releases/download/v0.9.24/diamond-linux64.tar.gz
tar xzf diamond-linux64.tar.gz
If already in your $PATH variable, BRAKER will find diamond, automatically. Otherwise, BRAKER can locate the diamond binary either by using an environment variable $DIAMOND_PATH, or by taking a command line argument (--DIAMOND_PATH=/your_path_to_diamond). In order to set the environment variable e.g. for your current bash session, type:
export DIAMOND_PATH=/your_path_to_diamond/
Add the above line to a startup script (e.g. ~/.bashrc) in order to set the environment variable for all bash sessions.
If you decide for BLAST+, install NCBI BLAST+ with sudo apt-get install ncbi-blast+.
If already in your $PATH variable, BRAKER will find blastp, automatically. Otherwise, BRAKER can locate the blastp binary either by using an environment variable $BLAST_PATH, or by taking a command line argument (--BLAST_PATH=/your_path_to_blast/). In order to set the environment variable e.g. for your current bash session, type:
export BLAST_PATH=/your_path_to_blast/
Add the above line to a startup script (e.g. ~/.bashrc) in order to set the environment variable for all bash sessions.
Following tools are required by GeneMark-ETP and it will try to locate them in your $PATH variable. So make sure to add their location to your $PATH, e.g.:
export PATH=$PATH:/your/path/to/Tool
For all tools below, add the above line to a startup script (e.g. ~/.bashrc) in order to extend your $PATH variable for all bash sessions.
These software tools are only mandatory if you run BRAKER with RNA-Seq and protein data!
StringTie2 is used by GeneMark-ETP to assemble aligned RNA-Seq alignments. A precompiled version of StringTie2 can be downloaded from https://ccb.jhu.edu/software/stringtie/#install.
The software package bedtools is required by GeneMark-ETP if you want to run BRAKER with both RNA-Seq and protein data. You can download bedtools from https://github.com/arq5x/bedtools2/releases. Here, you can either download a precompiled version bedtools.static.binary, e.g.
wget https://github.com/arq5x/bedtools2/releases/download/v2.30.0/bedtools.static.binary
mv bedtools.static.binary bedtools
chmod a+x
or you can download bedtools-2.30.0.tar.gz and compile it from source using make, e.g.
wget https://github.com/arq5x/bedtools2/releases/download/v2.30.0/bedtools-2.30.0.tar.gz
tar -zxvf bedtools-2.30.0.tar.gz
cd bedtools2
make
See https://bedtools.readthedocs.io/en/latest/content/installation.html for more information.
GffRead is a utility software required by GeneMark-ETP. It can be downloaded from https://github.com/gpertea/gffread/releases/download/v0.12.7/gffread-0.12.7.Linux_x86_64.tar.gz and installed with make, e.g.
wget https://github.com/gpertea/gffread/releases/download/v0.12.7/gffread-0.12.7.Linux_x86_64.tar.gz
tar xzf gffread-0.12.7.Linux_x86_64.tar.gz
cd gffread-0.12.7.Linux_x86_64
make
Samtools is not required for running BRAKER without GeneMark-ETP if all your files are formatted, correctly (i.e. all sequences should have short and unique fasta names). If you are not sure whether all your files are fomatted correctly, it might be helpful to have Samtools installed because BRAKER can automatically fix certain format issues by using Samtools.
As a prerequisite for Samtools, download and install htslib (e.g. git clone https://github.com/samtools/htslib.git, follow the htslib documentation for installation).
Download and install Samtools (e.g. git clone git:https://github.com/samtools/samtools.git), subsequently follow Samtools documentation for installation).
If already in your $PATH variable, BRAKER will find samtools, automatically. Otherwise, BRAKER can find Samtools either by taking a
command line argument (--SAMTOOLS_PATH=/your_path_to_samtools/), or by using an environment variable $SAMTOOLS_PATH. For exporting the variable, e.g. for your current bash session, type:
export SAMTOOLS_PATH=/your_path_to_samtools/
Add the above line to a startup script (e.g. ~/.bashrc) in order to set the environment variable for all bash sessions.
If Biopython is installed, BRAKER can generate FASTA-files with coding sequences and protein sequences predicted by AUGUSTUS and generate track data hubs for visualization of a BRAKER run with MakeHub R16.
These are optional steps. The first can be disabled with the command-line flag --skipGetAnnoFromFasta, the second can be activated by using the command-line options --makehub [email protected], Biopython is not required if neither of these optional steps shall be performed.
On Ubuntu, install Python3 package manager with:
`sudo apt-get install python3-pip`
Then, install Biopython with:
`sudo pip3 install biopython`
cdbfasta and cdbyank are required by BRAKER for correcting AUGUSTUS genes with in frame stop codons (spliced stop codons) using the AUGUSTUS script fix_in_frame_stop_codon_genes.py. This can be skipped with --skip_fixing_broken_genes.
On Ubuntu, install cdbfasta with:
sudo apt-get install cdbfasta
For other systems, you can for example obtain cdbfasta from https://github.com/gpertea/cdbfasta, e.g.:
git clone https://github.com/gpertea/cdbfasta.git
cd cdbfasta
make all
On Ubuntu, cdbfasta and cdbyank will be in your $PATH variable after installation, and BRAKER will automatically locate them. However, you have the option to specify the cdbfasta and cdbyank binary location in two other ways:
- Export an environment variable
$CDBTOOLS_PATH, e.g. in your~/.bashrcfile:
export CDBTOOLS_PATH=/path/to/cdbtools/
- Specify the command line option
--CDBTOOLS_PATH=/path/to/cdbtools/tobraker.pl.
Note: Support of stand-alone Spaln (ouside of ProtHint) within BRAKER is deprecated.
This tool is required if you run ProtHint or if you would like to run protein to genome alignments with BRAKER using Spaln outside of ProtHint. Using Spaln outside of ProtHint is a suitable approach only if an annotated species of short evolutionary distance to your target genome is available. We recommend running Spaln through ProtHint for BRAKER. ProtHint brings along a Spaln binary. If that does not work on your system, download Spaln from https://github.com/ogotoh/spaln. Unpack and install according to spaln/doc/SpalnReadMe22.pdf.
BRAKER will try to locate the Spaln executable by using an environment variable $ALIGNMENT_TOOL_PATH. Alternatively, this can be supplied as command line argument (--ALIGNMENT_TOOL_PATH=/your/path/to/spaln).
This tool is only required if you want either add UTRs (from RNA-Seq data) to predicted genes or if you want to train UTR parameters for AUGUSTUS and predict genes with UTRs. In any case, GUSHR requires the input of RNA-Seq data.
GUSHR is available for download at https://github.com/Gaius-Augustus/GUSHR. Obtain it by typing:
git clone https://github.com/Gaius-Augustus/GUSHR.git
GUSHR executes a GeMoMa jar file R19, R20, R21, and this jar file requires Java 1.8. On Ubuntu, you can install Java 1.8 with the following command:
sudo apt-get install openjdk-8-jdk
If you have several java versions installed on your system, make sure that you enable 1.8 prior running BRAKER with java by running
sudo update-alternatives --config java
and selecting the correct version.
If you switch --UTR=on, bamToWig.py will require the following tools that can be downloaded from https://hgdownload.soe.ucsc.edu/admin/exe:
-
twoBitInfo
-
faToTwoBit
It is optional to install these tools into your $PATH. If you don't, and you switch --UTR=on, bamToWig.py will automatically download them into the working directory.
If you wish to automaticaly generate a track data hub of your BRAKER run, the MakeHub software, available at https://github.com/Gaius-Augustus/MakeHub is required. Download the software (either by running git clone https://github.com/Gaius-Augustus/MakeHub.git, or by
picking a release from https://github.com/Gaius-Augustus/MakeHub/releases. Extract the release package if you downloaded a release (e.g. unzip MakeHub.zip or tar -zxvf MakeHub.tar.gz.
BRAKER will try to locate the make_hub.py script by using an environment variable $MAKEHUB_PATH. Alternatively, this can be supplied as command line argument (--MAKEHUB_PATH=/your/path/to/MakeHub/). BRAKER can also try to guess the location of MakeHub on your system.
