Cefmenoxime
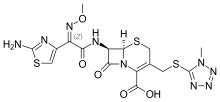 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intramuscular, intravenous |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | 100% (given IM) |
| Protein binding | 50% to 70% |
| Metabolism | Negligible |
| Elimination half-life | 1 hour |
| Excretion | Kidney, unchanged |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C16H17N9O5S3 |
| Molar mass | 511.55 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Cefmenoxime is a third-generation cephalosporin antibiotic.[1]
Synthesis
The alkylation of ethyl 2-hydroxyimino-3-oxobutanoate (1) with dimethylsulfate gives ethyl (2Z)-2-methoxyimino-3-oxo-butanoate (2). Halogenation with molecular bromine leads to ethyl 4-bromo-2-methoxyimino-3-oxobutanoate (3). Treatment with thiourea gives ethyl (Z)-2-(2-amino-4-thiazolyl)-2-methoxyiminoacetate (4) which is reacted with chloroacetyl chloride to give the amide (5). Saponification with potassium hydroxide gives (6) which is halogenated with phosphorus pentachloride to (7). Amide formation with the cephalosporin intermediate (8) then gives (9). Removal of the protecting group with benzyltriethylammonium bromide yields (10). The tert-butyl ester was deprotected with trifluoroacetic acid to give (11). Lastly, thioether formation with 5-mercapto-1-methyltetrazole (12) completes the synthesis of cefmenoxime.[2][3][4][5]
References
- ^ Campoli-Richards DM, Todd PA (August 1987). "Cefmenoxime. A review of its antibacterial activity, pharmacokinetic properties and therapeutic use". Drugs. 34 (2): 188–221. doi:10.2165/00003495-198734020-00002. PMID 3304966.
- ^ US 4098888, Ochiai M, Okada T, Aki O, Morimoto A, Kawakita K, Matsushita Y, "Thiazolylacetamido cephalosporin type compounds", issued 7 April 1978, assigned to Takeda Pharmaceutical Co Ltd.
- ^ Ochiai M, Aki O, Morimoto A, Okada T, Matsushita Y (November 1977). "New cephalosporin derivatives with high antibacterial activities". Chemical & Pharmaceutical Bulletin. 25 (11): 3115–3117. doi:10.1248/cpb.25.3115. PMID 603968.
- ^ Ochiai M, Morimoto A, Miyawaki T, Matsushita Y, Okada T, Natsugari H, et al. (February 1981). "Synthesis and structure-activity relationships of 7 beta-[2-(2-aminothiazol-4-yl)acetamido]cephalosporin derivatives. V. Synthesis and antibacterial activity of 7 beta-[2-(2-aminothiazol-4-yl)-2-methoxyiminoacetamido]-cephalosporin derivates and related compounds". The Journal of Antibiotics. 34 (2): 171–185. doi:10.7164/antibiotics.34.171. PMID 6271716.
- ^ Ochiai M, Morimoto A, Miyawaki T (February 1981). "Synthesis and structure-activity relationships of 7 beta-[2-(2-aminothiazol-4-yl)acetamido]cephalosporin derivatives. VI. Alternative syntheses of 7 beta-[2-(2-aminothiazol-4-yl)-(Z)-2-methoxyiminoacetamido]cephalosporin derivatives". The Journal of Antibiotics. 34 (2): 186–192. doi:10.7164/antibiotics.34.186. PMID 6271717.
Further reading
- Yokota N, Koguchi M, Suzuki Y, Fukayama S, Ishihara R, Deguchi K, et al. (May 1995). "[Antibacterial activities of cefmenoxime against recent fresh clinical isolates from patients in sinusitis]". The Japanese Journal of Antibiotics. 48 (5): 602–609. PMID 7637194.
- Paladino JA, Fell RE (March 1994). "Pharmacoeconomic analysis of cefmenoxime dual individualization in the treatment of nosocomial pneumonia". The Annals of Pharmacotherapy. 28 (3): 384–389. doi:10.1177/106002809402800316. PMID 8193431. S2CID 29444681.
- Duncker GI, Reich U, Krausse R (1994). "Cefmenoxime in corneal organ culture". Ophthalmologica. Journal International d'Ophtalmologie. International Journal of Ophthalmology. Zeitschrift Fur Augenheilkunde. 208 (5): 262–266. doi:10.1159/000310505. PMID 7816419.
External links
- Diseases Database (DDB): 30892