If you want BRAKER to download RNA-Seq libraries from NCBI's SRA, the SRA Toolkit is required. You can get a precompiled version of the SRA Toolkit from https://daehwankimlab.github.io/hisat2/download/#version-hisat2-221.
BRAKER will try to find executable binaries from the SRA Toolkit (fastq-dump, prefetch) by using an environment variable $SRATOOLS_PATH. Alternatively, this can be supplied as command line argument (--SRATOOLS_PATH=/your/path/to/SRAToolkit/). BRAKER can also try to guess the location of the SRA Toolkit on your system if the executables are in your $PATH variable.
If you want to use unaligned RNA-Seq reads, the HISAT2 software is required to map them to the genome. A precompiled version of HISAT2 can be downloaded from https://daehwankimlab.github.io/hisat2/download/#version-hisat2-221.
BRAKER will try to find executable HISAT2 binaries (hisat2, hisat2-build) by using an environment variable $HISAT2_PATH. Alternatively, this can be supplied as command line argument (--HISAT2_PATH=/your/path/to/HISAT2/). BRAKER can also try to guess the location of HISAT2 on your system if the executables are in your $PATH variable.
If you want to run TSEBRA within BRAKER in a BUSCO completeness maximizing mode, you need to install compleasm.
wget https://github.com/huangnengCSU/compleasm/releases/download/v0.2.4/compleasm-0.2.4_x64-linux.tar.bz2
tar -xvjf compleasm-0.2.4_x64-linux.tar.bz2 && \
Add the resulting folder compleasm_kit to your $PATH variable, e.g.:
export PATH=$PATH:/your/path/to/compleasm_kit
Compleasm requires pandas, which can be installed with:
pip install pandas
BRAKER (braker.pl) uses getconf to see how many threads can be run on your system. On Ubuntu, you can install it with:
sudo apt-get install libc-bin
In the following, we describe “typical” BRAKER calls for different input data types. In general, we recommend that you run BRAKER on genomic sequences that have been softmasked for Repeats. BRAKER should only be applied to genomes that have been softmasked for repeats!
This approach is suitable for genomes of species for which RNA-Seq libraries with good transcriptome coverage are available and for which protein data is not at hand. The pipeline is illustrated in Figure 2.
BRAKER has several ways to receive RNA-Seq data as input:
-
You can provide ID(s) of RNA-Seq libraries from SRA (in case of multiple IDs, separate them by comma) as argument to
--rnaseq_sets_ids. The libraries belonging to the IDs are then downloaded automatically by BRAKER, e.g.:braker.pl --species=yourSpecies --genome=genome.fasta \ --rnaseq_sets_ids=SRA_ID1,SRA_ID2 -
You can use local
FASTQfile(s) of unaligned reads as input. In this case, you have to provide BRAKER with the ID(s) of the RNA-Seq set(s) as argument to--rnaseq_sets_idsand the path(s) to the directories, where theFASTQfiles are located as argument to--rnaseq_sets_dirs. For each IDID, BRAKER will search in these directories for oneFASTQfile namedID.fastqif the reads are unpaired, or for twoFASTQfiles namedID_1.fastqandID_2.fastqif they are paired.For example, if you have a paired library called 'SRA_ID1' and an unpaired library named 'SRA_ID2', you have to have a directory
/path/to/local/fastq/files/, where the filesSRA_ID1_1.fastq,SRA_ID1_2.fastq, andSRA_ID2.fastqreside. Then, you could run BRAKER with following command:braker.pl --species=yourSpecies --genome=genome.fasta \ --rnaseq_sets_ids=SRA_ID1,SRA_ID2 \ --rnaseq_sets_dirs=/path/to/local/fastq/files/ -
There are two ways of supplying BRAKER with RNA-Seq data as
bamfile(s). First, you can do it in the same way as you would supplyFASTQfile(s): Provide the ID(s)/name(s) of yourbamfile(s) as argument to--rnaseq_sets_idsand specify directories where thebamfiles reside with--rnaseq_sets_dirs. BRAKER will automatically detect that these ID(s) arebamand notFASTQfile(s), e.g.:braker.pl --species=yourSpecies --genome=genome.fasta \ --rnaseq_sets_ids=BAM_ID1,BAM_ID2 \ --rnaseq_sets_dirs=/path/to/local/bam/files/Second, you can specify the paths to your
bamfile(s) directly, e.g. can either extract RNA-Seq spliced alignment information frombamfiles, or it can use such extracted information, directly.braker.pl --species=yourSpecies --genome=genome.fasta \ --bam=file1.bam,file2.bamPlease note that we generally assume that bam files were generated with HiSat2 because that is the aligner that would also be executed by BRAKER3 with fastq input. If you want for some reason to generate the bam files with STAR, use the option
--outSAMstrandField intronMotifof STAR to produce files that are compatible wiht StringTie in BRAKER3. -
In order to run BRAKER with RNA-Seq spliced alignment information that has already been extracted, run:
braker.pl --species=yourSpecies --genome=genome.fasta \ --hints=hints1.gff,hints2.gffThe format of such a hints file must be as follows (tabulator separated file):
chrName b2h intron 6591 8003 1 + . pri=4;src=E chrName b2h intron 6136 9084 11 + . mult=11;pri=4;src=E ...The source
b2hin the second column and the source tagsrc=Ein the last column are essential for BRAKER to determine whether a hint has been generated from RNA-Seq data.
It is also possible to provide RNA-Seq sets in different ways for the same BRAKER run, any combination of above options is possible. It is not recommended to provide RNA-Seq data with --hints if you run BRAKER in ETPmode (RNA-Seq and protein data), because GeneMark-ETP won't use these hints!
This approach is suitable for genomes of species for which no RNA-Seq libraries are available. A large database of proteins (with possibly longer evolutionary distance to the target species) should be used in this case. This mode is illustrated in figure 9.
Figure 9: BRAKER with proteins of any evolutionary distance. ProtHint protein mapping pipelines is used to generate protein hints. ProtHint automatically determines which alignments are from close relatives, and which are from rather distant relatives.
For running BRAKER in this mode, type:
braker.pl --genome=genome.fa --prot_seq=proteins.fa
We recommend using OrthoDB as basis for proteins.fa. The instructions on how to prepare the input OrthoDB proteins are documented here: https://github.com/gatech-genemark/ProtHint#protein-database-preparation.
You can of course add additional protein sequences to that file, or try with a completely different database. Any database will need several representatives for each protein, though.
Instead of having BRAKER run ProtHint, you can also start BRAKER with hints already produced by ProtHint, by providing ProtHint's prothint_augustus.gff output:
braker.pl --genome=genome.fa --hints=prothint_augustus.gff
The format of prothint_augustus.gff in this mode looks like this:
2R ProtHint intron 11506230 11506648 4 + . src=M;mult=4;pri=4
2R ProtHint intron 9563406 9563473 1 + . grp=69004_0:001de1_702_g;src=C;pri=4;
2R ProtHint intron 8446312 8446371 1 + . grp=43151_0:001cae_473_g;src=C;pri=4;
2R ProtHint intron 8011796 8011865 2 - . src=P;mult=1;pri=4;al_score=0.12;
2R ProtHint start 234524 234526 1 + . src=P;mult=1;pri=4;al_score=0.08;
The prediction of all hints with src=M will be enforced. Hints with src=C are 'chained evidence', i.e. they will only be incorporated if all members of the group (grp=...) can be incorporated in a single transcript. All other hints have src=P in the last column. Supported features in column 3 are intron, start, stop and CDSpart.
If RNA-Seq (and only RNA-Seq) data is provided to BRAKER as a bam-file, and if the genome is softmasked for repeats, BRAKER can automatically train UTR parameters for AUGUSTUS. After successful training of UTR parameters, BRAKER will automatically predict genes including coverage information form RNA-Seq data. Example call:
braker.pl --species=yourSpecies --genome=genome.fasta \
--bam=file.bam --UTR=on
Warnings:
-
This feature is experimental!
-
--UTR=on is currently not compatible with bamToWig.py as released in AUGUSTUS 3.3.3; it requires the current development code version from the github repository (git clone https://github.com/Gaius-Augustus/Augustus.git).
-
--UTR=on increases memory consumption of AUGUSTUS. Carefully monitor jobs if your machine was close to maxing RAM without --UTR=on! Reducing the number of cores will also reduce RAM consumption.
-
UTR prediction sometimes improves coding sequence prediction accuracy, but not always. If you try this feature, carefully compare results with and without UTR parameters, afterwards (e.g. in UCSC Genome Browser).
For running BRAKER without UTR parameters, it is not very important whether RNA-Seq data was generated by a stranded protocol (because spliced alignments are ’artificially stranded’ by checking the splice site pattern). However, for UTR training and prediction, stranded libraries may provide information that is valuable for BRAKER.
After alignment of the stranded RNA-Seq libraries, separate the resulting bam file entries into two files: one for plus strand mappings, one for minus strand mappings. Call BRAKER as follows:
braker.pl --species=yourSpecies --genome=genome.fasta \
--bam=plus.bam,minus.bam --stranded=+,- \
--UTR=on
You may additionally include bam files from unstranded libraries. Those files will not used for generating UTR training examples, but they will be included in the final gene prediction step as unstranded coverage information, example call:
braker.pl --species=yourSpecies --genome=genome.fasta \
--bam=plus.bam,minus.bam,unstranded.bam \
--stranded=+,-,. --UTR=on
Warning: This feature is experimental and currently has low priority on our maintenance list!
The native mode for running BRAKER with RNA-Seq and protein data. This will call GeneMark-ETP, which will use RNA-Seq and protein hints for training GeneMark-ETP. Subsequently, AUGUSTUS is trained on 'high-confindent' genes (genes with very high extrinsic evidence support) from the GeneMark-ETP prediction and a set of genes is predicted by AUGUSTUS. In a last step, the predictions of AUGUSTUS and GeneMark-ETP are combined using TSEBRA.
Alignment of RNA-Seq reads
GeneMark-ETP utilizes Stringtie2 to assemble RNA-Seq data, which requires that the aligned reads (BAM files) contain the XS (strand) tag for spliced reads. Therefore, if you align your reads with HISAT2, you must enable the --dta option, or if you use STAR, you must use the --outSAMstrandField intronMotif option. TopHat alignments include this tag by default.
To call the pipeline in this mode, you have to provide it with a protein database using --prot_seq (as described in BRAKER with protein data), and RNA-Seq data either by their SRA ID so that they are downloaded by BRAKER, as unaligned reads in FASTQ format, and/or as aligned reads in bam format (as described in BRAKER with RNA-Seq data). You could also specify already processed extrinsic evidence using the --hints option. However, this is not recommend for a normal BRAKER run in ETPmode, as these hints won't be used in the GeneMark-ETP step. Only use --hints when you want to skip the GenMark-ETP step!
Examples of how you could run BRAKER in ETPmode:
braker.pl --genome=genome.fa --prot_seq=orthodb.fa \
--rnaseq_sets_ids=SRA_ID1,SRA_ID2 \
--rnaseq_sets_dirs=/path/to/local/RNA-Seq/files/
braker.pl --genome=genome.fa --prot_seq=orthodb.fa \
--rnaseq_sets_ids=SRA_ID1,SRA_ID2,SRA_ID3
braker.pl --genome=genome.fa --prot_seq=orthodb.fa \
--bam=/path/to/SRA_ID1.bam,/path/to/SRA_ID2.bam
A preliminary protocol for integration of assembled subreads from PacBio ccs sequencing in combination with short read Illumina RNA-Seq and protein database is described at https://github.com/Gaius-Augustus/BRAKER/blob/master/docs/long_reads/long_read_protocol.md
We forked GeneMark-ETP and hard coded that StringTie will perform long read assembly in that particular version. If you want to use this 'fast-hack' version for BRAKER, you have to prepare the BAM file with long read to genome spliced alignments outside of BRAKER, e.g.:
T=48 # adapt to your number of threads
minimap2 -t${T} -ax splice:hq -uf genome.fa isoseq.fa > isoseq.sam
samtools view -bS --threads ${T} isoseq.sam -o isoseq.bam
Pull the adapted container:
singularity build braker3_lr.sif docker:https://teambraker/braker3:isoseq
Calling BRAKER3 with a BAM file of spliced-aligned IsoSeq Reads:
singularity exec -B ${PWD}:${PWD} braker3_lr.sif braker.pl --genome=genome.fa --prot_seq=protein_db.fa –-bam=isoseq.bam --threads=${T}
Warning Do NOT mix short read and long read data in this BRAKER/GeneMark-ETP variant!
Warning The accuracy of gene prediction here heavily depends on the depth of your isoseq data. We verified with PacBio HiFi reads from 2022 that given sufficient completeness of the assembled transcriptome you will reach similar results as with short reads. However, we also observed a drop in accuracy compared to short reads when using other long read data sets with higher error rates and less sequencing depth.
Please run braker.pl --help to obtain a full list of options.
Compute AUGUSTUS ab initio predictions in addition to AUGUSTUS predictions with hints (additional output files: augustus.ab_initio.*. This may be useful for estimating the quality of training gene parameters when inspecting predictions in a Browser.
One or several command line arguments to be passed to AUGUSTUS, if several arguments are given, separate them by whitespace, i.e. "--first_arg=sth --second_arg=sth". This may be be useful if you know that gene prediction in your particular species benefits from a particular AUGUSTUS argument during the prediction step.
Specifies the maximum number of threads that can be used during computation. BRAKER has to run some steps on a single thread, others can take advantage of multiple threads. If you use more than 8 threads, this will not speed up all parallelized steps, in particular, the time consuming optimize_augustus.pl will not use more than 8 threads. However, if you don’t mind some threads being idle, using more than 8 threads will speed up other steps.
GeneMark-ETP option: run algorithm with branch point model. Use this option if you genome is a fungus.
Use the present config and parameter files if they exist for 'species'; will overwrite original parameters if BRAKER performs an AUGUSTUS training.
Execute CRF training for AUGUSTUS; resulting parameters are only kept for final predictions if they show higher accuracy than HMM parameters. This increases runtime!
Change the parameter
Generate UTR training examples for AUGUSTUS from RNA-Seq coverage information, train AUGUSTUS UTR parameters and predict genes with AUGUSTUS and UTRs, including coverage information for RNA-Seq as evidence. This is an experimental feature!
If you performed a BRAKER run without --UTR=on, you can add UTR parameter training and gene prediction with UTR parameters (and only RNA-Seq hints) with the following command:
braker.pl --genome=../genome.fa --addUTR=on \
--bam=../RNAseq.bam --workingdir=$wd \
--AUGUSTUS_hints_preds=augustus.hints.gtf \
--threads=8 --skipAllTraining --species=somespecies
Modify augustus.hints.gtf to point to the AUGUSTUS predictions with hints from previous BRAKER run; modify flaning_DNA value to the flanking region from the log file of your previous BRAKER run; modify some_new_working_directory to the location where BRAKER should store results of the additional BRAKER run; modify somespecies to the species name used in your previous BRAKER run.
Add UTRs from RNA-Seq converage information to AUGUSTUS gene predictions using GUSHR. No training of UTR parameters and no gene prediction with UTR parameters is performed.
If you performed a BRAKER run without --addUTR=on, you can add UTRs results of a previous BRAKER run with the following command:
braker.pl --genome=../genome.fa --addUTR=on \
--bam=../RNAseq.bam --workingdir=$wd \
--AUGUSTUS_hints_preds=augustus.hints.gtf --threads=8 \
--skipAllTraining --species=somespecies
Modify augustus.hints.gtf to point to the AUGUSTUS predictions with hints from previous BRAKER run; modify some_new_working_directory to the location where BRAKER should store results of the additional BRAKER run; this run will not modify AUGUSTUS parameters. We recommend that you specify the original species of the original run with --species=somespecies. Otherwise, BRAKER will create an unneeded species parameters directory Sp_*.
If --UTR=on is enabled, strand-separated bam-files can be provided with --bam=plus.bam,minus.bam. In that case, --stranded=... should hold the strands of the bam files (+ for plus strand, - for minus strand, . for unstranded). Note that unstranded data will be used in the gene prediction step, only, if the parameter --stranded=... is set. This is an experimental feature! GUSHR currently does not take advantage of stranded data.
--makehub --email=[email protected]
If --makehub and [email protected] (with your valid e-mail adress) are provided, a track data hub for visualizing results with the UCSC Genome Browser will be generated using MakeHub (https://github.com/Gaius-Augustus/MakeHub).
By default, GeneMark-ES/ET/EP/ETP uses a probability of 0.001 for predicting the donor splice site pattern GC (instead of GT). It may make sense to increase this value for species where this donor splice site is more common. For example, in the species Emiliania huxleyi, about 50% of donor splice sites have the pattern GC (https://media.nature.com/original/nature-assets/nature/journal/v499/n7457/extref/nature12221-s2.pdf, page 5).
Use a species-specific lineage, e.g. arthropoda_odb10 for an arthropod. BRAKER does not support auto-typing of the lineage.
Specifying a BUSCO-lineage invokes two changes in BRAKER R28:
-
BRAKER will run compleasm with the specified lineage in genome mode and convert the detected BUSCO matches into hints for AUGUSTUS. This may increase the number of BUSCOs in the augustus.hints.gtf file slightly.
-
BRAKER will invoke best_by_compleasm.py to check whether the braker.gtf file that is by default generated by TSEBRA has the lowest amount of missing BUSCOs compared to the augustus.hints.gtf and the genemark.gtf file. If not, the following decision schema is applied to re-run TSEBRA to minimize the missing BUSCOs in the final output of BRAKER (always braker.gtf). If an alternative and better gene set is created, the original braker.gtf gene set is moved to a directory called braker_original. Information on what happened during the best_by_compleasm.py run is written to the file best_by_compleasm.log.
Please note that using BUSCO to assess the quality of a gene set, in particular when comparing BRAKER to other pipelines, does not make sense once you specified a BUSCO lineage. We recommend that you use other measures to assess the quality of your gene set, e.g. by comparing it to a reference gene set or running OMArk.
BRAKER produces several important output files in the working directory.
-
braker.gtf: Final gene set of BRAKER. This file may contain different contents depending on how you called BRAKER
-
in ETPmode: Final gene set of BRAKER consisting of genes predicted by AUGUSTUS and GeneMark-ETP that were combined and filtered by TSEBRA.
-
otherwise: Union of augustus.hints.gtf and reliable GeneMark-ES/ET/EP predictions (genes fully supported by external evidence). In
--esmode, this is the union of augustus.ab_initio.gtf and all GeneMark-ES genes. Thus, this set is generally more sensitive (more genes correctly predicted) and can be less specific (more false-positive predictions can be present). This output is not necessarily better than augustus.hints.gtf, and it is not recommended to use it if BRAKER was run in ESmode.
-
-
braker.codingseq: Final gene set with coding sequences in FASTA format
-
braker.aa: Final gene set with protein sequences in FASTA format
-
braker.gff3: Final gene set in gff3 format (only produced if the flag
--gff3was specified to BRAKER. -
Augustus/*: Augustus gene set(s) in as gtf/conding/aa files
-
GeneMark-E*/genemark.gtf: Genes predicted by GeneMark-ES/ET/EP/EP+/ETP in GTF-format.
-
hintsfile.gff: The extrinsic evidence data extracted from RNAseq.bam and/or protein data.
-
braker_original/*: Genes predicted by BRAKER (TSEBRA merge) before compleasm was used to improve BUSCO completeness
-
bbc/*: output folder of best_by_compleasm.py script from TSEBRA that is used to improve BUSCO completeness in the final output of BRAKER
Output files may be present with the following name endings and formats:
-
Coding sequences in FASTA-format are produced if the flag
--skipGetAnnoFromFastawas not set. -
Protein sequence files in FASTA-format are produced if the flag
--skipGetAnnoFromFastawas not set.
For details about gtf format, see https://www.sanger.ac.uk/Software/formats/GFF/. A GTF-format file contains one line per predicted exon. Example:
HS04636 AUGUSTUS initial 966 1017 . + 0 transcript_id "g1.1"; gene_id "g1";
HS04636 AUGUSTUS internal 1818 1934 . + 2 transcript_id "g1.1"; gene_id "g1";
The columns (fields) contain:
seqname source feature start end score strand frame transcript ID and gene ID
If the --makehub option was used and MakeHub is available on your system, a hub directory beginning with the name hub_ will be created. Copy this directory to a publicly accessible web server. A file hub.txt resides in the directory. Provide the link to that file to the UCSC Genome Browser for visualizing results.
An incomplete example data set is contained in the directory BRAKER/example. In order to complete the data set, please download the RNA-Seq alignment file (134 MB) with wget:
cd BRAKER/example
wget https://topaz.gatech.edu/GeneMark/Braker/RNAseq.bam
In case you have trouble accessing that file, there's also a copy available from another server:
cd BRAKER/example
wget https://bioinf.uni-greifswald.de/augustus/datasets/RNAseq.bam
The example data set was not compiled in order to achieve optimal prediction accuracy, but in order to quickly test pipeline components. The small subset of the genome used in these test examples is not long enough for BRAKER training to work well.
Data corresponds to the last 1,000,000 nucleotides of Arabidopsis thaliana's chromosome Chr5, split into 8 artificial contigs.
RNA-Seq alignments were obtained by VARUS.
The protein sequences are a subset of OrthoDB v10 plants proteins.
List of files:
genome.fa- genome file in fasta formatRNAseq.bam- RNA-Seq alignment file in bam format (this file is not a part of this repository, it must be downloaded separately from https://topaz.gatech.edu/GeneMark/Braker/RNAseq.bam)RNAseq.hints- RNA-Seq hints (can be used instead of RNAseq.bam as RNA-Seq input to BRAKER)proteins.fa- protein sequences in fasta format
The below given commands assume that you configured all paths to tools by exporting bash variables or that you have the necessary tools in your $PATH.
The example data set also contains scripts tests/test*.sh that will execute below listed commands for testing BRAKER with the example data set. You find example results of AUGUSTUS and GeneMark-ES/ET/EP/ETP in the folder results/test*. Be aware that BRAKER contains several parts where random variables are used, i.e. results that you obtain when running the tests may not be exactly identical. To compare your test results with the reference ones, you can use the compare_intervals_exact.pl script as follows:
# Compare CDS features
compare_intervals_exact.pl --f1 augustus.hints.gtf --f2 ../../results/test${N}/augustus.hints.gtf --verbose
# Compare transcripts
compare_intervals_exact.pl --f1 augustus.hints.gtf --f2 ../../results/test${N}/augustus.hints.gtf --trans --verbose
Several tests use --gm_max_intergenic 10000 option to make the test runs faster. It is not recommended to use this option in real BRAKER runs, the speed increase achieved by adjusting this option is negligible on full-sized genomes.
We give runtime estimations derived from computing on Intel(R) Xeon(R) CPU E5530 @ 2.40GHz.
The following command will run the pipeline according to Figure 3:
braker.pl --genome genome.fa --bam RNAseq.bam --threads N --busco_lineage=lineage_odb10
This test is implemented in test1.sh, expected runtime is ~20 minutes.
The following command will run the pipeline according to Figure 4:
braker.pl --genome genome.fa --prot_seq proteins.fa --threads N --busco_lineage=lineage_odb10
This test is implemented in test2.sh, expected runtime is ~20 minutes.
The following command will run a pipeline that first trains GeneMark-ETP with protein and RNA-Seq hints and subsequently trains AUGUSTUS on the basis of GeneMark-ETP predictions. AUGUSTUS predictions are also performed with hints from both sources, see Figure 5.
Run with local RNA-Seq file:
braker.pl --genome genome.fa --prot_seq proteins.fa --bam ../RNAseq.bam --threads N --busco_lineage=lineage_odb10
This test is implemented in test3.sh, expected runtime is ~20 minutes.
Download RNA-Seq library from Sequence Read Archive (~1gb):
braker.pl --genome genome.fa --prot_seq proteins.fa --rnaseq_sets_ids ERR5767212 --threads N --busco_lineage=lineage_odb10
This test is implemented in test3_4.sh, expected runtime is ~35 minutes.
The training step of all pipelines can be skipped with the option --skipAllTraining. This means, only AUGUSTUS predictions will be performed, using pre-trained, already existing parameters. For example, you can predict genes with the command:
braker.pl --genome=genome.fa --bam RNAseq.bam --species=arabidopsis \
--skipAllTraining --threads N
This test is implemented in test4.sh, expected runtime is ~1 minute.
The following command will run the pipeline with no extrinsic evidence:
braker.pl --genome=genome.fa --esmode --threads N
This test is implemented in test5.sh, expected runtime is ~20 minutes.
The following command will run BRAKER with training UTR parameters from RNA-Seq coverage data:
braker.pl --genome genome.fa --bam RNAseq.bam --UTR=on --threads N
This test is implemented in test6.sh, expected runtime is ~20 minutes.
The following command will add UTRs to augustus.hints.gtf from RNA-Seq coverage data:
braker.pl --genome genome.fa --bam RNAseq.bam --addUTR=on --threads N
This test is implemented in test7.sh, expected runtime is ~20 minutes.
There is currently no clean way to restart a failed BRAKER run (after solving some problem). However, it is possible to start a new BRAKER run based on results from a previous run -- given that the old run produced the required intermediate results. We will in the following refer to the old working directory with variable ${BRAKER_OLD}, and to the new BRAKER working directory with ${BRAKER_NEW}. The file what-to-cite.txt will always only refer to the software that was actually called by a particular run. You might have to combine the contents of ${BRAKER_NEW}/what-to-cite.txt with ${BRAKER_OLD}/what-to-cite.txt for preparing a publication. The following figure illustrates at which points BRAKER run may be intercepted.
Figure 10: Points for intercepting a BRAKER run and reusing intermediate results in a new BRAKER run.
This option is only possible for BRAKER in ETmode or EPmode and not in ETPmode!
If you have access to an existing BRAKER output that contains hintsfiles that were generated from extrinsic data, such as RNA-Seq or protein sequences, you can recycle these hints files in a new BRAKER run. Also, hints from a separate ProtHint run can be directly used in BRAKER.
The hints can be given to BRAKER with --hints ${BRAKER_OLD}/hintsfile.gff option. This is illustrated in the test files test1_restart1.sh, test2_restart1.sh, test4_restart1.sh. The other modes (for which this test is missing) cannot be restarted in this way.
The GeneMark result can be given to BRAKER with --geneMarkGtf ${BRAKER_OLD}/GeneMark*/genemark.gtf option if BRAKER is run in ETmode or EPmode. This is illustrated in the test files test1_restart2.sh, test2_restart2.sh, test5_restart2.sh.
In ETPmode, you can either provide BRAKER with the results of the GeneMarkETP step manually, with --geneMarkGtf ${BRAKER_OLD}/GeneMark-ETP/proteins.fa/genemark.gtf, --traingenes ${BRAKER_OLD}/GeneMark-ETP/training.gtf, and --hints ${BRAKER_OLD}/hintsfile.gff (see test3_restart1.sh for an example), or you can specify the previous GeneMark-ETP results with the option --gmetp_results_dir ${BRAKER_OLD}/GeneMark-ETP/ so that BRAKER can search for the files automatically (see test3_restart2.sh for an example).
The trained species parameters for AGUSTUS can be passed with --skipAllTraining and --species $speciesName options. This is illustrated in test*_restart3.sh files. Note that in ETPmode you have to specify the GeneMark files as described in Option 2!
Before reporting bugs, please check that you are using the most recent versions of GeneMark-ES/ET/EP/ETP, AUGUSTUS and BRAKER. Also, check the list of Common problems, and the Issue list on GitHub before reporting bugs. We do monitor open issues on GitHub. Sometimes, we are unable to help you, immediately, but we try hard to solve your problems.
If you found a bug, please open an issue at https://github.com/Gaius-Augustus/BRAKER/issues (or contact [email protected] or [email protected]).
Information worth mentioning in your bug report:
Check in braker/yourSpecies/braker.log at which step braker.pl crashed.
There are a number of other files that might be of interest, depending on where in the pipeline the problem occurred. Some of the following files will not be present if they did not contain any errors.
-
braker/yourSpecies/errors/bam2hints.*.stderr- will give details on a bam2hints crash (step for converting bam file to intron gff file) -
braker/yourSpecies/hintsfile.gff- is this file empty? If yes, something went wrong during hints generation - does this file contain hints from source “b2h” and of type “intron”? If not: GeneMark-ET will not be able to execute properly. Conversely, GeneMark-EP+ will not be able to execute correctly if hints from the source "ProtHint" are missing. -
braker/yourSpecies/spaln/*err- errors reported by spaln -
braker/yourSpecies/errors/GeneMark-{ET,EP,ETP}.stderr- errors reported by GeneMark-ET/EP+/ETP -
braker/yourSpecies/errors/GeneMark-{ET,EP,ETP).stdout- may give clues about the point at which errors in GeneMark-ET/EP+/ETP occured -
braker/yourSpecies/GeneMark-{ET,EP,ETP}/genemark.gtf- is this file empty? If yes, something went wrong during executing GeneMark-ET/EP+/ETP -
braker/yourSpecies/GeneMark-{ET,EP}/genemark.f.good.gtf- is this file empty? If yes, something went wrong during filtering GeneMark-ET/EP+ genes for training AUGUSTUS -
braker/yourSpecies/genbank.good.gb- try a “grep -c LOCUS genbank.good.gb” to determine the number of training genes for training AUGUSTUS, should not be low -
braker/yourSpecies/errors/firstetraining.stderr- contains errors from first iteration of training AUGUSTUS -
braker/yourSpecies/errors/secondetraining.stderr- contains errors from second iteration of training AUGUSTUS -
braker/yourSpecies/errors/optimize_augustus.stderr- contains errors optimize_augustus.pl (additional training set for AUGUSTUS) -
braker/yourSpecies/errors/augustus*.stderr- contain AUGUSTUS execution errors -
braker/yourSpecies/startAlign.stderr- if you provided a protein fasta file, something went wrong during protein alignment -
braker/yourSpecies/startAlign.stdout- may give clues on at which point protein alignment went wrong
-
BRAKER complains that the RNA-Seq file does not correspond to the provided genome file, but I am sure the files correspond to each other!
Please check the headers of the genome FASTA file. If the headers are long and contain whitespaces, some RNA-Seq alignment tools will truncate sequence names in the BAM file. This leads to an error with BRAKER. Solution: shorten/simplify FASTA headers in the genome file before running the RNA-Seq alignment and BRAKER.
-
GeneMark fails!
(a) GeneMark by default only uses contigs longer than 50k for training. If you have a highly fragmented assembly, this might lead to "no data" for training. You can override the default minimal length by setting the BRAKER argument
--min_contig=10000.(b) see "[something] failed to execute" below.
-
[something] failed to execute!
When providing paths to software to BRAKER, please use absolute, non-abbreviated paths. For example, BRAKER might have problems with
--SAMTOOLS_PATH=./samtools/or--SAMTOOLS_PATH=~/samtools/. Please useSAMTOOLS_PATH=/full/absolute/path/to/samtools/, instead. This applies to all path specifications as command line options tobraker.pl. Relative paths and absolute paths will not pose problems if you export a bash variable, instead, or if you append the location of tools to your $PATH variable. -
GeneMark-ETP in BRAKER dies with '/scratch/11232323': No such file or directory.
This appears to be related to sorting large files, and it's a system configuration depending problem. Solve it with
export TMPDIR=/tmp/before calling BRAKER via Singularity. -
BRAKER cannot find the Augustus script XYZ...
Update Augustus from github with
git clone https://github.com/Gaius-Augustus/Augustus.git. Do not use Augustus from other sources. BRAKER is highly dependent on an up-to-date Augustus. Augustus releases happen rather rarely, updates to the Augustus scripts folder occur rather frequently. -
Does BRAKER depend on Python3?
It does. The python scripts employed by BRAKER are not compatible with Python2.
-
Why does BRAKER predict more genes than I expected?
If transposable elements (or similar) have not been masked appropriately, AUGUSTUS tends to predict those elements as protein coding genes. This can lead to a huge number genes. You can check whether this is the case for your project by BLASTing (or DIAMONDing) the predicted protein sequences against themselves (all vs. all) and counting how many of the proteins have a high number of high quality matches. You can use the output of this analysis to divide your gene set into two groups: the protein coding genes that you want to find and the repetitive elements that were additionally predicted.
-
I am running BRAKER in Anaconda and something fails...
Update AUGUSTUS and BRAKER from github with
git clone https://github.com/Gaius-Augustus/Augustus.gitandgit clone https://github.com/Gaius-Augustus/BRAKER.git. The Anaconda installation is great, but it relies on releases of AUGUSTUS and BRAKER - which are often lagging behind. Please use the current GitHub code, instead. -
Why and where is the GenomeThreader support gone?
BRAKER is a joint project between teams from University of Greifswald and Georgia Tech. While the group of Mark Bordovsky from Georgia Tech contributes GeneMark expertise, the group of Mario Stanke from University of Greifswald contributes AUGUSTUS expertise. Using GenomeThreader to build training genes for AUGUSTUS in BRAKER circumvents execution of GeneMark. Thus, the GenomeThreader mode is strictly speaking not part of the BRAKER project. The previous functionality of BRAKER with GenomeThreader has been moved to GALBA at https://github.com/Gaius-Augustus/GALBA. Note that GALBA has also undergone extension for using Miniprot instead of GenomeThreader.
-
My BRAKER gene set has too many BUSCO duplicates!
AUGUSTUS within BRAKER can predict alternative splicing isoforms. Also the merge of the AUGUSTUS and GeneMark gene set by TSEBRA within BRAKER may result in additional isoforms for a single gene. The BUSCO duplicates usually come from alternative splicing isoforms, i.e. they are expected.
-
Augustus and/or etraining within BRAKER complain that the file
aug_cmdln_parameters.jsonis missing. Even though I am using the latest Singularity container!BRAKER copies the AUGUSTUS_CONFIG_PATH folder to a writable location. In older versions of Augustus, that file was indeed not existing. If the local writable copy of a folder already exists, BRAKER will not re-copy it. Simply delete the old folder. (It is often
~/.augustus, so you can simply dorm -rf ~/.augustus; the folder might be residing in $PWD if your home directory was not writable). -
I sit behind a firewall, compleasm cannot download the BUSCO files, what can I do? See Issue #785 (comment)
Since BRAKER is a pipeline that calls several Bioinformatics tools, publication of results obtained by BRAKER requires that not only BRAKER is cited, but also the tools that are called by BRAKER. BRAKER will output a file what-to-cite.txt in the BRAKER working directory, informing you about which exact sources apply to your run.
-
Always cite:
-
Stanke, M., Diekhans, M., Baertsch, R. and Haussler, D. (2008). Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics, doi: 10.1093/bioinformatics/btn013.
-
Stanke. M., Schöffmann, O., Morgenstern, B. and Waack, S. (2006). Gene prediction in eukaryotes with a generalized hidden Markov model that uses hints from external sources. BMC Bioinformatics 7, 62.
-
-
If you provided any kind of evidence for BRAKER, cite:
- Gabriel, L., Bruna, T., Hoff, K. J., Borodovsky, M., Stanke, M. (2021) TSEBRA: transcript selector for BRAKER. BMC Bioinformatics 22, 1-12.
-
If you provided both short read RNA-Seq evidence and a large database of proteins, cite:
-
Gabriel, L., Bruna, T., Hoff, K. J., Ebel, M., Lomsadze, A., Borodovsky, M., Stanke, M. (2023). BRAKER3: Fully Automated Genome Annotation Using RNA-Seq and Protein Evidence with GeneMark-ETP, AUGUSTUS and TSEBRA. bioRxiV, doi: 10.1101/2023.06.10.54444910.1101/2023.01.01.474747.
-
Bruna, T., Lomsadze, A., Borodovsky, M. (2023). GeneMark-ETP: Automatic Gene Finding in Eukaryotic Genomes in Consistence with Extrinsic Data. bioRxiv, doi: 10.1101/2023.01.13.524024.
-
Kovaka, S., Zimin, A. V., Pertea, G. M., Razaghi, R., Salzberg, S. L., & Pertea, M. (2019). Transcriptome assembly from long-read RNA-seq alignments with StringTie2. Genome biology, 20(1):1-13.
-
Pertea, G., & Pertea, M. (2020). GFF utilities: GffRead and GffCompare. F1000Research, 9.
-
Quinlan, A. R. (2014). BEDTools: the Swiss‐army tool for genome feature analysis. Current protocols in bioinformatics, 47(1):11-12.
-
-
If the only source of evidence for BRAKER was a large database of protein sequences, cite:
- Bruna, T., Hoff, K.J., Lomsadze, A., Stanke, M., & Borodovsky, M. (2021). BRAKER2: Automatic Eukaryotic Genome Annotation with GeneMark-EP+ and AUGUSTUS Supported by a Protein Database. NAR Genomics and Bioinformatics 3(1):lqaa108, doi: 10.1093/nargab/lqaa108.
-
If the only source of evidence for BRAKER was RNA-Seq data, cite:
-
Hoff, K.J., Lange, S., Lomsadze, A., Borodovsky, M. and Stanke, M. (2016). BRAKER1: unsupervised RNA-Seq-based genome annotation with GeneMark-ET and AUGUSTUS. Bioinformatics, 32(5):767-769.
-
Lomsadze, A., Paul D.B., and Mark B. (2014) Integration of Mapped Rna-Seq Reads into Automatic Training of Eukaryotic Gene Finding Algorithm. Nucleic Acids Research 42(15): e119--e119
-
-
If you called BRAKER3 with an IsoSeq BAM file, or if you envoked the
--busco_lineageoption, cite:- Bruna, T., Gabriel, L., Hoff, K. J. (2024). Navigating Eukaryotic Genome Annotation Pipelines: A Route Map to BRAKER, Galba, and TSEBRA. arXiv, doi: 10.48550/arXiv.2403.19416 .
-
If you called BRAKER with the
--busco_lineageoption, in addition, cite:-
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V., & Zdobnov, E. M. (2015). BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics, 31(19), 3210-3212.
-
Li, H. (2023). Protein-to-genome alignment with miniprot. Bioinformatics, 39(1), btad014.
-
Huang, N., & Li, H. (2023). compleasm: a faster and more accurate reimplementation of BUSCO. Bioinformatics, 39(10), btad595.
-
-
If any kind of AUGUSTUS training was performed by BRAKER, check carefully whether you configured BRAKER to use NCBI BLAST or DIAMOND. One of them was used to filter out redundant training gene structures.
-
If you used NCBI BLAST, please cite:
-
Altschul, A.F., Gish, W., Miller, W., Myers, E.W. and Lipman, D.J. (1990). A basic local alignment search tool. J Mol Biol 215:403--410.
-
Camacho, C., Coulouris, G., Avagyan, V., Ma, N., Papadopoulos, J., Bealer, K., and Madden, T.L. (2009). Blast+: architecture and applications. BMC bioinformatics, 10(1):421.
-
-
If you used DIAMOND, please cite:
- Buchfink, B., Xie, C., Huson, D.H. (2015). Fast and sensitive protein alignment using DIAMOND. Nature Methods 12:59-60.
-
-
If BRAKER was executed with a genome file and no extrinsic evidence, cite, then GeneMark-ES was used, cite:
-
Lomsadze, A., Ter-Hovhannisyan, V., Chernoff, Y.O. and Borodovsky, M. (2005). Gene identification in novel eukaryotic genomes by self-training algorithm. Nucleic Acids Research, 33(20):6494--6506.
-
Ter-Hovhannisyan, V., Lomsadze, A., Chernoff, Y.O. and Borodovsky, M. (2008). Gene prediction in novel fungal genomes using an ab initio algorithm with unsupervised training. Genome research, pages gr--081612, 2008.
-
Hoff, K.J., Lomsadze, A., Borodovsky, M. and Stanke, M. (2019). Whole-Genome Annotation with BRAKER. Methods Mol Biol. 1962:65-95, doi: 10.1007/978-1-4939-9173-0_5.
-
-
If BRAKER was run with proteins as source of evidence, please cite all tools that are used by the ProtHint pipeline to generate hints:
-
Bruna, T., Lomsadze, A., & Borodovsky, M. (2020). GeneMark-EP+: eukaryotic gene prediction with self-training in the space of genes and proteins. NAR Genomics and Bioinformatics, 2(2), lqaa026.
-
Buchfink, B., Xie, C., Huson, D.H. (2015). Fast and sensitive protein alignment using DIAMOND. Nature Methods 12:59-60.
-
Lomsadze, A., Ter-Hovhannisyan, V., Chernoff, Y.O. and Borodovsky, M. (2005). Gene identification in novel eukaryotic genomes by self-training algorithm. Nucleic Acids Research, 33(20):6494--6506.
-
Iwata, H., and Gotoh, O. (2012). Benchmarking spliced alignment programs including Spaln2, an extended version of Spaln that incorporates additional species-specific features. Nucleic acids research, 40(20), e161-e161.
-
Gotoh, O., Morita, M., Nelson, D. R. (2014). Assessment and refinement of eukaryotic gene structure prediction with gene-structure-aware multiple protein sequence alignment. BMC bioinformatics, 15(1), 189.
-
-
If BRAKER was executed with RNA-Seq alignments in bam-format, then SAMtools was used, cite:
-
Li, H., Handsaker, B., Wysoker, A., Fennell, T., Ruan, J., Homer, N., Marth, G., Abecasis, G., Durbin, R.; 1000 Genome Project Data Processing Subgroup (2009). The Sequence Alignment/Map format and SAMtools. Bioinformatics, 25(16):2078-9.
-
Barnett, D.W., Garrison, E.K., Quinlan, A.R., Strömberg, M.P. and Marth G.T. (2011). BamTools: a C++ API and toolkit for analyzing and managing BAM files. Bioinformatics, 27(12):1691-2
-
-
If BRAKER downloaded RNA-Seq libraries from SRA using their IDs, cite SRA, SRA toolkit, and HISAT2:
-
Leinonen, R., Sugawara, H., Shumway, M., & International Nucleotide Sequence Database Collaboration. (2010). The sequence read archive. Nucleic acids research, 39(suppl_1), D19-D21.
-
SRA Toolkit Development Team (2020). SRA Toolkit. https://trace.ncbi.nlm.nih.gov/Traces/sra/sra.cgi?view=software.
-
Kim, D., Paggi, J. M., Park, C., Bennett, C., & Salzberg, S. L. (2019). Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nature biotechnology, 37(8):907-915.
-
-
If BRAKER was executed using RNA-Seq data in FASTQ format, cite HISAT2:
- Kim, D., Paggi, J. M., Park, C., Bennett, C., & Salzberg, S. L. (2019). Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nature biotechnology, 37(8):907-915.
-
If BRAKER called MakeHub for creating a track data hub for visualization of BRAKER results with the UCSC Genome Browser, cite:
- Hoff, K. J. (2019). MakeHub: fully automated generation of UCSC genome browser assembly hubs. Genomics, Proteomics and Bioinformatics, 17(5), 546-549.
-
If BRAKER called GUSHR for generating UTRs, cite:
-
Keilwagen, J., Hartung, F., Grau, J. (2019) GeMoMa: Homology-based gene prediction utilizing intron position conservation and RNA-seq data. Methods Mol Biol. 1962:161-177, doi: 10.1007/978-1-4939-9173-0_9.
-
Keilwagen, J., Wenk, M., Erickson, J.L., Schattat, M.H., Grau, J., Hartung F. (2016) Using intron position conservation for homology-based gene prediction. Nucleic Acids Research, 44(9):e89.
-
Keilwagen, J., Hartung, F., Paulini, M., Twardziok, S.O., Grau, J. (2018) Combining RNA-seq data and homology-based gene prediction for plants, animals and fungi. BMC Bioinformatics, 19(1):189.
-
All source code, i.e. scripts/*.pl or scripts/*.py are under the Artistic License (see https://www.opensource.org/licenses/artistic-license.php).
[F1] EX = ES/ET/EP/ETP, all available for download under the name GeneMark-ES/ET/EP ↩
[F2] Please use the latest version from the master branch of AUGUSTUS distributed by the original developers, it is available from github at https://github.com/Gaius-Augustus/Augustus. Problems have been reported from users that tried to run BRAKER with AUGUSTUS releases maintained by third parties, i.e. Bioconda. ↩
[F4] install with sudo apt-get install cpanminus ↩
[F6] The binary may e.g. reside in bamtools/build/src/toolkit ↩
[R0] Bruna, Tomas, Hoff, Katharina J., Lomsadze, Alexandre, Stanke, Mario, and Borodovsky, Mark. 2021. “BRAKER2: automatic eukaryotic genome annotation with GeneMark-EP+ and AUGUSTUS supported by a protein database." NAR Genomics and Bioinformatics 3(1):lqaa108.↩
[R1] Hoff, Katharina J, Simone Lange, Alexandre Lomsadze, Mark Borodovsky, and Mario Stanke. 2015. “BRAKER1: Unsupervised Rna-Seq-Based Genome Annotation with Genemark-et and Augustus.” Bioinformatics 32 (5). Oxford University Press: 767--69.↩
[R2] Lomsadze, Alexandre, Paul D Burns, and Mark Borodovsky. 2014. “Integration of Mapped Rna-Seq Reads into Automatic Training of Eukaryotic Gene Finding Algorithm.” Nucleic Acids Research 42 (15). Oxford University Press: e119--e119.↩
[R3] Stanke, Mario, Mark Diekhans, Robert Baertsch, and David Haussler. 2008. “Using Native and Syntenically Mapped cDNA Alignments to Improve de Novo Gene Finding.” Bioinformatics 24 (5). Oxford University Press: 637--44.↩
[R4] Stanke, Mario, Oliver Schöffmann, Burkhard Morgenstern, and Stephan Waack. 2006. “Gene Prediction in Eukaryotes with a Generalized Hidden Markov Model That Uses Hints from External Sources.” BMC Bioinformatics 7 (1). BioMed Central: 62.↩
[R5] Barnett, Derek W, Erik K Garrison, Aaron R Quinlan, Michael P Strömberg, and Gabor T Marth. 2011. “BamTools: A C++ Api and Toolkit for Analyzing and Managing Bam Files.” Bioinformatics 27 (12). Oxford University Press: 1691--2.↩
[R6] Li, Heng, Handsaker, Bob, Alec Wysoker, Tim Fennell, Jue Ruan, Nils Homer, Gabor Marth, Goncalo Abecasis, and Richard Durbin. 2009. “The Sequence Alignment/Map Format and Samtools.” Bioinformatics 25 (16). Oxford University Press: 2078--9.↩
[R7] Gremme, G. 2013. “Computational Gene Structure Prediction.” PhD thesis, Universität Hamburg.↩
[R8] Gotoh, Osamu. 2008a. “A Space-Efficient and Accurate Method for Mapping and Aligning cDNA Sequences onto Genomic Sequence.” Nucleic Acids Research 36 (8). Oxford University Press: 2630--8.↩
[R9] Iwata, Hiroaki, and Osamu Gotoh. 2012. “Benchmarking Spliced Alignment Programs Including Spaln2, an Extended Version of Spaln That Incorporates Additional Species-Specific Features.” Nucleic Acids Research 40 (20). Oxford University Press: e161--e161.↩
[R10] Osamu Gotoh. 2008b. “Direct Mapping and Alignment of Protein Sequences onto Genomic Sequence.” Bioinformatics 24 (21). Oxford University Press: 2438--44.↩
[R11] Slater, Guy St C, and Ewan Birney. 2005. “Automated Generation of Heuristics for Biological Sequence Comparison.” BMC Bioinformatics 6(1). BioMed Central: 31.↩
[R12] Altschul, S.F., W. Gish, W. Miller, E.W. Myers, and D.J. Lipman. 1990. “Basic Local Alignment Search Tool.” Journal of Molecular Biology 215:403--10.↩
[R13] Camacho, Christiam, et al. 2009. “BLAST+: architecture and applications.“ BMC Bioinformatics 1(1): 421.↩
[R14] Lomsadze, A., V. Ter-Hovhannisyan, Y.O. Chernoff, and M. Borodovsky. 2005. “Gene identification in novel eukaryotic genomes by self-training algorithm.” Nucleic Acids Research 33 (20): 6494--6506. doi:10.1093/nar/gki937.↩
[R15] Ter-Hovhannisyan, Vardges, Alexandre Lomsadze, Yury O Chernoff, and Mark Borodovsky. 2008. “Gene Prediction in Novel Fungal Genomes Using an Ab Initio Algorithm with Unsupervised Training.” Genome Research. Cold Spring Harbor Lab, gr--081612.↩
[R16] Hoff, K.J. 2019. MakeHub: Fully automated generation of UCSC Genome Browser Assembly Hubs. Genomics, Proteomics and Bioinformatics, in press, preprint on bioarXive, doi: https://doi.org/10.1101/550145.↩
[R17] Bruna, T., Lomsadze, A., & Borodovsky, M. 2020. GeneMark-EP+: eukaryotic gene prediction with self-training in the space of genes and proteins. NAR Genomics and Bioinformatics, 2(2), lqaa026. doi: https://doi.org/10.1093/nargab/lqaa026.↩
[R18] Kriventseva, E. V., Kuznetsov, D., Tegenfeldt, F., Manni, M., Dias, R., Simão, F. A., and Zdobnov, E. M. 2019. OrthoDB v10: sampling the diversity of animal, plant, fungal, protist, bacterial and viral genomes for evolutionary and functional annotations of orthologs. Nucleic Acids Research, 47(D1), D807-D811.↩
[R19] Keilwagen, J., Hartung, F., Grau, J. (2019) GeMoMa: Homology-based gene prediction utilizing intron position conservation and RNA-seq data. Methods Mol Biol. 1962:161-177, doi: 10.1007/978-1-4939-9173-0_9.↩
[R20] Keilwagen, J., Wenk, M., Erickson, J.L., Schattat, M.H., Grau, J., Hartung F. (2016) Using intron position conservation for homology-based gene prediction. Nucleic Acids Research, 44(9):e89.↩
[R21] Keilwagen, J., Hartung, F., Paulini, M., Twardziok, S.O., Grau, J. (2018) Combining RNA-seq data and homology-based gene prediction for plants, animals and fungi. BMC Bioinformatics, 19(1):189.↩
[R22] SRA Toolkit Development Team (2020). SRA Toolkit. https://trace.ncbi.nlm.nih.gov/Traces/sra/sra.cgi?view=software.[↩](#a22)
[R23] Kim, D., Paggi, J. M., Park, C., Bennett, C., & Salzberg, S. L. (2019). Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nature biotechnology, 37(8):907-915.↩
[R24] Quinlan, A. R. (2014). BEDTools: the Swiss‐army tool for genome feature analysis. Current protocols in bioinformatics, 47(1):11-12.↩
[R25] Kovaka, S., Zimin, A. V., Pertea, G. M., Razaghi, R., Salzberg, S. L., & Pertea, M. (2019). Transcriptome assembly from long-read RNA-seq alignments with StringTie2. Genome biology, 20(1):1-13.↩
[R26] Pertea, G., & Pertea, M. (2020). GFF utilities: GffRead and GffCompare. F1000Research, 9.↩
[R27] Huang, N., & Li, H. (2023). compleasm: a faster and more accurate reimplementation of BUSCO. Bioinformatics, 39(10), btad595.↩
[R28] Bruna, T., Gabriel, L. & Hoff, K. J. (2024). Navigating Eukaryotic Genome Annotation Pipelines: A Route Map to BRAKER, Galba, and TSEBRA. arXiv, https://doi.org/10.48550/arXiv.2403.19416 .↩
![braker2-team-2[fig10]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/mario.png)
![braker2-team-1[fig11]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/alex-katharina-tomas.png)
![braker2-team-3[fig12]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/lars.jpg)
![braker2-team-4[fig13]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/mark.png)
![braker2-main-a[fig1]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/braker-es.png)
![braker2-main-b[fig2]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/braker1.png)
![braker2-main-c[fig3]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/braker2_ep.png)
![braker3-main-a[fig4]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/braker3_etp.png)
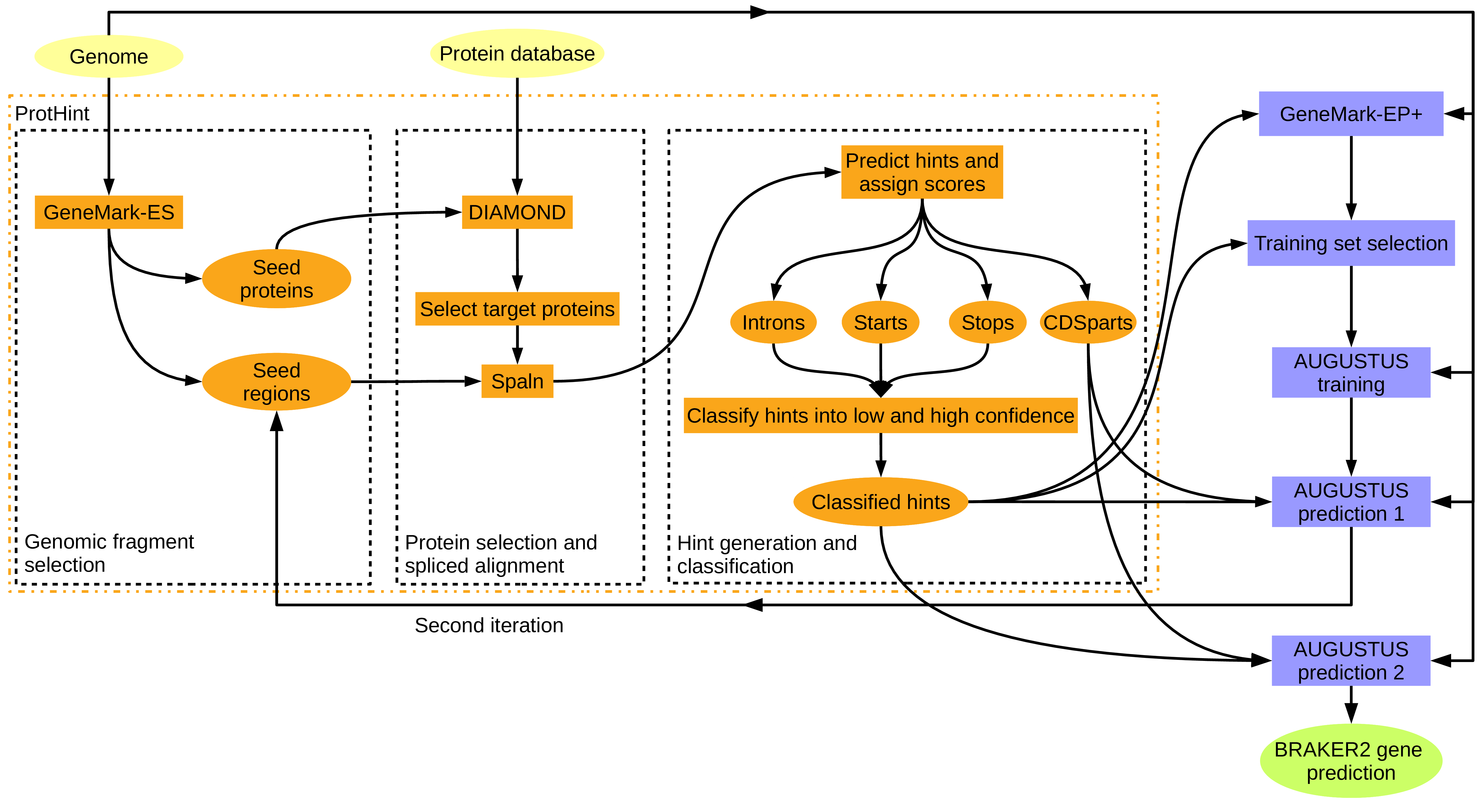
![best_by_busco[fig14]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/best_by_compleasm.jpg)
![braker-intercept[fig8]](https://github.com/Gaius-Augustus/BRAKER/raw/master/docs/figs/braker-intercept.png)